-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(8): 2538-2542
doi:10.5923/j.ajmms.20251508.25
Received: Jun. 27, 2025; Accepted: Jul. 20, 2025; Published: Aug. 4, 2025

Impact of Autoimmune Thyroid Disease on Ovarian Reserve in Reproductive-Age Women with Hypothyroidism
Khalimova Zamira Yusufovna1, Gafurova Dildorakhon Kamalidinovna2
1Professor, Deputy Director for Scientific Affairs of the Republican Specialized Scientific and Practical Medical Center of Endocrinology named after Academician Y.Kh. Turakulov of the Ministry of Health of the Republic of Uzbekistan
2PHD Student, Department of Hospital Therapy and Endocrinology of Andijan State Medical Institute, Uzbekistan
Correspondence to: Gafurova Dildorakhon Kamalidinovna, PHD Student, Department of Hospital Therapy and Endocrinology of Andijan State Medical Institute, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The aim of the study was to evaluate the relationship between autoimmune thyroid disease and the state of ovarian reserve in women of reproductive age with hypothyroidism. In a retrospective study, we analyzed the data of 50 patients aged 16-49 years, divided into four quartiles by the level of anti-Müllerian hormone (AMH). The control group included 20 healthy women of comparable age. Ovarian reserve was assessed by the number of antral follicles (AF) using transvaginal ultrasound. Statistical analysis did not reveal significant differences in anthropometric parameters and hormonal status of the thyroid gland between quartiles. However, women with the lowest AMH level had a statistically significant increase in thyroid peroxidase antibodies (TPO AT) with a tendency to increase with age, which indicates an increased risk of hypothyroidism progression and a decrease in ovarian reserve in this group. The obtained data confirm the need to monitor autoimmune processes of the thyroid gland when assessing the reproductive potential of women with hypothyroidism.
Keywords: Fertility, Hypothyroidism, Thyroid autoimmunity, Ovarian reserve
Cite this paper: Khalimova Zamira Yusufovna, Gafurova Dildorakhon Kamalidinovna, Impact of Autoimmune Thyroid Disease on Ovarian Reserve in Reproductive-Age Women with Hypothyroidism, American Journal of Medicine and Medical Sciences, Vol. 15 No. 8, 2025, pp. 2538-2542. doi: 10.5923/j.ajmms.20251508.25.
Article Outline
1. Introduction
- According to researchers, thyroid hormones (TH) are involved in the normal growth, development and function of many organs, including the gonads. Their circulating levels are tightly regulated by feedback mechanisms acting along the hypothalamic-pituitary-thyroid (HPT) axis. Moreover, cells and tissues can locally tune TH signaling by regulating the life stage-specific expression of iodothyronine deiodinases (DIO, enzymes involved in TH metabolism), TH transporters and, finally, TH receptors (TR). Their local modulation represents an additional and/or alternative mechanism for maintaining the peripheral T3 quota necessary for physiological processes, regardless of fluctuations in circulating TH levels [1]. Thus, organ/tissue-specific TH signaling is a result of thyroid hormone synthesis and their peripheral metabolism.Autoimmunity is believed to be involved in the pathogenesis of 4–30% of cases of premature ovarian failure (POF) [1]. Alterations in the immune system can cause ovarian dysfunction or even removal of follicles, causing a decrease in ovarian reserve [2]. The most common autoimmune disease in women of reproductive age is thyroid dysfunction with a prevalence of 5–20% [3]. Altered thyroid function affects a wide range of functions, including growth, development, and metabolism [4]. Hypothyroidism occurs either due to thyroid autoimmune disease (TAID) or due to insufficient iodine intake [5]. It is well known that prolonged reduction in thyroid hormone concentrations results in a wide range of reproductive changes, including abnormal folliculogenesis, changes in ovulation and fertilization rates, and ovarian failure [6]. There have been reports of changes in the menstrual cycle in patients with Hashimoto's thyroiditis [7], and women with thyroid-related diseases have a higher incidence of infertility compared to healthy individuals [8].The presence of thyroid hormone receptors in the ovaries suggests that thyroid hormones are important for ovarian function [9]. Currently, the physiopathology underlying hypothyroidism for follicular development and ovarian follicular reserve is unclear, and studies using animal models are contradictory. For example, some reports claim that low thyroid hormone levels cause a significant decrease in basal luteinizing hormone (LH) release, leading to ovarian atrophy [10] or prolonged periods of vaginal diestrus [11]. Other reports show small changes in LH and follicle-stimulating hormone (FSH) release and the presence of mature follicles and corpus luteum [12]. In humans, some studies support an association between hypothyroidism and decreased ovarian reserve (DOR) [13]. UOR is defined as a reduced response to ovarian stimulation in women of reproductive age with regular menstrual cycles compared to women of the same age.Ovarian reserve (OR) can be accurately assessed by measuring anti-Müllerian hormone (AMH), which is secreted by granulosa cells of the ovarian follicles [14] and whose levels are stable throughout the cycle. There is preliminary evidence that thyroid disease is associated with low OR, and there is a possible association between TAID and AMH [15]. In addition, the prevalence of AMH in patients with low thyroid hormone levels is not fully established.Accelerated decline in fertility and early menopause are associated with loss of functional ovarian reserve (FOR), leading to premature ovarian ageing. This decline is often asymptomatic, and the underlying mechanisms are still poorly understood. Genetic and environmental factors contribute to this phenomenon. Both factors influence the number of follicles formed in early life and the hormonal balance required for their maintenance and maturation during reproductive age [17]. Some epidemiological studies, although contradictory, indicate a significantly higher prevalence of hypothyroidism, both overt and subclinical, in women with a genetic cause of decreased ovarian reserve (OR) [18].TGs likely play a role in the onset and progression of premature ovarian failure (POF) due to their cross-talk with other hormonal pathways (e.g., estrogen, prolactin, IGF-1, and GnRH) to impair folliculogenesis. The prevalence of hypothyroidism ranges from 0.3% to 4.3% in adult women and is often associated with the presence of thyroid antibodies (e.g., AtTPO, AtTG). Although debated, the association between increased infertility/ovarian dysfunction with hypothyroidism/thyroid antibodies has been studied, suggesting an association of high thyroid antibody levels and several reproductive dysfunctions including POF [19–21].Ovarian reserve function can reflect endocrine function and fertility in women and is often age-related and will gradually decline. Diminished ovarian reserve (DOR) is defined as decreased reproductive potential with a poor response to ovarian stimulation. Some young women still have DOR, but the cause and mechanism are unknown. Previous studies have shown that TSH levels in infertile women were higher than those in normal fertile women. Moreover, elevated serum TSH levels were associated with UOR in infertile patients [22].The above formed the basis for this study.The aim of the study is to evaluate the relationship between autoimmune thyroid disease and the state of ovarian reserve in women of reproductive age with hypothyroidism.
2. Material and Methods of Research
- The study retrospectively analyzed the results of examination and treatment of 50 women of reproductive age (16-49 years) with hypothyroidism. Participants were divided into four age quartiles based on AMH levels (Q1–Q4):Q1 – the lowest quartile, n=12Q2 – middle quartile, n=11Q3 – high quartile, n=13Q4 – the highest, n=1420 healthy women of similar age formed the control group.Consultative reception and examination of women with reproductive dysfunction were carried out at the clinical sites of the endocrinology departments of the Andijan State Medical Institute. Inclusion criteria: normal sexual life without contraception and absence of pregnancy for more than 12 months.Exclusion criteria: (a) patients with polycystic ovary syndrome; (b) patients with a previous history of thyroid disease, or the presence of goiter and/or nodules, or thyroid surgery; (c) history of hypothalamic and pituitary diseases; (d) patients with autoimmune diseases, diabetes and adrenal dysfunction; (e) patients with a history of diseases and chromosomal abnormalities; (f) factors that adversely affect thyroid hormone and ovarian function.We measured serum thyroid hormone and AMH levels.Serum luteinizing hormone, follicle-stimulating hormone, estradiol, progesterone, prolactin, and testosterone levels were analyzed on days 2–5 of the menstrual cycle to screen infertile patients.Using all available data on ovarian reserve, we selected all patients with ovarian reserve defined by the following criteria: (1) a woman with any of the risk factors for poor ovarian response and/or (2) an abnormal ovarian reserve test (i.e., antral follicle count (AFC) <5-7 follicles or AMH <0.5-1.1 ng/mL).Antral follicle (AF) size and number were measured using a Philips HD9 transvaginal ultrasound probe with a 12.0 MHz vaginal probe (Philips Healthcare, Amsterdam, The Netherlands).Results were presented as mean ± standard deviation for normally distributed variables and as median and interquartile range for non-normally distributed variables. Correlation analysis was performed using the Pearson method.
3. Research Results
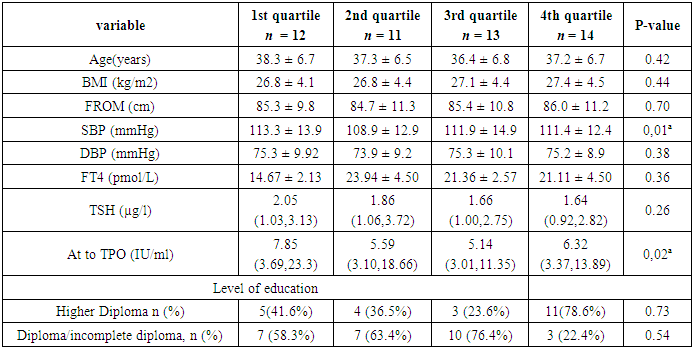
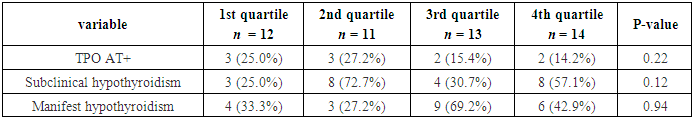
- At baseline, there were no statistically significant differences in anthropometric measurements, diastolic blood pressure, parity, educational level, and thyroid hormone levels among participants in different quartiles (Table 1).
|
|
|
4. Discussion of Results
- Conflicting reports suggest that both hypothyroidism and hyperthyroidism, assessed by circulating T4, T3, and thyroid stimulating hormone (TSH), may affect ovarian reserve functionality, determining infertility. The relationship between ovarian T3 levels and functional ovarian reserve (FOR) is poorly understood, although alterations in local T3 metabolism and signaling have been associated with dysfunctions of several organs [16]. The relationship between ovarian T3 levels and functional ovarian reserve (FOR) is poorly understood, although alterations in local T3 metabolism and signaling have been associated with dysfunctions of several organs.Taken together, the review data suggest that abnormal TG levels, particularly during puberty and the reproductive years, may lead to ovarian dysfunction throughout life. This may be mediated by a variety of mechanisms, ranging from alterations in circulating TG levels and/or their peripheral metabolism/signaling to their interactions with signaling pathways that play a key role in maintaining GF. Well-documented studies indicate that thyroid dysfunction, particularly early in life, may be associated with subfertility or infertility, menstrual/estrus irregularities, and anovulation. Well-documented studies indicate that thyroid dysfunction, particularly early in life, may be associated with subfertility or infertility, menstrual/estrus irregularities, and anovulation [23].There are several studies with conflicting results regarding the association between hypothyroidism and thyroid autoimmunity with low ovarian reserve [23]; however, the impact of low ovarian reserve on thyroid function has been largely unexplored so far.
5. Conclusions
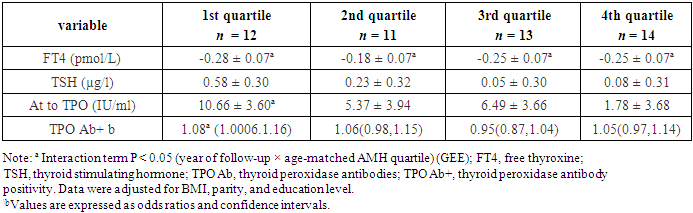
- 1. According to the review, TG levels, especially during puberty and fertile age, may lead to ovarian dysfunction throughout life. 2. Women with the lowest ovarian reserves had higher levels of TPO antibodies, with a positive trend in this antibody over time compared to other quartiles, indicating that this group may be at higher risk of developing hypothyroidism over time.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML