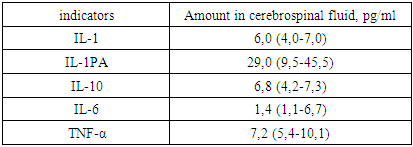-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(8): 2455-2459
doi:10.5923/j.ajmms.20251508.06
Received: Jun. 19, 2025; Accepted: Jul. 21, 2025; Published: Aug. 2, 2025

Cytokines in the Pathogenetic Development of Parkinson's Disease
Abdukadirov Eldor Israilovich1, Matmurodov Rustam Jumanazarovich2, Inoyatova Sitora Oybekovna1, Togayeva Adel Yusupovna3
1Associate Professor, Department of Neurological Diseases, Folk Remedies, Tashkent State Medical University, Uzbekistan
2Professor of the Department of Neurology and Medical Psychology, Tashkent State Medical University, Uzbekistan
3Department of Neurological Diseases, Folk Remedies, Ordinator, Tashkent State Medical University, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This study investigates the role of pro-inflammatory and anti-inflammatory cytokines in the pathogenesis and progression of different clinical subtypes of Parkinson’s disease (PD). A total of 160 PD patients were examined, with subgrouping based on clinical phenotype (tremor-dominant and akinetic-rigid-dominant forms), disease stage, and duration. Levels of cytokines including IL-1β, IL-6, IL-10, IL-1RA, and TNF-α were measured in serum and cerebrospinal fluid (CSF). Results revealed elevated IL-1β and IL-6 levels and decreased IL-1RA in PD patients compared to controls, indicating a shift towards a pro-inflammatory state. These changes were particularly significant in patients with advanced stages and in the akinetic-rigid subtype. A correlation was also found between cytokine levels and various clinical features, including non-motor symptoms, depression, and disease lateralization. The findings support the involvement of chronic neuroinflammation in PD pathogenesis and highlight cytokine profiling as a potential biomarker for differentiating PD subtypes and monitoring disease progression.
Keywords: Parkinson’s disease, Cytokines, IL-1β, IL-6, IL-10, IL-1RA, TNF-α, Neuroinflammation, Disease subtypes, Cerebrospinal fluid, Disease progression, Non-motor symptoms
Cite this paper: Abdukadirov Eldor Israilovich, Matmurodov Rustam Jumanazarovich, Inoyatova Sitora Oybekovna, Togayeva Adel Yusupovna, Cytokines in the Pathogenetic Development of Parkinson's Disease, American Journal of Medicine and Medical Sciences, Vol. 15 No. 8, 2025, pp. 2455-2459. doi: 10.5923/j.ajmms.20251508.06.
1. Introduction
- Parkinson's Disease (PD) is a chronic, progressive neurodegenerative disorder primarily characterized by dopaminergic degeneration of the substantia nigra. Its clinical manifestations include hypokinesia, muscle rigidity, tremors, postural instability, as well as various non-motor symptoms [9].The clinical picture of the disease is diverse, typically presenting in combinations of the following symptom types: akinetic-rigid, tremor-dominant, and mixed forms, with differences in progression rate and a range of motor and non-motor features. Lateralization of symptoms is also a distinctive characteristic.Due to the clinical heterogeneity of PD, there are both general pathophysiological mechanisms common to the disease and unique pathogenic chains associated with specific clinical variants. Beyond phenotypic differences, biochemical identification of clinical forms of PD holds significant fundamental and practical importance, serving as a basis for targeted symptomatic therapy.Recent studies on the pathogenesis of PD have paid special attention to the immunological mechanisms involved in disease progression [1], particularly the role of pro-inflammatory and anti-inflammatory cytokines in the neurodegeneration process [2].The akinetic-rigid and tremor-dominant forms of PD differ in terms of epidemiology, pathomorphology, and pathophysiology. Some researchers even propose that these described clinical forms may represent independent nosological variants of primary parkinsonism [3].During the progression and treatment of PD, gradual changes in the clinical picture are observed, with the emergence of new symptoms and the intensification of existing ones. These include iatrogenic and neurogenic symptoms that often do not respond well to therapy and exert a growing impact on patients, leading to a complex clinical picture. This complexity necessitates adjustments to treatment algorithms based on the form and course of the disease [4].Although cytokine profiles in PD have been studied, most research is limited by the duration of observation, the number of clinical studies on the topic, and varying concentration data. There are conflicting and scattered findings regarding cytokine levels in blood and cerebrospinal fluid. For instance, Stypula et al. (1996) reported significantly elevated levels of interleukin (IL)-1β, IL-2, and IL-6 in the blood of PD patients [5].However, many studies do not consider the impact of the medications patients are taking. In recent years, researchers have focused not only on the changes in cytokine levels but also on their association with the clinical manifestations of PD [6]. For example, Lindqvist D. et al. found that elevated levels of IL-6 and tumor necrosis factor-alpha (TNF-α) in PD patients were positively correlated with certain non-motor symptoms [7].Despite these findings, the full interrelation of cytokine profile markers with the phenotypic heterogeneity and pathomorphosis of PD remains unclear. Questions remain regarding cytokine levels in blood and cerebrospinal fluid in relation to disease subtype, stage, age of onset, progression speed, symptom lateralization, and severity of motor and non-motor manifestations, as well as the specific response to anti-parkinsonian therapy.Research Objective: To improve the diagnosis of cytokine metabolism and oxidative stress markers in different forms and variants of Parkinson's disease, and to optimize therapy accordingly.
2. Research Subjects and Methods
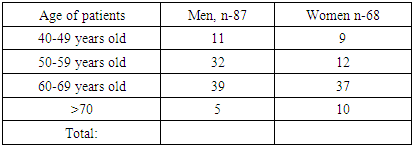
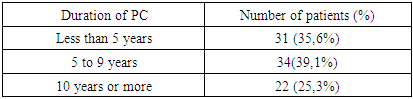
- From 2023 to 2025, a total of 160 patients diagnosed with this syndrome were examined. Patients meeting the diagnostic criteria for parkinsonism were selected for the study. The clinical-biochemical correlation group consisted of 87 patients. Among those with PD, there were 47 (54%) females and 40 (46%) males. The age range of participants was 43 to 82 years, with a mean age of 65 (range 57–73) years. The distribution of patients by age and sex is presented in Table 1.
|
|
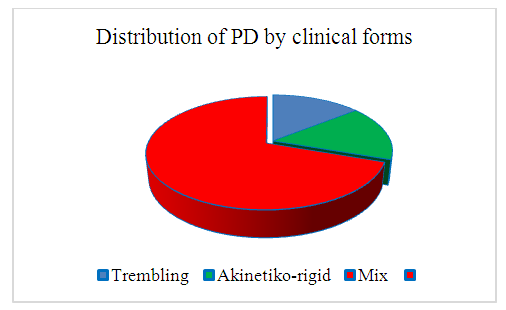 | Figure 1. Distribution of patients according to clinical forms of Parkinson's disease (n=87) |
|
|
|
|
|
3. Conclusions
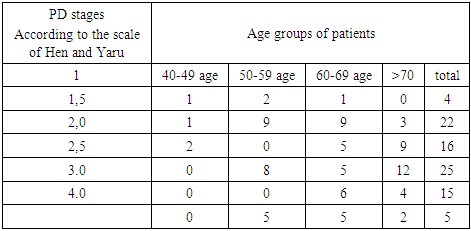
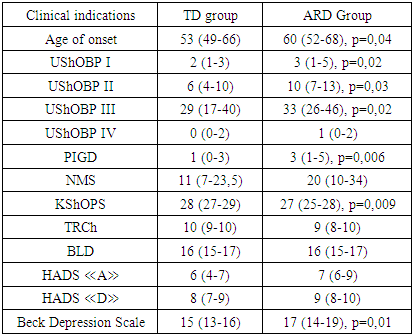
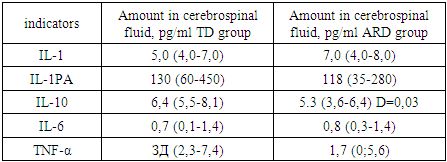
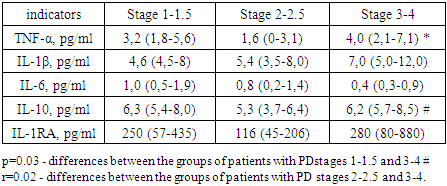
- Our study provides more precise insights into cognitive impairment among patients with the akinetic-rigid dominant (ARD) form of PD. This association is supported by studies investigating brain activity in PD. For instance, J. Prodoehl (2013) showed that patients with ARD have reduced activity in the prefrontal cortex and globus pallidus compared to those with the tremor-dominant (TD) form, based on MRI data using voxel-based analysis [10]. Similarly, Rosenberg-Katz et al. (2013), using voxel-based morphometry, demonstrated distinct regions of atrophy in different PD subtypes in gray matter [3].The leading pathogenic mechanisms in PD include oxidative stress and the excessive accumulation of neurotoxic substances, such as reactive oxygen species (ROS), inflammatory mediators, and iron ions. Pro-inflammatory and anti-inflammatory cytokines present in biological fluids of PD patients are believed to play dual roles — both promoting neurodegeneration and exerting neuroprotective effects.According to our study, the concentration of IL-6 in the serum and CSF of PD patients was significantly elevated compared to controls, consistent with findings by T.N. Torgan (2013), D. Lindqvist (2012), and R. Scalzo (2010). Earlier reports by Stypula G. et al. (1996) and Blum-Degen D. et al. (1995) also confirmed increased IL-6 levels in the blood of PD patients [7].No significant differences in IL-10 concentrations between patient and control groups were found overall. However, in the TD group, IL-10 levels were higher than in the ARD group. This aligns with findings by Rentzos M. et al. (2009), who reported that IL-10 levels were approximately 1.5 times higher in the tremor-dominant form of PD compared to the akinetic-rigid form [205]. This elevated IL-10 in the TD form may reflect distinct immune-pathogenic mechanisms underlying PD subtypes. Prior studies have demonstrated not only clinical, but also pathomorphological and epidemiological differences between ARD and TD phenotypes [4].Regarding the effect of IL-10 on motor dysfunction in PD, our findings are consistent with Menza et al., who reported a correlation between UPDRS motor scores and IL-10 levels. According to our data, IL-1β levels in serum were significantly higher in PD patients than in controls, regardless of disease onset age, duration, or clinical form, indicating that elevated IL-1β may be a general marker of PD pathology.In our study, serum TNF-α concentrations did not differ significantly from the control group (p=0.9), consistent with findings by D. Lindqvist [6]. Interestingly, we observed a sex difference in TNF-α levels, with higher levels in men — a phenomenon not previously described. While no direct relationship was found between TNF-α levels and CSF concentrations or affective symptoms, we did observe that TNF-α levels were lower in patients with cognitive impairment compared to cognitively intact PD patients. This pattern was more pronounced in those with ARD and mild dementia, suggesting that TNF-α may be related to cognitive deficits. Moreover, a significant association was found between TNF-α levels and clinical lateralization: patients with right-dominant symptoms had higher TNF-α concentrations (3.1 [1.6–5.4] pg/mL) than those with left-dominant symptoms (1.8 [0–3.2] pg/mL) (p=0.04).A strong relationship between disease stage (Hoehn and Yahr) and cytokine levels was observed only for first-generation pro-inflammatory cytokines such as IL-1β and TNF-α, which act as upstream regulators of cytokine synthesis. These cytokines stimulate the expression of MHC complex antigens, cytokine receptors, chemokines, prostaglandins, and leukocyte recruitment to inflammatory sites.Although we observed a trend toward a relationship between disease stage and IL-6 levels — another key first-generation cytokine — this did not reach statistical significance. The absence of strong associations with second-generation cytokines may be due to the long premotor phase of PD (10–15 years), during which initial pro- and anti-inflammatory activity is modulated by numerous compensatory mechanisms. Over time, as the pathological process spreads, these mechanisms are exhausted, leading to chronic neuroinflammation.Therefore, our study confirms the presence of alterations in cytokine profiles in both serum and CSF of patients with PD. Across all clinical subtypes, elevated levels of IL-1β and IL-6 and reduced levels of IL-1RA were observed. These consistent immune shifts suggest that increased pro-inflammatory cytokines and insufficient anti-inflammatory compensation (e.g., low IL-1RA) contribute to the chronic, progressive neuroinflammatory process underlying PD pathogenesis.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML