-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(8): 2532-2537
doi:10.5923/j.ajmms.20251508.24
Received: Jul. 9, 2025; Accepted: Jul. 30, 2025; Published: Aug. 4, 2025

Comparative Study of GSTP1 Gene Polymorphism Indices (IIe105Val) in Pregnant Women with Different Forms of Preeclampsia
Negmatshaeva Mubina Sinobek kizi1, Akhmedov Farkhod Kakhramonovich2, Akhmadzhonova Gulnoza Murodovna3, Negmatshaeva Habiba Nabievna4
1Andijan State Medical Institute, Andijan, Uzbekistan
2Doctor of Medical Sciences, Associate Professor, Bukhara State Medical Institute, Bukhara, Uzbekistan
3Doctor of Medical Sciences, Associate Professor, Andijan State Medical Institute, Andijan, Uzbekistan
4Candidate of Medical Sciences Andijan State Medical Institute, Andijan, Uzbekistan
Correspondence to: Akhmedov Farkhod Kakhramonovich, Doctor of Medical Sciences, Associate Professor, Bukhara State Medical Institute, Bukhara, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background. Genetic factors for preeclampsia are being actively studied worldwide, but results are often inconsistent and not confirmed by repeated studies in different populations, highlighting the need for further study of the genetic causes of this pregnancy complication. Aim. To study the indicators of GSTP1 gene polymorphism (IIe105Val) in pregnant women with preeclampsia. Materials and methods. Genetic studies were conducted and GSTP1 gene polymorphisms (IIe105Val) were studied in 87 pregnant women of the main group and 90 women with physiological pregnancy who participated in our study. In turn, we divided 87 pregnant women of the main group into 2 groups. More precisely, Group I included 47 pregnant women with severe preeclampsia. Group II consisted of 40 pregnant women with moderate preeclampsia. Results. In our study, we analyzed the prognostic significance of the studied variants of polymorphism genes for the development of preeclampsia in pregnant women complicated by the preeclampsia group in the ethnic Uzbek population Thus, our study found a positive correlation between interleukin-2 (IL-2) and fetal growth retardation. It also showed that IL-2 is a reliable marker for fetal growth retardation alone. Thus, the GSTP1 gene polymorphism (IIe105Val) in the mutant Val/Val genotype had an AUC of more than 0.71, which proves its important role in the risk of developing preeclampsia. Conclusions. Thus, some polymorphisms of the GSTP1 gene (IIe105Val) may be risk factors for the development of preeclampsia and, as an independent genetic marker, may increase the risk of developing preeclampsia in pregnant women.
Keywords: GSTP1, Preeclampsia, IIe105Val, Pregnant women, Hypertension in Pregnancy
Cite this paper: Negmatshaeva Mubina Sinobek kizi, Akhmedov Farkhod Kakhramonovich, Akhmadzhonova Gulnoza Murodovna, Negmatshaeva Habiba Nabievna, Comparative Study of GSTP1 Gene Polymorphism Indices (IIe105Val) in Pregnant Women with Different Forms of Preeclampsia, American Journal of Medicine and Medical Sciences, Vol. 15 No. 8, 2025, pp. 2532-2537. doi: 10.5923/j.ajmms.20251508.24.
Article Outline
1. Introduction
- Preeclampsia (PE) is a systemic disorder occurring during pregnancy and is a major cause of maternal, fetal and neonatal mortality, particularly in low- and middle-income countries [1,2,3,4,17,18,19,20]. PE is characterized by the development or progression of hypertension after 20 weeks of gestation, accompanied by proteinuria or evidence of end-organ damage such as neurological disorders, pulmonary edema, hematological abnormalities, acute kidney injury, liver injury or uteroplacental dysfunction, as defined by the International Society for the Study of Hypertension in Pregnancy (ISSHP) [5,6,7,8,9,21,22,23,24,25]. The UK National Institute for Health and Care Excellence (NICE) defines PE as hypertension after 20 weeks of gestation with proteinuria or organ dysfunction, including renal failure, liver dysfunction, neurological or hematological complications, and uteroplacental dysfunction [10,11,12,13,14,15,16,26,27,28]. The American College of Obstetricians and Gynecologists (ACOG) does not consider proteinuria a mandatory criterion for the diagnosis of PE, emphasizing the importance of elevated blood pressure in combination with complications such as thrombocytopenia, renal failure, liver dysfunction, pulmonary edema, or cerebral/visual symptoms [Ng, K.W., Chaturvedi, N., 2024].The genetic aspects of preeclampsia are actively studied worldwide, but the research results are often contradictory and not confirmed in different populations, which indicates the need for further study of the genetic factors of this complication [Abramova M.Yu., Churnosov M.I., 2022]. Studies have shown that polymorphism of the glutathione S-transferase pi-1 (GSTP1) gene, as well as the superoxide dismutase 2 (SOD2) and glutathione S-transferase M1 (GSTM1) genes, can contribute to endothelial dysfunction and placental hypoxia, playing a significant role in the pathogenesis of preeclampsia [29,30,31,32,33,34]. In this regard, we decided to investigate the role of the GSTP1 gene polymorphism (IIe105Val) in the Uzbek population.Objective of the study: To study the indicators of GSTP1 gene polymorphism (IIe105Val) in pregnant women with preeclampsia.
2. Materials and Methods
- All patients underwent a full clinical examination in the conditions of the Municipal Maternity Hospital No. 2 of the city of Andijan, Andijan region. The diagnosis of preeclampsia was established on the basis of complaints, anamnesis and additional examination methods, and the severity was assessed according to the WHO classification of hypertensive conditions in pregnancy (ICD-10) and the national clinical protocol. In all cases, preeclampsia was combined with various extragenital diseases.Inclusion criteria:- Blood pressure ≥130/90 mmHg;- Diastolic pressure >90 mm Hg;- Proteinuria >0.033 g/l.Patients with isolated oedema syndrome were not included, since oedema is not considered as a criterion for assessing the severity of preeclampsia in most countries.The main group consisted of 87 pregnant women aged 18–40 years, the control group consisted of 90 healthy women with normal pregnancy, comparable in gestational age and intergenetic interval. Genetic studies of the GSTP1 gene polymorphism (IIe105Val) were performed in 87 women of the main group and 90 women of the control group. The main group was divided into two subgroups depending on the severity of preeclampsia: Subgroup I included 47 women with severe preeclampsia, subgroup II — 40 women with moderate preeclampsia.
3. Research Methods
- Clinical and laboratory studies included a complete blood count and urine analysis, determination of hematocrit, protein levels in single and daily urine, microalbuminuria, blood enzymes, creatinine, total protein, urea, as well as genetic analysis of the GSTP1 gene polymorphism (IIe105Val).The data were processed on a Pentium-IV computer using Microsoft Office Excel-2012 with statistical analysis functions. The methods of variation parametric and nonparametric statistics were used, including calculation of the arithmetic mean (M), standard deviation (σ), standard error of the mean (m), relative values (%, frequency). The statistical significance of differences in mean values was determined by the Student's criterion (t) with the calculation of the probability of error (P), taking into account the normality of distribution (kurtosis criterion) and equality of variances (Fisher's F-criterion). The significance level was accepted at P<0.05. For qualitative values, the χ² (chi-square) criterion was used.
4. Results
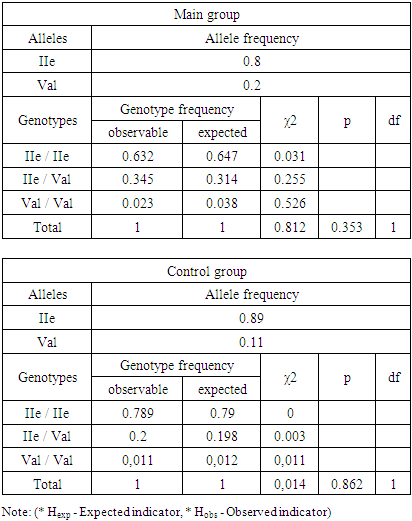
- The distribution of allele and genotype frequencies of the GSTP1 gene polymorphism (IIe105Val) was studied among pregnant women with moderate and severe preeclampsia, as well as with normal pregnancy. In the main group, which included women with moderate and severe preeclampsia, the homozygous genotype IIe/IIe was 63.22%, the heterozygous genotype IIe/Val was 34.48%, and the homozygous mutant genotype Val/Val was 2.3%.
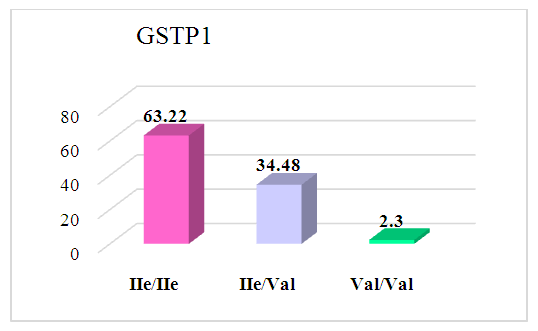 | Figure 1. The result of the distribution of GSTP1 genotypes in the main group (n = 87) |
|
|
|
5. Discussion
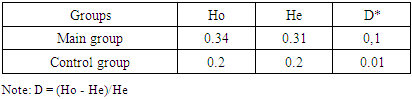
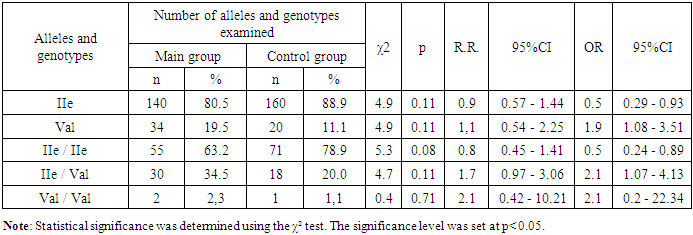
- Data analysis showed that heterozygous IIe/Val genotype was associated with an increased risk of preeclampsia (OR=1.5; 95% CI: 0.67–3.47). Comparison of genotype and allele frequencies of the IIe105Val locus of the GSTP1 gene between the second subgroup (n=40) and the control group is presented below. In the second subgroup (pregnant women with mild preeclampsia), the proportion of the IIe allele was 73.8%, and the Val allele was 26.3%, while in the control group it was 88.9% and 11.1%, respectively. The IIe allele was predominant in the control group (χ²=9.6; p=0.01; RR=0.8; 95% CI: 0.4–1.71; OR=0.4; 95% CI: 0.18–0.68). The frequency of the mutant Val allele was higher in the second subgroup (χ²=9.6; p=0.01; RR=1.2; 95% CI: 0.64–2.27; OR=2.8; 95% CI: 1.47–5.53).The homozygous IIe/IIe genotype was more common in the control group (78.9% versus 52.5% in the second subgroup; χ²=9.3; p=0.01; RR=0.7; 95% CI: 0.25–1.75; OR=0.3; 95% CI: 0.14–0.65). The heterozygous IIe/Val genotype was more common in the second subgroup (42.5% versus 20.0% in the control group; χ²=7.1; p=0.01; RR=2.1; 95% CI: 0.81–5.58; OR=3.0; 95% CI: 1.33–6.55). The homozygous Val/Val genotype was more often detected in the second subgroup (5.0% versus 1.0% in the control group; χ²=1.9; p=0.27; RR=4.5; 95% CI: 0.86–23.49; OR=4.7; 95% CI: 0.51–43.16).Comparison of the subgroups revealed that the frequency of the IIe allele was higher in the first subgroup (86.2%) compared to the second (73.8%; χ²=4.2; p=0.10; RR=1.2; 95% CI: 0.48–2.82; OR=2.2; 95% CI: 1.04–4.73). The Val allele, on the contrary, prevailed in the second subgroup (26.3% versus 13.8%; χ²=4.2; p=0.10; RR=0.9; 95% CI: 0.45–1.63; OR=1.5; 95% CI: 0.21–0.96). The homozygous IIe/IIe genotype was more common in the first subgroup (72.3% versus 52.8% in the second; χ²=3.7; p=0.21; RR=1.4; 95% CI: 0.55–3.44; OR=2.4; 95% CI: 0.98–5.72). The heterozygous IIe/Val genotype was more common in the second subgroup (42.5% versus 27.7%; χ²=2.1; p=0.30; RR=1.7; 95% CI: 0.26–1.61; OR=1.5; 95% CI: 0.21–1.26). The homozygous Val/Val genotype was absent in the first subgroup, but was found in the second.
6. Prognostic Significance
- Evaluation of the prognostic value of the IIe105Val polymorphism of the GSTP1 gene showed that the mutant Val allele can increase the risk of preeclampsia. In the main group, AUC was 0.65 (SE=0.89; SP=0.58; OR=1.94; 95% CI: 1.08–3.5; p=0.37). In the first subgroup, AUC=0.62 (SE=0.89; SP=0.64; OR=1.28; 95% CI: 0.61–2.68; p=0.61), in the second — AUC=0.68 (SE=0.89; SP=0.76; OR=2.85; 95% CI: 1.47–5.54; p=0.49). For the mutant Val/Val genotype in the main group, AUC=0.71 (SE=0.72; SP=0.99; OR=2.09; 95% CI: 0.2–22.15; p=0.49), in the first subgroup, AUC=0.72 (SE=0.75; SP=0.99; OR=4.68; 95% CI: 0.51–43.06; p=0.66). The average AUC value (0.65–0.71) indicates a high prognostic value of the mutant Val allele and the Val/Val genotype as markers of preeclampsia risk.
7. Conclusions
- The study revealed that in the Uzbek population, the mutant Val allele and the homozygous Val/Val genotype of the GSTP1 gene (IIe105Val) are associated with an increased risk of preeclampsia. The IIe allele and IIe/IIe genotype probably do not play a key role in the development of preeclampsia, but may have a protective effect. Polymorphism of the GSTP1 gene (IIe105Val) can be used as a genetic marker of preeclampsia risk, especially in the case of the mutant Val allele and Val/Val genotype.Conclusions. Our study revealed that in the Uzbek population of pregnant women with preeclampsia, the mutant homozygous Val/Val genotype of the GSTP1 gene polymorphism (IIe105Val) was more common than in the control group. Statistical data, including OR and AUC, indicate that the mutant Val allele and the Val/Val genotype of the GSTP1 gene are associated with an increased risk of preeclampsia. At the same time, the IIe allele and the homozygous IIe/IIe genotype do not seem to play a significant role in the development of preeclampsia, although the IIe allele may have a protective effect. Nevertheless, preeclampsia was observed in women with the mutant Val allele and the Val/Val genotype. Thus, the GSTP1 gene polymorphism (IIe105Val) may act as a genetic marker that increases the risk of preeclampsia in pregnant women.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML