-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(8): 2449-2454
doi:10.5923/j.ajmms.20251508.05
Received: Jun. 26, 2025; Accepted: Jul. 27, 2025; Published: Aug. 2, 2025

The Role of IL-1β in Patients with COVID-19 Associated Reactive Arthritis
Abdullaev Zh. U., Mirakhmedova Kh. T., Saidrasulova G. B.
Tashkent State Medical University, Tashkent, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

To evaluate the role of IL-1β in patients with Covid-19 associated inflammatory arthritis. Materials and methods. The study included 155 patients. The main group consisted of 135 patients who had suffered COVID-19, the control group consisted of 20 healthy people. General clinical, laboratory and instrumental studies of patients were assessed. Results. It was found that the average age of patients was 54.7±3.8. When comparing gender differences, it was found that 71.9% of the patients were male and 28.1 % were female, indicating a 2.56-fold increase in the number of women. The time between COVID-19 infection and the onset of the first joint syndrome was 23.5±4.6 days. The duration of the joint syndrome was 170.6±4.6 days. RF, ACPA, and ANA were negative in all patients. Elevated serum inflammatory markers (ESR and/or CRP) were noted in 92.6% of the study group. When the average amount of IL-1β was analysed and compared between groups, it was found that the average amount of IL-1β in the main group was almost 12 times higher than in the control group and was statistically significant (p<0.05). A moderate positive correlation was found between the IL-1β and the duration of morning stiffness (r=0.38). However, no correlation (r=0.072) was observed between the VAS scale and the IL-1β, while a positive moderate correlation was also found between this cytokine and CRP and IgG to SARS-CoV-2 (r=0.21) and (r=0.65), respectively. Conclusion. COVID-19 may trigger inflammatory arthritis, specifically reactive arthritis. The increased serum level of IL-1β may be a potential indicator and cause of exacerbation of joint syndrome in patients suffering from COVID-19. The relationship between IL-1β and clinical, laboratory indicators in patients highlights its role in the manifestation and worsening of joint syndrome.
Keywords: COVID-19, IL-1β, Reactive arthritis, Inflammatory arthritis, SARS-CoV-2
Cite this paper: Abdullaev Zh. U., Mirakhmedova Kh. T., Saidrasulova G. B., The Role of IL-1β in Patients with COVID-19 Associated Reactive Arthritis, American Journal of Medicine and Medical Sciences, Vol. 15 No. 8, 2025, pp. 2449-2454. doi: 10.5923/j.ajmms.20251508.05.
1. Introduction
- In rheumatology, infectious-etiology arthritis is classified separately, and the causative agents may vary widely. Among such types of arthritis are those with viral etiology. Viral arthritis typically presents as acute-onset polyarticular arthritis. It has been established that several viruses may cause arthritis, including parvoviruses, alphaviruses, hepatitis B virus, Epstein-Barr virus, and tropical viruses such as Zika virus [1]. Hepatitis C virus and alphaviruses can lead to chronic arthritis, while parvovirus B19, hepatitis B, and rubella virus usually cause self-limiting arthritis. Nevertheless, virus-associated arthritis accounts for only about 1% of all cases [2]. Some studies have identified that parvovirus B19, hepatitis C virus, and Epstein-Barr virus may increase the risk of rheumatoid arthritis (RA). Meanwhile, cytomegalovirus and hepatitis B virus do not appear to influence RA development, though Chikungunya virus may trigger persistent inflammatory arthritis. Respiratory viruses are often associated with arthralgia and myalgia. Among patients infected with COVID-19, myalgia or arthralgia was observed in 15–44% during the infectious phase [3].However, recent sources suggest that SARS-CoV-2 infection, through immune-inflammatory mechanisms, may also affect the musculoskeletal system, possibly leading to inflammatory arthritis during the infectious or post-infectious period. Another research also indicates that respiratory infections may increase the risk of developing autoimmune inflammatory arthritis, such as RA. In patients with pre-existing inflammatory arthritis, infections may exacerbate or trigger disease flares [4,5].After COVID-19 infection (post-COVID-19), inflammatory arthritis has been frequently observed. According to Ursini et al., 51% of patients exhibited asymmetric monoarthritis or oligoarthritis. In 20%, RA-like symptoms were present, and in 11%, axial symptoms were predominant [6]. Jacopo et al. proposed in their research that post-COVID-19 syndrome may act as a trigger for the development of inflammatory arthritis [7]. Other sources report that COVID-19 and even vaccines against it can induce inflammatory arthritis [8] and cause rare systemic disorders, such as vasculitis, Still’s disease, and dermatomyositis [9].SARS-CoV-2 infection occurs in two clinical phases:1. The initial phase of viral replication and development.2. The immune response of the body to the virus [10].Scientific data collected so far indicate that SARS-CoV-2 can attack the musculoskeletal system by stimulating immune-mediated inflammatory processes, potentially leading to inflammatory arthritis during both infectious and post-infectious phases [11]. In clinical studies, patients with SARS-CoV-2 infection have shown an increase in various cytokines, especially IL-1β, which stimulates neutrophils and macrophages and activates the innate immune system. IL-1β is one of the main mediators of inflammation and is the most extensively studied cytokine of the IL-1 family, associated with many rheumatic diseases [12]. IL-1β contributes to chondrolysis and joint destruction. Studies show that activated monocytes produce IL-1β. American researchers found that in synovial tissue biopsies from patients with RA-like leukocyte-rich, leukocyte-poor, and OA-like arthritis, IL-1β was dominant in monocytes across all three types. These findings support the effectiveness of IL-1β inhibitors in RA and their significance in OA. Additionally, the pathological role of IL-1β has been explored in other types of arthritis such as gout and osteoarthritis. Interleukin-1β plays a key role in the development of arthritis. For instance, in OA pathogenesis, IL-1β is an inflammatory and catabolic cytokine, promoting cartilage degradation by stimulating matrix-degrading enzymes.Along with IL-6 and TNF-α, IL-1β contributes to chronic inflammatory processes like RA and Crohn’s disease. IL-1β and IL-8 together enhance Th1 and Th17 responses in autoimmune diseases, leading to more severe tissue damage. IL-1β also disrupts TGF-β signaling and, along with IL-23, suppresses regulatory T cells while promoting Th17 cell polarization.IL-1β plays a key role in multiple inflammation checkpoints:• Stimulates the production of collagenases and metalloproteinases.• Increases expression of cyclooxygenase-2 (COX-2), C-reactive protein, and various protease inhibitors that contribute significantly to inflammation [13].The cytokine release from epithelial cells damaged by COVID-19 leads to elevated levels of IL-1β, IL-6, IL-8, and other cytokines and chemokines. This activates macrophages, attracts neutrophils to the site of inflammation, and supports the recruitment of monocytes and macrophages.For example, the SARS virus nucleocapsid protein can stimulate NF-κB transcription factor, thereby increasing IL-6 expression. IL-6, in turn, promotes the differentiation and activation of monocytes into macrophages, amplifying the inflammatory process. As alveolar damage progresses, monocytes and macrophages continue producing inflammatory cytokines initially Il-1β. Il-1β cytokine affects systemic and local to joints [14].
2. Materials and Methods
- The study was conducted in 2020-2024 at the Multidisciplinary Clinic of the Tashkent Medical Academy with the participation of patients who received inpatient and outpatient treatment in the departments of internal rehabilitation, rheumatology, cardio-rheumatology and the Republican Rheumatology Center IADK. The study was carried out with a total of 155 participants. 135 patients with varying degrees of severity of COVID-19 and joint syndrome were selected for the study, and 20 healthy participants were chosen as the main group for comparison (below research design-pic. 1).
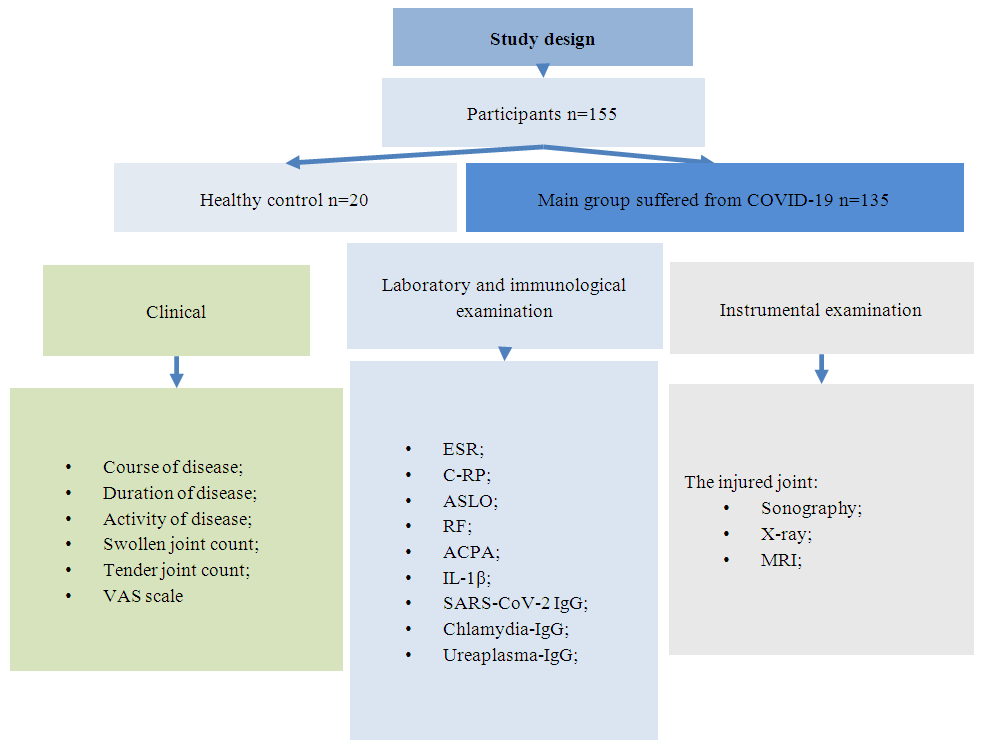 | Picture 1. Study design |
3. Results
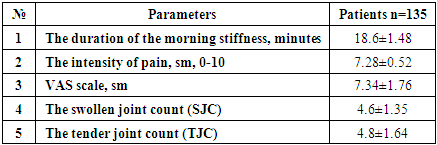
- The study found that the mean age of patients was 54.7±3.8. When comparing gender differences, it was found that 71.9% of the patients were female and 28.1 % were male, indicating a 2.56-fold increase in the number of female. The time between COVID-19 infection and the onset of the first joint syndrome was 23.5±4.6 days (min-5 days, max-3 months). The duration of the joint syndrome was 170.6±4.6 days (min-2.5 months, max-12 months). RF, ACPA, and ANA were negative in all patients. Elevated serum inflammatory markers (ESR and/or CRP) were noted in 92.6% of the study group.The course of the disease was analyzed in the study. Acute, subacute and chronic course of the disease was observed in patients participating in the study (pic. 2).
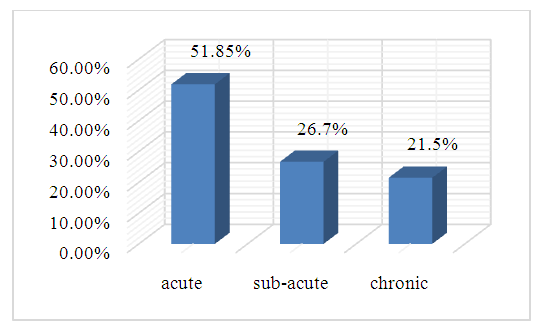 | Picture 2. The course of joint syndrome in patients with COVID-19 associated reactive arthritis |
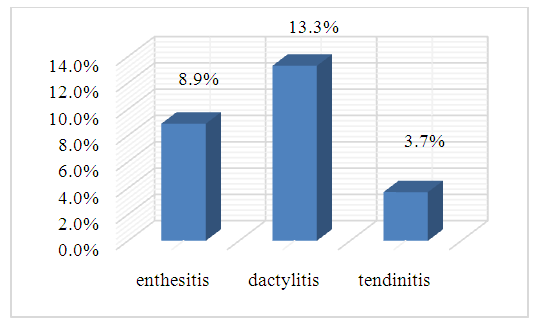 | Picture 3. Clinical manifestation of joint syndrome in patients with COVID-19 associated reactive arthritis |
|
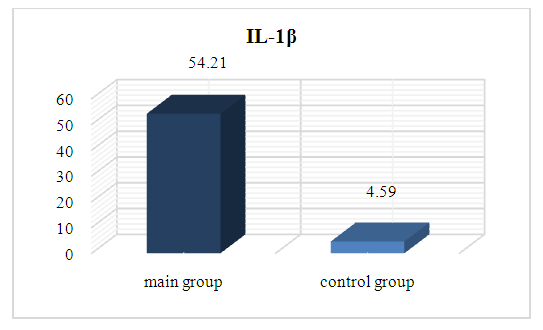 | Picture 4. The mean levels of IL-1β in patients and control |
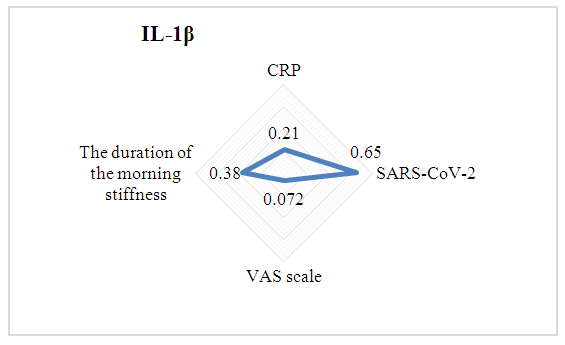 | Picture 5. The relationship between IL-1β and clinical, laboratorical parameters of patients |
4. Discussion
- During pandemic period and until since, patients with active COVID-19 disease and post-Covid-19 period were complaints to more than 200 symptoms, impacting multiple structures and functions of the body, such as cardiovascular, thrombotic, and cerebrovascular diseases, diabetes mellitus, myalgic encephalomyelitis and chronic fatigue syndrome, and autonomic system impairments such as postural orthostatic tachycardia syndrome. Apart from that, at least 65 million individuals worldwide are estimated to have long COVID, with cases increasing daily [15]. Some symptoms may be last for years [16] and exactly clinical signs are expected to be lifelong [15]. Post-COVID-19 inflammatory arthritis is characterized by joint inflammation following an COVID-19 infectious episode. During COVID-19 pandemic and after pandemic period, there has been encountered a syndrome of inflammatory arthritis with elevated inflammatory markers. There has been information about patients affected by post-covid-19 inflammatory arthritis often report persistent diffuse joint pain and fatigue [17].In our study the mean age of patients was 54.7±3.8. However, a small controlled study by American researcher Siddhanth Yadav and Sarah L. Bonn reported joint syndrome in patients aged 19, 45, 46, 54, and 61 years after COVID-19. All of these patients were female. These scientists put forward the idea that COVID-19 could be a trigger for Reactive arthritis or Rheumatoid Arthritis [17].In our study female was 2.5 times higher than male. In one article by European researchers reported that 54% of women were affected by COVID-19-associated joint syndrome [18].When examining the time to the onset of post-COVID-19 joint syndrome in this study, the average was found to be 23.5 ± 4.6 days (minimum 5 days, maximum 3 months). Research conducted by Turkish scientists indicates that signs of inflammatory arthritis typically emerged more than one week after COVID-19. However, it is important to note that the sample size in this article is small [19]. Additionally, while the presence of COVID-19 was confirmed through nasal swab PCR testing in that scientific study, our study confirmed past COVID-19 infection serologically. In controlled studies by Danish researchers, this period is reported to be 16 days. Another brief scientific article mentions that a duration of 2 weeks elapsed between the illness and the onset of joint syndrome [20].COVID-19 associated reactive arthritis usually presents acutely and typically resolves within 6 months. In a small percentage of cases, the condition may become chronic. Research indicates that a chronic course is more commonly observed in women among patients with COVID-19 associated reactive arthritis. The studies discussed did not offer specific details regarding the progression of COVID-19 associated reactive arthritis. However, there is evidence suggesting that treatment measures were maintained for 5 to 42 days [21].It should be noted that, when studying the radiographic signs of COVID-19 associated reactive arthritis, no reliable radiographic changes were detected, with only 2 patients reported to have sclerotic changes [22,23]. However, MRI revealed signs such as synovitis, soft tissue edema, and osseitis [24]. As mentioned above, tendinitis and enthesitis were detected in our study using clinical ultrasound and MRI examinations. In many studies, enthesitis manifested as Achilles’ tendon injury.In COVID-19 and inflammatory arthritis, tissue damage is observed as a result of increased IL-1β cytokine production by activated immune cells. The cytokine IL-1β is a potent pro-inflammatory cytokine that causes bone and cartilage destruction and joint damage. Elevated levels of IL-1β in synovial fluid are positively correlated with markers of inflammation. During inflammatory cases, proinflammatory and inflammatory cytokines increase and being relationship with inflammatory markers. In our study also found similar results. As well, our study serves to complement this theory.
5. Conclusions
- COVID-19 may trigger inflammatory arthritis, specifically reactive arthritis. The increased serum level of IL-1β may be a potential indicator and cause of exacerbation of joint syndrome in patients suffering from COVID-19. The relationship between IL-1β and clinical, laboratory indicators in patients highlights its role in the manifestation and worsening of joint syndrome.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML