-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(7): 2404-2408
doi:10.5923/j.ajmms.20251507.65
Received: Jul. 4, 2025; Accepted: Jul. 26, 2025; Published: Jul. 30, 2025

Early Postnatal Liver Morphometry in First-Generation Rats Born to Females with Induced Diabetes Mellitus
Muborakxon A. Ubaydullayeva, Feruza Kh. Azizova
Histology and Medical Biology Department, Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Muborakxon A. Ubaydullayeva, Histology and Medical Biology Department, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The aim of this study was to identify morphometric changes in the liver of first-generation offspring of rats born to dams with alloxan-induced diabetes mellitus during the early postnatal period. Histological, morphometric, and digital analysis methods were employed. Liver samples of neonatal rats were examined on postnatal days 3, 7, 14, 21, and 30. The obtained data revealed significant alterations in hepatic structural development in offspring exposed to maternal diabetes. A significant increase in lobule area and diameter, sinusoid dilation, as well as elevated numbers of mitoses and binucleated hepatocytes were observed in the early stages. Disorganization of the hepatic cord structure, cytoplasmic vacuolization of hepatocytes, and delayed cellular maturation were also noted. These findings indicate compensatory and adaptive responses triggered by intrauterine metabolic stress. Enlargement of central veins and increased sinusoidal volume suggest vascular remodeling aimed at counteracting hypoxic and hemodynamic disturbances. Thus, maternal diabetes mellitus exerts a significant impact on the structural and functional development of the liver in offspring, potentially leading to long-term consequences and warranting further investigation using molecular markers of proliferation and apoptosis.
Keywords: Maternal diabetes, Liver morphometry, Postnatal development, Hepatocytes, Morphogenesis
Cite this paper: Muborakxon A. Ubaydullayeva, Feruza Kh. Azizova, Early Postnatal Liver Morphometry in First-Generation Rats Born to Females with Induced Diabetes Mellitus, American Journal of Medicine and Medical Sciences, Vol. 15 No. 7, 2025, pp. 2404-2408. doi: 10.5923/j.ajmms.20251507.65.
Article Outline
1. Introduction
- The liver is one of the most important organs responsible for metabolism, detoxification, hematopoiesis, and immune regulation. In mammals, including laboratory rats and humans, its morphogenesis continues not only during embryogenesis but also in the early postnatal period, which is crucial for the formation of the morphofunctional maturity of the organ [6].Recent studies emphasize that it is in the neonatal period that the formation of liver lobules, vascular architecture, and spatial organization of hepatocytes is completed. New lobules occur mainly at the periphery of the liver during the first week of life, with active involvement of the mesothelium and Wnt-signaling molecules. A key role in this process is played by the proliferation of endothelial cells originating from the central veins, which determines the completion of angiogenesis and the final number of liver structural units [1].The histological stages of hepatobiliary system development in rats during the first 30 days of postnatal life include the trabecular organization of hepatocytes, formation of portal tracts, and development of mature bile ducts [2]. The use of MTT assay made it possible to determine the peak of metabolic activity of hepatocytes by days 7–10 of life [18].Additional insight into the molecular aspects of liver morphogenesis is presented by using single-cell transcriptomics, it was shown that active gene expression responsible for metabolism, protein synthesis, and transport is observed between days 7 and 14 of life, and by day 30, the liver reaches functional maturity [13].However, the sensitivity of the developing liver to metabolic disturbances, especially under pregnancy pathologies, remains extremely high. So, zinc deficiency in the prenatal period causes pronounced morphological changes: hepatocyte destruction, mitochondrial damage, hypoxia, and increased apoptosis [19]. Also its important to say that vascular development of the liver is an active and spatially organized process that is critically important for complete morphogenesis [3].One of the most significant metabolic stressors during gestational and postnatal periods is diabetes mellitus. According to IDF (2024), type 1 diabetes continues to spread at an epidemic rate, especially in developing countries, including Uzbekistan [8]. The glycemic control status of the pregnant woman directly affects the growth and morphogenesis of fetal organs, including the liver [12].Classical studies have shown that maternal hyperglycemia stimulates excessive fetal growth, hepatomegaly, and morphofunctional immaturity of the liver [5], [11]. Fetal hyperinsulinemia causes hepatocyte hyperplasia and glycogen accumulation, contributing to liver enlargement [10].Diabetic fetopathy has been described as a combination of structural and functional disorders, including hepatomegaly, hypoxia, and disproportions in organogenesis [16]. The development of steatosis, vascular disorganization, inflammation, and fibrosis in fetuses and newborns of diabetic mothers has been confirmed [14].Particularly alarming is the observation that persistent disorders of liver histoarchitecture are present even in cases of mild diabetes. Kupffer cells play a crucial role in regulating regeneration and inflammation, as well as participating in stem cell activation and tissue remodeling [9]. Moreover, molecular-level findings indicate that in newborns from mothers with gestational diabetes, key metabolite levels change even under adequate glycemic control, suggesting underlying metabolic reprogramming [4].Altogether, these data clearly indicate that the liver is a target organ under maternal hyperglycemia. Despite existing experimental models, data on the effect of diabetes on structural and functional liver development in early postnatal ontogenesis remain limited and fragmented. Especially scarce are the data on histomorphological and molecular changes occurring in the first 30 days of life under previous intrauterine hyperglycemia.
2. The Aim of the Study
- The aim of this study was to identify morphometric changes in the liver of first-generation offspring of rats born to dams with alloxan-induced diabetes mellitus during the early postnatal period.
3. Materials and Methods
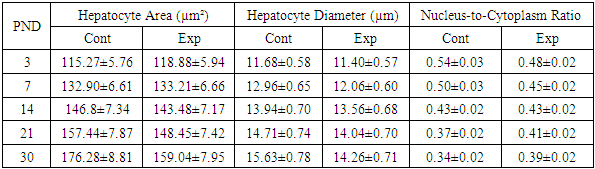
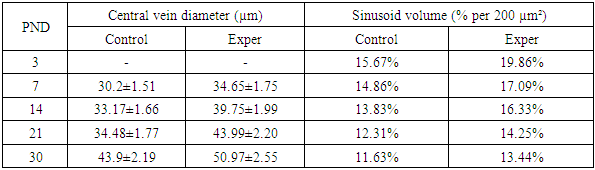
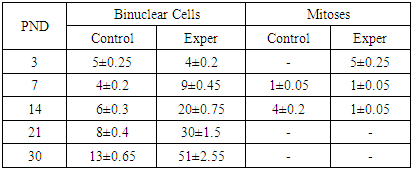
- The study was conducted on 50 (25 control, 25 experimental) nulliparous white outbred female rats weighing 160–180 g and their 253 offspring at various periods of early postnatal development. The experimental group included rats born to females with alloxan-induced diabetes, and the control group included intact rats of the same age born to healthy females.The study on laboratory animals was conducted in accordance with the “European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes” dated 18.03.1986.Experimental diabetes in the females of the experimental group was induced by a single intraperitoneal injection of alloxan trihydrate (Lachema, Czechoslovakia) at a dose of 13 mg per 100 g of body weight. The control group received an equivalent volume of saline.After confirming the induction of diabetes, females from both groups were mated with healthy males in a 1:4 ratio, and pregnancy development was monitored. Pregnancy and delivery in nearly all animals proceeded without complications.After birth, dams with pups were preliminarily divided into 3-, 7-, 14-, 21-, and 30-day groups according to postnatal age. Pups were fed breast milk until day 14, then received mixed feeding and from day 21 were switched to standard laboratory diet.Offspring from all groups were euthanized at 3, 7, 14, 21, and 30 days after birth under light ether anesthesia. The timepoints were selected based on the age classification of laboratory animals. During necropsy, the liver was removed, which served as the material for the study. Macroscopic examination with measurement of morphometric parameters was performed. Histological analysis was carried out on tissues fixed in 10% neutral buffered formalin, processed through ethanol series, and embedded in paraffin blocks. Sections of ~5.0 μm thickness were cut and stained with hematoxylin and eosin according to standard protocols, then examined under a microscope with photodocumentation of representative areas.Histological slides were digitized using whole slide imaging (WSI) technology with the Aperio system (Leica Microsystems, Germany).Primary processing and morphometric analysis of the obtained images were performed using open-source software QuPath version 0.5.1 (University of Edinburgh, Scotland). The area and diameter of the hepatic lobule were measured at 4x magnification; the diameter and area of hepatocytes, nuclear-cytoplasmic ratio (NCR), and central vein diameter were measured at 40x; the number of binuclear hepatocytes and mitoses were counted in 20 fields totaling 1 mm²; average sinusoid width and sinusoid density (per unit area) were also calculated.
4. Results
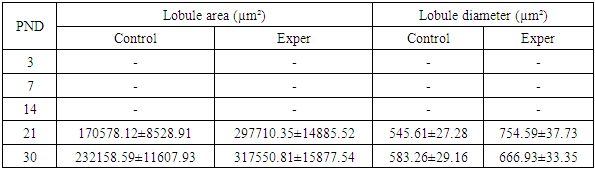
- It should be noted that induced maternal diabetes mellitus negatively affects fertility processes in females — up to 62% (compared to 92% in the control group), and prolongs the gestation period up to 24–25 days (vs. 21–22 days in the control group). Also notable was the reduction in the number of pups per litter to 5.2 ± 0.31, whereas in the control group, this indicator was significantly higher — 8.7 ± 0.42. Maternal diabetes mellitus also led to increased postnatal mortality — 10.8%, which significantly exceeded that of the control group — 2.2%. The death of first-generation rats in both groups was recorded within 14 days after birth. Naturally, all deceased pups were excluded from the experiments.Morphometric changes in liver lobulesAlthough it was conditionally possible to differentiate the hepatic lobule by postnatal day (PND) 14 in the control group, it was not possible to calculate its area and diameter due to poor triad organization at the lobular periphery. Calculations were performed for postnatal days 21 and 30 of postnatal development. On postnatal days 21 and 30, there was a significant increase in lobular area and diameter in the experimental group compared to the control. On postnatal day 21, the lobular area in the experimental group was 297,710.35 ± 14,885.52 µm², while in the control group — 170,578.12 ± 8,528.91 µm². The lobular diameter was 754.59 ± 37.73 µm in the experimental group versus 545.61 ± 27.28 µm in the control. On day 30, the lobular area in the experiment reached 317,550.81 ± 15,877.54 µm², and the diameter — 666.93 ± 33.35 µm, also exceeding the control values (232,158.59 ± 11,607.93 µm² and 583.26 ± 29.16 µm, respectively) Table 1. These data indicate accelerated growth and hypertrophy of the structural liver units in offspring of diabetic mothers.
|
|
|
|
5. Discussion
- The morphological analysis data confirm that alloxan-induced maternal diabetes mellitus affects the structural development of the liver in offspring during the early postnatal period. The disruption of the spatial organization of the trabecular structure at early stages, its disorganization, and delayed stabilization by day 21 compared to the control group indicate a delay in histogenesis, which is consistent with the results previously presented in the studies of Yu.A. Mitrofanov et al. [17] and I.V. Vasilieva [22], which showed the influence of the hyperglycemic maternal environment on the development of fetal parenchymal organs.The persistence of cytoplasmic vacuolization and disruption of hepatocyte homogeneity in the experimental group at later stages of postnatal development may be due to metabolic rearrangements under conditions of impaired carbohydrate and lipid metabolism, as also observed in offspring with gestational diabetes in experimental models [20]. Meanwhile, in the control group, the cytoplasm became homogeneous by day 21, indicating the completion of hepatocyte maturation.Indicators of mitotic activity and an increased number of binucleated hepatocytes in the experimental group at early stages indicate the activation of compensatory proliferation, similar to previously described results demonstrating regenerative liver activity under intrauterine hypoxia and metabolic stress [7].Marked accumulation of collagen fibers around the portal tracts in the experimental group of rats may indicate early signs of perivenular fibrosis, which was also noted in several studies on chronic hyperglycemia and its role in the development of steatohepatitis [21].Disruption in the timing and sequence of bile duct and portal tract formation under diabetic conditions supports data on the destructive impact of hyperglycemia on the mesenchymal and epithelial components of the hepatobiliary system in early ontogenesis [15].The observed significant increase in the area and diameter of hepatic lobules in the experimental group, especially on days 21–30, is consistent with the data of Kovalenko T.V. (2008) [10] and El-Sayyad et al. (2014) [15], who described hepatomegaly as one of the characteristic manifestations of diabetic fetopathy. Probably, lobular hypertrophy and vascular remodeling, expressed as dilation of central veins, result from intrauterine hyperglycemia and enhanced insulin stimulation of hepatocytes in response to metabolic overload described by Bofarda A. [2].The obtained morphometric data demonstrate a significant increase in the area and diameter of liver lobules in the offspring born to females with alloxan-induced diabetes mellitus, accompanied by sinusoidal dilation and increased volume at all observation periods. The increase in lobular area may be due to vascular remodeling aimed at compensating for hypoxic and metabolic disorders arising under hyperglycemia. These results are consistent with the findings of Patel et al. (2015) [18], indicating that intrauterine hyperglycemia is accompanied by remodeling of the liver microvasculature, including sinusoidal dilation and increased permeability.Increased mitotic activity at early stages in the offspring of the experimental group indicates stress-induced activation of hepatocyte proliferation, supported by the findings of El-Sayyad et al. (2014) [5], who observed hyperplasia and vacuolization of hepatocytes in newborns from diabetic mothers, accompanied by cytoskeletal instability and mitochondrial destruction.Particular attention should be paid to the pronounced increase in the number of binucleated hepatocytes in the experimental group from day 7 of life. This may be interpreted as an adaptive response to hypoxic and metabolic stresses initiated by intrauterine disorders. Hepatocyte binucleation is a marker of liver regenerative activity; however, its excessive expression, as shown by Rateb & Kamal (2022) [19], may indicate compensatory hyperplasia under cellular stress.An interesting finding is the early mitotic activity in the experimental group (5±0.25 mitoses/mm² on day 3), whereas mitoses were absent in the control group during this period. This may reflect an accelerated but disorganized cell cycle under hyperinsulinemia, as described in the work of Kazakova E.E. (2021) [9]. However, by day 14, mitoses were virtually absent in the experiment, unlike the control where their number increased.Data on increased central vein diameter and sinusoidal engorgement in animals of the experimental group are consistent with ultrastructural analysis results from Rateb & Kamal (2022) [19], where such changes were interpreted as signs of portal hypertension and microcirculatory disorders. The consistently high sinusoidal volume throughout the observation period in the experimental group may indicate prolonged microcirculatory impairment, correlating with the observations of Lucchesi et al. (2015) [14], who recorded persistent sinusoidal dilation and slowed blood flow in the liver of newborns with diabetic fetopathy.
6. Conclusions
- The results of morphological and morphometric analysis revealed some differences in liver development between the control and experimental groups, indicating the influence of maternal diabetes on liver formation in the early postnatal period.Morphometric data suggest that maternal diabetes exerts multifaceted effects on liver morphogenesis during the early postnatal period. Although signs of accelerated growth and proliferation are observed in the first weeks of life, they are accompanied by structural disorganization, delayed functional differentiation, and signs of vascular disarray. These changes may underlie a predisposition to metabolic and inflammatory liver diseases later in life.To further investigate the impact of maternal diabetes mellitus on proliferative and apoptotic processes in the liver of first-generation offspring, immunohistochemical studies with relevant markers are recommended.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML