-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(8): 2464-2469
doi:10.5923/j.ajmms.20251508.08
Received: Jun. 22, 2025; Accepted: Jul. 20, 2025; Published: Aug. 2, 2025

Features of Target Organ Damage in Salt-Sensitive Arterial Hypertension in the Population of the Aral Sea Region
Ataniyazov Khurshid Khamidovich1, Khamidullaeva Gulnoz Abdusattarovna2
1PhD., Head of the Department of Arterial Hypertension, Regional Branch of the Republik of Karakalpakstan of the Republican Specialized Scientific and Practical Medical Center for Cardiology of the Republic of Uzbekistan
2DSc., Associate Professor, Head of the Arterial Hypertension, Department of the Republican Specialized Cardiology Scientific and Practical Medical Center, Tashkent, Uzbekistan
Correspondence to: Ataniyazov Khurshid Khamidovich, PhD., Head of the Department of Arterial Hypertension, Regional Branch of the Republik of Karakalpakstan of the Republican Specialized Scientific and Practical Medical Center for Cardiology of the Republic of Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The dry climate of the Aral Sea region, salt and sandy sea winds have a negative effect on the human body and cause an increase in blood pressure, making it difficult to control and causing salt-sensitive arterial hypertension. Background: The aim of the work was to study the characteristics of target organ damage in patients with salt-sensitive arterial hypertension in the practice of primary health care institutions in the Aral Sea region. Methods: The study included 1021 patients aged 40–70 years who were diagnosed with hypertension in district family clinics. The results of laboratory-instrumental analysis of all patients included in the study were analyzed. Results: The results of our study showed that the development of target organ damage in patients with salt-sensitive arterial hypertension in the Aral Sea region is directly related to the amount of table salt consumed. Conclusions: In patients with salt-sensitive arterial hypertension, target organ damage was clearly expressed, dyslipidemia was high, and glucose tolerance was impaired.
Keywords: Blood pressure 1, Health care 2, Arterial hypertension 3, Target organs 4
Cite this paper: Ataniyazov Khurshid Khamidovich, Khamidullaeva Gulnoz Abdusattarovna, Features of Target Organ Damage in Salt-Sensitive Arterial Hypertension in the Population of the Aral Sea Region, American Journal of Medicine and Medical Sciences, Vol. 15 No. 8, 2025, pp. 2464-2469. doi: 10.5923/j.ajmms.20251508.08.
1. Introduction
- Arterial hypertension (AH) is one of the main risk factors for cardiovascular diseases, including myocardial infarction and stroke. According to the World Health Organization, AH occurs in 36-45% of the working population, depending on age, gender, and social status [1]. AH occurs in 80% of patients with coronary heart disease, diabetes, and stroke. The problem of AH remains relevant in economically developed countries. To reduce the complications of AH, it is necessary to prevent cardiovascular diseases from an early age in the population. The relevance of the problem of AH lies in the damage to the target organs in the disease. To determine the level of cardiovascular risk factors in AH, it is necessary to study the level of arterial pressure, target organ damage, and the presence or absence of comorbidities [3]. In the treatment of AH, it is very important to change the lifestyle of patients, regardless of their age, gender, blood pressure, and cardiovascular risk. The sooner hypertensive patients follow a healthy lifestyle, the more effective it will be. In the development of AH, the role of the accumulation of sodium and water in the body, the sympathetic nervous system, and the activation of the renin-angiotensin-aldosterone system (RAAS) has been studied in scientific studies. Epidemiological and cohort studies revealed that the population consumes table salt 2-3 times more than the norm. High salt intake increases the risk of cardiovascular disease by 14% and stroke by 23% [2]. Salt-sensitive AH is a general population problem in economically developed countries. Variants of salt-sensitive AH include metabolic syndrome, diabetes, chronic kidney disease, and isolated systolic AH in the elderly. Excessive table salt consumption causes AH equally in population with normal and above normal body mass index [4]. Excess sodium in the body leads to myocardial left ventricular hypertrophy [5]. In diabetic nephropathy, salt sensitivity further increases renal protein excretion [6]. In AH, the heart, blood vessels, kidneys, brain, and retina are primarily damaged. Left ventricular hypertrophy is the most commonly affected target organ in AH.According to the results of the Framing study, left ventricular hypertrophy is a major risk factor for heart attack and stroke [7]. The LIFE randomized trial demonstrated that left ventricular hypertrophy is a more important predictor of cardiovascular complications than hypertensive syndrome [8]. Left ventricular hypertrophy is a risk factor for cardiovascular mortality in ischemic heart disease and stroke [9,10]. Patients with left ventricular hypertrophy are 10 times more likely to develop heart failure [11]. Also, the geometric model of the left ventricle depends on the relative thickness of the walls of the left ventricle. Normal values for the relative thickness of the left ventricular wall are 0.40-0.44, and values higher than this value indicate cardiac remodeling [12,13]. High consumption of table salt causes eccentric remodeling of the left ventricle [14]. AH is the main factor causing atherosclerosis of blood vessels. It has been proven in studies that there is a clear connection between blood pressure increase and the development of atherosclerosis in blood vessels, as well as myocardial hypertrophy [15,16]. AH, age, hypercholesterolemia, smoking, and diabetes affect the thickness of the intima-media of the carotid arteries. In hypertensive patients, the intima-media is significantly thicker than in healthy subjects. An intima-media thickness of more than 0.9 mm is a risk factor for heart attack and stroke [17]. Kidneys are the main target organs in AH. In AH, there are compensatory hemodynamic changes aimed at hyperperfusion and hyperfiltration in the kidneys. As a result, renal arterioles narrow. High blood pressure in the arterioles increases the intraglobuler pressure and the endotheliocytes in the glomerular capillaries are injured. High pressure in the balls causes endothelial dysfunction, hyperfiltration, and increased permeability of proteins in the basement membrane. The above processes constitute the pathogenetic mechanism of albuminuria.Efferent arterioles in the glomeruli undergo vasoconstriction under the influence of angiotensin II, which leads to ischemia, apoptosis, and ultimately glomerulosclerosis of glomerular endothelial cells. Endotheliocyte dysfunction is also affected by uric acid, low-density lipoproteins, insulin resistance, and obesity [18].It is reasonable to assume that the dry climate of the Aral Sea region, the Harmsel winds, which contain salt and sand, can have a negative effect on the human body and cause an increase in blood pressure, making it difficult to control it [19].The relationship between excess consumption of table salt and left ventricular hypertrophy in patients with hypertensive crisis in the Aral Sea region, which suffered from an environmental disaster, was studied in our previous research [19]. The effect of sodium on kidney, heart, and vascular damage in patients with salt-sensitive AH in the Aral Sea region remains a problem that has not been fully studied.Thus, the main goal of our study was to study the characteristics of target organ damage in patients with salt-sensitive AH in the practice of primary medical and sanitary facilities in the Aral Sea region.
2. Materials and Methods
- The scientific study was conducted in family clinics of the Ellikkalya district medical association of Karakalpakstan. A total of 1021 patients aged 40-70 years diagnosed with AH in district family clinics were included in the study. The results of laboratory-instrumental analysis of all patients included in the study were analyzed.Echocardiography, left ventricular myocardial mass index, and intima-media thickness of the common carotid and brachiocephalic arteries were performed using standard methods to determine target organ damage. General clinical blood and urine tests, blood biochemical tests - creatinine, urea, fasting glucose, lipid spectrum, blood sodium, serum uric acid, urine biochemical tests - urine creatinine, urine sodium and microalbuminuria were performed. The glomerular filtration rate was calculated according to the EPI formula (ml/min/1.73m2). Arterial blood pressure was measured by the method of N.S. Kortkov from the wrist artery.The taste threshold for table salt was tested using the R. Henkin method by placing one drop of a sodium chloride (NaCl) solution of increasing concentration from 0.02% to 5.12% on the anterior third of the tongue. The test began with a low-concentration (0.02%) NaCl solution and the concentration was increased by 2 times until the patient first felt a salty taste. The threshold for the first taste of salt is determined by the amount of NaCl. The table salt taste threshold was divided into 3 groups: low threshold ≤0.08%, medium table salt taste threshold=0.16%, and high table salt taste threshold ≥ 0.32%.Statistical processing of the results was carried out using the Statistica 6.0 software package. The main characteristics are presented as mean (M) and standard deviation (SD). The frequency of symptoms in the study group was estimated using the Pearson χ² method. The Pearson correlation test was used to determine whether there was a correlation between two quantitative indicators, as well as to assess its closeness and statistical significance. For all analyses, p values <0.05 were considered statistically significant.
3. Result and Discussion
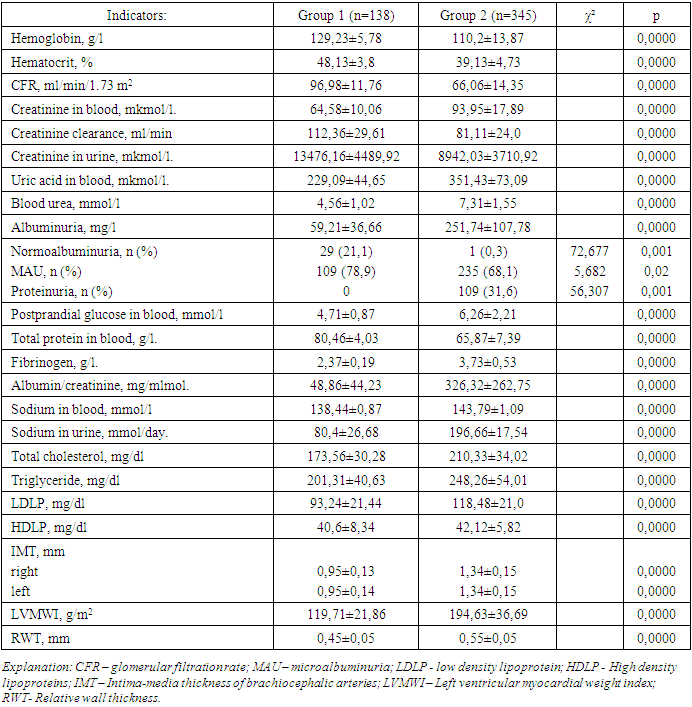
- The average age of 1021 patients included in the study was 57.68±8.06 years, of which n=627 (61.4%) were women and n=394 (38.6%) were men. The duration of AH was 7.88±3.35 years.Taste sensitivity threshold for table salt (TSTTS) was determined in patients as follows: TSTTS 0.08% in 11 patients, TSTTS 0.16% in 138 patients, TSTT 0.32% in 410 patients, TSTTS 0.64% in 345 patients, TSTTS 1.28% in 29 patients 2.56%, and TSTTS was 5.12% in 5 patients.In order to separately analyze the clinical and laboratory-instrumental indicators of patients with TSTTS 0.16%, 0.32%, 0.64% and 1.28%, we divided the patients into 2 groups: group 1 included 138 hypertensive patients with TSTTS equal to 0.16%, and 345 hypertensive patients with TSTTS equal to 0.64% to group 2.The average age of 138 hypertensive patients included in group 1 was 57.38±7.89 years, the duration of AH was 5.94±2.60 years. The mean age of 345 hypertensive patients included in group 2 was 55.12±8.83 years, the duration of AH was 8.56±2.98 years (respectively, р1=0.006; р2=0.0000). Of the patients in group 1, 42 (30.4%) were men and 96 (69.6%) were women, while of the patients in group 2, 124 (35.9%) were men and 221 (64.1%) were women. In patients belonging to group 1, systolic arterial pressure (SAP) was 151.67±9.93 mm.Hg, diastolic arterial pressure (DAP) was 91.52±3.94 mm.Hg, average arterial pressure (AAP) was 111.57±5.48 mm.Hg. In patients in group 2, the SAP was 175.59±13.09 mm.Hg, the DAP was 103.42±8.52 mm.Hg, and the AAP was 127.48±9.58 mm.Hg, which significantly differed from group 1 (p1=0.0000; p2=0.0001; p3=0.0000, respectively).The amount of hemoglobin in patients belonging to group 1 was 129.23±5.78 g/l, and it was found to be 110.2±13.87 g/l in patients of group 2 (р=0.0000).The rate of glomerular filtration (GFR) in group 1 patients was 96.98±11.76 ml/min/1.73 m2, and in group 2 patients it was equal to 66.06±14.35 ml/min/1.73 m2 (р=0.0000).Creatinine clearance in group 1 patients was 112.36±29.61 ml/min, and in group 2 patients it was 81.11±24.0 ml/min (р=0.0000).Blood uric acid was found to be 229.09±44.65 mkmol/l in group 1 patients, and 351.43±73.09 mkmol/l in group 2 patients (p=0.0000).Albuminuria averaged 59.21±36.66 mg/l in patients of group 1, and 251.74±107.78 mg/l in patients of group 2, significantly different from group 1 (p=0.0000).Normoalbuminuria was observed in 29 (21.1%) patients in group 1, and in 1 (0.3%) patient in group 2 (χ²=72.677, p=0.001).Microalbuminuria (MAU) was detected in 109 (78.9%) patients in group 1 and in 235 (68.1%) patients in group 2 (χ²=5.682, p=0.02).Proteinuria was detected in 109 (31.6%) patients in group 2, and there were no patients with proteinuria in group 1 (χ²=56.307, p=0.001).1st stage of chronic kidney disease (CKD) was observed in 97 (70.3%) of group 1 patients, and 21 (6.1%) of group 2 patients (χ²=220,080, p=0,001).CKD stage 2 was detected in 41 (29.7%) patients of group 1 and 197 (57.1%) patients of group 2 (χ²=29,589, p=0,001).Stage 3A of CKD was observed in 103 patients (29.8%), and stage 3B in 24 (6.9%) patients of group 2, and stage 3 of CKD was not detected in patients of group 1 (respectively, χ²1=327.296, p1=0.001; χ²2=63.137, р2=0.001).Intima-media thickness (IMT) of brachiocephalic arteries in group 1 patients was 0.95±0.13 mm on the right side and 0.95±0.14 mm on the left side. In group 2 patients, IMT was 1.34±0.15 mm on the right side, and 1.34±0.15 mm on the left side was found to be (p=0.0000).Left ventricular myocardial weight index (LVMWI) was 119.71±21.86 g/m2 in patients of group 1, and 194.63±36.69 g/m2 in patients of group 2 and was significantly different from group 1 (p=0.0000). The results of laboratory-instrumental analysis of patients are shown in Table 1.
|
4. Conclusions
- Our scientific research conducted at primary health care facilities along the Aral Sea coast revealed that the population of this region consumes salt and products high in salt, which over the years has led to the development of salt-sensitive AH, and the development of disease complications is related to the amount of salt consumed. In patients with salt-sensitive arterial hypertension, we observed that target organ damage was clearly expressed, dyslipidemia was high, and glucose tolerance was impaired. This indicates that family physicians should perform tasks such as taking patients with AH into separate groups in family clinics, prescribing them the correct medications, providing patients with individual, regular non-drug treatment recommendations and monitoring their compliance, and regularly evaluating the effectiveness of the treatment regimens provided to patients on the regression of target organ damage based on laboratory and instrumental examination results.Patronage nurses and family doctors who work directly with the population in family clinics should be retrained in dietetics courses, so that they can provide our people with ongoing guidance on healthy eating in their work.At the same time, the implementation of the Henkin method for determining the taste threshold for table salt in the practice of primary health care institutions plays an important role in preventing the population from cardiovascular diseases, predicting target organ damage, and providing an effective approach to treatment.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML