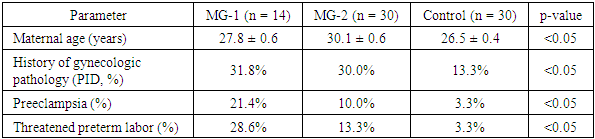-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(7): 2397-2403
doi:10.5923/j.ajmms.20251507.64
Received: Jun. 25, 2025; Accepted: Jul. 22, 2025; Published: Jul. 30, 2025

Prognostic Approaches and Optimization of Management in Pregnant Women with Focal Myocarditis
An Andrey Vladimirovich1, Sharipova Feruza Xayrullayevna2
1Department of Obstetrics and Gynecology, Tashkent Medical Academy, Tashkent, Uzbekistan
2Obstetrician Gynecologist, Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology, Tashkent, Uzbekistan
Correspondence to: Sharipova Feruza Xayrullayevna, Obstetrician Gynecologist, Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Cardiovascular diseases remain a leading cause of maternal morbidity and mortality, with focal myocarditis representing a particularly high-risk condition during pregnancy due to its variable clinical presentation and potential for severe complications. The aim of this study was to optimize the management of pregnant women with focal myocarditis by identifying prognostic criteria and evaluating the effectiveness of personalized obstetric strategies. A prospective observational study was conducted involving 74 pregnant women, divided into three groups: Myocarditis Group 1 (MG-1, n = 14) with myocarditis diagnosed during pregnancy, Myocarditis Group 2 (MG-2, n = 30) with myocarditis diagnosed before conception, and a control group (n = 30) without cardiovascular pathology. All participants underwent comprehensive clinical evaluation, electrocardiography, echocardiography, and Doppler ultrasonography. Patients with myocarditis demonstrated significantly increased left ventricular end-diastolic dimension (50.7 ± 3.9 mm vs. 48.7 ± 2.6 mm; p = 0.022), elevated pulmonary artery pressure (17.6 ± 6.1 mmHg vs. 12.7 ± 1.9 mmHg; p < 0.001), and reduced ejection fraction (65.1 ± 5.8% vs. 68.1 ± 2.6%; p < 0.05) compared to controls. Obstetric complications such as preeclampsia (21.4% vs. 3.3%) and threatened preterm labor (28.6% vs. 3.3%) were significantly more frequent in MG-1 (p < 0.05). A predictive logistic regression model was developed, identifying maternal age >25 years, viral infection during pregnancy, pelvic inflammatory disease, tachycardia >80 bpm, and specific ECG and EchoCG changes as key predictors, achieving a specificity of 91.6% and diagnostic accuracy of 80.4%. Implementation of individualized management based on this model improved maternal and fetal outcomes and reduced healthcare resource utilization. These results support the use of risk-adapted strategies and predictive tools for managing pregnant women with myocarditis.
Keywords: Myocarditis, Pregnancy, Prognosis, Echocardiography, Obstetric complications, Hemodynamics, Fetal outcomes, Risk stratification, Predictive model, Maternal morbidity
Cite this paper: An Andrey Vladimirovich, Sharipova Feruza Xayrullayevna, Prognostic Approaches and Optimization of Management in Pregnant Women with Focal Myocarditis, American Journal of Medicine and Medical Sciences, Vol. 15 No. 7, 2025, pp. 2397-2403. doi: 10.5923/j.ajmms.20251507.64.
Article Outline
1. Introduction
- Recent evidence underscores that cardiovascular disorders remain a leading contributor to maternal mortality and adverse perinatal outcomes, thereby requiring heightened clinical vigilance and the implementation of interdisciplinary care models [1], [2], [3]. Within this group, focal myocarditis holds particular clinical relevance due to its capacity to manifest either prior to conception or during pregnancy, often exerting a deleterious influence on both maternal and fetal health parameters [4], [5].Focal myocarditis (FM) is a relatively rare but clinically significant condition in pregnant women, with an estimated global prevalence ranging from 0.5% to 1.3% among cardiomyopathies diagnosed during pregnancy [Ref]. However, actual rates may be higher due to diagnostic challenges and subclinical presentation. In Central Asia, including Uzbekistan, specific epidemiological data on FM in pregnancy remain limited, but regional hospital-based studies suggest an increasing trend in cases associated with viral infections and chronic inflammatory conditions. Importantly, FM is linked to a higher incidence of adverse pregnancy outcomes such as preeclampsia, preterm birth, and fetal growth restriction, warranting greater clinical awareness and risk stratification.The clinical spectrum of focal myocarditis is notably heterogeneous, ranging from subclinical arrhythmogenic presentations to overt manifestations of heart failure. Such variability complicates timely detection and poses significant challenges for the management of pregnant patients [4], [6]. Existing literature consistently highlights that gestation in the context of myocarditis is associated with a markedly elevated risk of obstetric complications, including but not limited to preeclampsia, preterm labor, fetal growth restriction, and intrauterine fetal demise [3], [7].Current clinical guidelines from the European Society of Cardiology (ESC) suggest that peripartum cardiomyopathy—a condition with overlapping clinical features—requires specialized diagnostic protocols and individualized treatment pathways, encompassing early recognition, risk stratification, and targeted prevention of cardiovascular decompensation [8], [9]. Furthermore, optimal care for women of reproductive age with underlying cardiovascular pathology mandates coordinated involvement of a multidisciplinary team, including cardiologists, obstetricians, gynecologists, and primary care providers, throughout the entire continuum from preconception to the postpartum period [2], [3].Globally, variability in diagnostic accuracy and treatment outcomes for myocarditis during pregnancy is influenced by institutional resources and the level of specialized training among medical personnel, which, in turn, directly affects prognostic trajectories and perinatal outcomes [5], [10]. Additionally, maternal age has been identified as a pivotal risk factor for both the onset and progression of myocarditis, while coexisting inflammatory gynecological conditions—particularly pelvic inflammatory disease—serve to aggravate the overall prognosis [6], [11].Given the clinical complexity and the potential for severe maternal-fetal complications, there is an evident need for robust strategies aimed at early prediction, accurate diagnosis, and individualized management of myocarditis in pregnancy. In this regard, the development and implementation of predictive mathematical models that integrate clinical, hemodynamic, laboratory, and instrumental data are of paramount importance [6].The objective of this study is to refine the clinical management of pregnant women diagnosed with focal myocarditis by formulating reliable prognostic criteria and evaluating the clinical effectiveness of a structured, risk-adapted therapeutic approach.
2. Materials and Methods
2.1. Study Design and Population
- This investigation was structured as a single-center prospective cohort observational study, conducted at the Perinatal Center of Tashkent. A total of 74 pregnant women, admitted for childbirth, were enrolled following comprehensive clinical screening and diagnostic evaluation.Participants were categorized into two primary cohorts based on the timing and presence of myocarditis:Main Group (MG): Comprised 44 women with a confirmed diagnosis of focal myocarditis.MG-1: Included 14 patients (31.8%) in whom myocarditis was identified during pregnancy.MG-2: Included 30 patients (68.2%) with a history of chronic focal myocarditis diagnosed prior to conception.Control Group (CG): Included 30 pregnant women without any clinical, instrumental, or laboratory evidence of myocarditis or other cardiovascular pathology.While the study design was prospective, data collection was limited to the antenatal and intrapartum periods. Postpartum follow-up was not within the scope of this study, which may limit the ability to evaluate long-term maternal cardiovascular outcomes.
2.2. Clinical Evaluation and Data Collection
- All participants underwent thorough clinical evaluation, which incorporated detailed history-taking. This included obstetric, gynecological, and general somatic histories. Key demographic and clinical data were documented, including maternal age, gravidity and parity, previous pregnancy outcomes, and comorbid gynecologic or infectious diseases before or during the current pregnancy.Anthropometric parameters—such as height, weight, and body mass index (BMI)—were recorded alongside data from external obstetric examinations. Vital signs including systolic and diastolic blood pressure (SBP and DBP) and resting heart rate (HR) were measured at baseline and reassessed at designated follow-up points.
2.3. Instrumental Investigations
- All participants underwent a standardized instrumental diagnostic protocol in accordance with international clinical guidelines.Electrocardiography (ECG): Standard 12-lead ECG was conducted using the Edan SE-3 device (Edan Instruments Inc., China), following the recommendations of the American Heart Association (AHA) for resting electrocardiogram interpretation in adults. The following parameters were evaluated: cardiac rhythm, heart rate, conduction disturbances (e.g., bundle branch block), repolarization abnormalities (e.g., ST-T changes), and arrhythmic activity.Echocardiography (EchoCG): Comprehensive transthoracic echocardiographic examinations were performed using the Mindray DC-70 system (Mindray Bio-Medical Electronics Co., Ltd., China), based on guidelines from the European Association of Cardiovascular Imaging and the European Society of Cardiology (ESC). The following measurements were obtained:• Left ventricular dimensions (LVEDD and LVESD)• Interventricular septal and posterior wall thickness• Left atrial and right ventricular diameters• Aortic root diameter• Left ventricular volumes (LVEDV, LVESV)• Left ventricular ejection fraction (LVEF, Simpson’s biplane method)• Mean pulmonary artery pressure (mPAP, estimated from tricuspid regurgitation velocity)• Diastolic function indices (E/A ratio, deceleration time)• Presence and degree of valvular insufficiencies (mitral, tricuspid)• Pericardial effusionDoppler Ultrasound Assessment of the Fetoplacental Complex: Ultrasound and Doppler evaluation was conducted according to ISUOG (International Society of Ultrasound in Obstetrics and Gynecology) guidelines. Standard fetal biometric parameters and anatomical surveys were performed. Doppler studies assessed hemodynamics in the uterine arteries, fetal umbilical artery, middle cerebral artery, ductus venosus, thoracic aorta, and across the tricuspid valve. Perfusion abnormalities were classified according to standard grading: Grade I-a (increased resistance without diastolic notch) and Grade I-b (presence of diastolic notch and high pulsatility index), as described in recent obstetric literature.Operator Training and Blinding: All procedures were carried out by certified physicians with a minimum of five years of experience in obstetric imaging and maternal-fetal medicine. Before the study commenced, internal calibration was conducted to ensure methodological consistency across investigators. Where feasible, examiners were blinded to group assignment to minimize observer bias. Echocardiograms were interpreted by cardiologists, while Doppler evaluations were assessed in collaboration with obstetricians specializing in fetal medicine.
2.4. Statistical Analysis
- All statistical procedures were executed using Microsoft Excel and Statistica for Windows, version 6.0. Distribution normality of continuous variables was assessed via the Kolmogorov–Smirnov and Shapiro–Wilk tests. Quantitative variables were expressed as mean ± standard deviation (M±SD), and categorical data as absolute values and percentages.Comparisons of normally distributed continuous variables between groups were performed using the Student's t-test, with a threshold for statistical significance set at p < 0.05. Categorical variables were compared using the chi-square (χ²) test with Yates correction.To identify predictors of adverse pregnancy outcomes, binary logistic regression modeling was employed. Diagnostic metrics—including sensitivity, specificity, overall accuracy, and odds ratios (ORs) with 95% confidence intervals (CIs)—were computed. Pearson correlation coefficients were calculated to explore associations between continuous variables.A prognostic scoring model was developed based on weighted risk factors derived from regression coefficients, enabling classification of patients into distinct risk categories.
2.5. Ethical Considerations
- The study protocol adhered to the ethical standards of the Declaration of Helsinki and received approval from the institutional ethics committee of the Perinatal Center in Tashkent. Written informed consent was obtained from all participants prior to enrollment in the study.
3. Results
3.1. Demographic Characteristics
- Comparative demographic analysis revealed a statistically significant age difference between the study groups. Patients in both myocarditis subgroups demonstrated a higher mean age compared to the control group (29.4 ± 0.6 years vs. 26.5 ± 0.4 years; p < 0.05). Within the myocarditis cohort, individuals with chronic disease (MG-2) were older on average than those with newly diagnosed myocarditis during pregnancy (MG-1), suggesting a potential correlation between disease chronicity and maternal age.
|
3.2. Gynecological and Obstetric Comorbidities
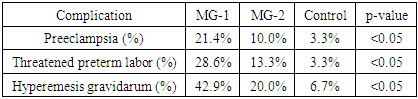
- The frequency of gynecological pathologies was notably elevated among women with myocarditis relative to controls. Pelvic inflammatory disease (PID) emerged as the predominant condition, identified in 31.8% (n = 7) of MG-1 and 30.0% (n = 9) of MG-2, compared to only 13.3% (n = 4) in the control group (p < 0.05). Less frequently encountered complications—such as endometritis, adnexitis, ectopic pregnancy, and colpitis—were predominantly found in the myocarditis groups.Obstetric complications displayed a similar trend. The incidence of preeclampsia was markedly higher in MG-1 (21.4%, n = 3) than in MG-2 (10.0%, n = 3), and significantly exceeded that of the control group (3.3%, n = 1; p < 0.05). Threatened preterm labor was also more common among MG-1 patients (28.6%, n = 4), versus 13.3% (n = 4) in MG-2 and 3.3% (n = 1) in the control group.
3.3. Hemodynamic Characteristics
- Evaluation of baseline hemodynamic parameters revealed that women with myocarditis had significantly elevated heart rates compared to healthy controls (96.9 ± 17.1 bpm vs. 88.8 ± 11.9 bpm; p < 0.05). No statistically significant intergroup differences were found in systolic or diastolic blood pressure measurements.
3.4. Echocardiographic Parameters
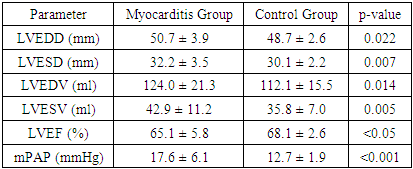
- Echocardiographic findings demonstrated pronounced alterations in cardiac morphology and function among women with myocarditis. Notably, the myocarditis group exhibited:• Increased LVEDD (50.7 ± 3.9 mm vs. 48.7 ± 2.6 mm; p = 0.022)• Elevated LVESD (32.2 ± 3.5 mm vs. 30.1 ± 2.2 mm; p = 0.007)• Higher LVEDV (124.0 ± 21.3 ml vs. 112.1 ± 15.5 ml; p = 0.014)• Elevated LVESV (42.9 ± 11.2 ml vs. 35.8 ± 7.0 ml; p = 0.005)• Raised mean pulmonary arterial pressure (17.6 ± 6.1 mm Hg vs. 12.7 ± 1.9 mm Hg; p < 0.001)
|
3.5. Electrocardiographic and Echocardiographic Abnormalities by Disease Timing
- Electrocardiographic disturbances—including diffuse myocardial changes, metabolic abnormalities, tachyarrhythmias, and conduction defects—were more frequently detected in MG-1. Echocardiography in this subgroup also revealed a high prevalence of mitral/tricuspid regurgitation (68%) and diastolic dysfunction (61%), reflecting acute myocardial compromise.In contrast, MG-2 patients showed a lower incidence but greater severity of ECG alterations, notably an increased rate of left bundle branch block. Chronic myocardial adaptation was evident on echocardiography, with a higher frequency of diastolic dysfunction, suggestive of sustained structural remodeling.
3.6. Pregnancy Complications in Early and Late Gestation
- During the first half of pregnancy, complications such as threatened miscarriage and hyperemesis gravidarum were more prominent in the myocarditis cohort. Mild forms of hyperemesis gravidarum were especially common in MG-1 (42.9%, n = 6), compared to MG-2 (20.0%, n = 6) and the control group (6.7%, n = 2; p < 0.05).
|
3.7. Doppler Assessment of Fetoplacental Perfusion
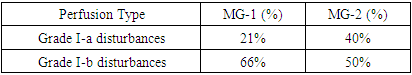
- Doppler ultrasound evaluation of uteroplacental and fetal blood flow revealed impaired hemodynamics in both myocarditis subgroups. Grade I-b perfusion defects predominated in MG-1 (66%), followed by grade I-a disturbances (21%). Conversely, MG-2 exhibited a higher proportion of grade I-a impairments (40%) and a lower incidence of grade I-b changes (50%). These patterns indicate compromised fetoplacental circulation, especially in acute inflammatory presentations.Table 4 summarizes the distribution of fetoplacental perfusion abnormalities. Grade I-b disorders predominated in MG-1, whereas MG-2 showed a greater proportion of Grade I-a disturbances.
|
3.8. Predictive Modeling of Adverse Outcomes
- Based on multivariate logistic regression, a predictive algorithm was constructed to estimate the likelihood of unfavorable obstetric outcomes in patients with myocarditis. Statistically significant predictors included:• Maternal age >25 years• History of viral infection during gestation• Presence of inflammatory gynecological disease• Preeclampsia• Threatened preterm birth• Tachycardia (>80 bpm)• ECG anomalies (diffuse changes, extrasystoles)• Valvular regurgitation on echocardiographyThe resultant model demonstrated:• Sensitivity: 57.8%• Specificity: 91.6%• Diagnostic accuracy: 80.4%Patients were stratified according to their total risk score:• 1–15 points: Low risk of adverse pregnancy outcomes• 16–45 points: High risk, requiring enhanced obstetric surveillanceWomen classified as high-risk based on this model should receive individualized care plans, including precise scheduling of labor induction, continuous intrapartum monitoring, and specialized postpartum follow-up to optimize clinical outcomes for both mother and child.
4. Discussion
- The findings of this study provide critical insights into the clinical management of pregnant women diagnosed with focal myocarditis, particularly in the setting of recent viral infections and underlying chronic inflammatory gynecological conditions. The data obtained are consistent with contemporary international literature, which emphasizes the pivotal role of thorough cardiovascular assessment throughout pregnancy in reducing maternal and fetal morbidity [2], [4], [8].It should be noted that, although the present study was prospective in nature, it did not include longitudinal postpartum data. Consequently, the long-term impact of focal myocarditis on maternal cardiac health and subsequent pregnancies remains to be elucidated. Future studies employing a longitudinal design could provide valuable insights into chronic cardiovascular sequelae in this population. A key hemodynamic alteration observed in affected patients was a persistently elevated heart rate, alongside echocardiographic evidence of ventricular remodeling. Specifically, increases in LVEDD and LVESD, along with a moderate reduction in LVEF, were indicative of compromised myocardial contractile function. These parameters align with prior reports documenting structural and functional deterioration of the myocardium in the context of gestational myocarditis [6], [9].The study also established a strong association between focal myocarditis and a heightened incidence of obstetric complications. Conditions such as preeclampsia, threatened preterm labor, and fetoplacental perfusion disorders were markedly more frequent among women with myocarditis, particularly those with acute disease onset. These findings reaffirm the well-documented link between maternal cardiovascular dysfunction and unfavorable perinatal outcomes [3], [6]. Moreover, the concomitant presence of chronic inflammatory gynecologic pathologies, especially pelvic inflammatory disease (PID), likely exerts an additive pathophysiological burden, suggesting a multifaceted interaction between systemic inflammation and cardiac function during gestation.In response to the need for early identification of high-risk pregnancies, the logistic regression model developed within this study demonstrated high specificity (91.6%) and acceptable diagnostic accuracy (80.4%) for predicting adverse outcomes. Risk factors such as advanced maternal age, a history of viral infections during pregnancy, electrocardiographic abnormalities, echocardiographic signs of myocardial dysfunction, and gynecological comorbidities emerged as independent predictors. These findings are concordant with validated international risk stratification frameworks employed in both cardiology and obstetric practice [6], [7], [10].From a practical standpoint, the study highlights the necessity of a tailored approach to labor and delivery planning for high-risk individuals. In line with European Society of Cardiology (ESC) guidelines, a preference should be given to non-invasive mechanical methods and carefully monitored pharmacologic induction using agents such as misoprostol or oxytocin. These strategies help mitigate cardiovascular stress and optimize maternal safety [3], [6], [7]. Cesarean section, while often considered in women with cardiac pathology, should not be routinely applied but rather reserved for cases of overt hemodynamic compromise or fetal distress. This selective approach reflects recent expert consensus discouraging indiscriminate surgical intervention in this population due to its limited maternal benefit and potential neonatal risks [2], [3].Postpartum care represents another essential component of management. Continuous cardiovascular monitoring, cautious administration of anticoagulants, and promotion of early mobilization are recommended to reduce the incidence of thromboembolic and hemodynamic complications. The therapeutic use of beta-blockers and low-molecular-weight heparins, when individually indicated, is consistent with current clinical guidance advocating for personalized risk-adjusted strategies in the postpartum period [1], [7], [11].Lastly, the integration of predictive modeling into routine clinical workflow offers not only medical but also economic benefits. The application of the developed risk stratification system significantly reduced the incidence of severe maternal-fetal complications, shortened hospital stays, and optimized the allocation of healthcare resources. These outcomes affirm the potential for broad implementation of the proposed model in obstetric cardiology settings, reinforcing both its clinical utility and its socioeconomic value.
5. Conclusions
- This study has demonstrated that focal myocarditis represents a clinically significant factor influencing both maternal and perinatal outcomes during pregnancy. The identification of key prognostic indicators—including maternal age, cardiovascular and gynecological comorbidities, electrocardiographic abnormalities, and echocardiographic dysfunction—enables early risk detection and timely intervention.The developed logistic regression–based predictive model proved effective in stratifying patients by risk level, thereby allowing for the implementation of individualized obstetric management. This targeted approach, encompassing tailored protocols for labor induction, delivery planning, and postpartum surveillance, has been shown to reduce the incidence of severe complications in both the mother and the fetus.Importantly, the integration of this structured management strategy into routine clinical practice offers not only clinical benefits but also tangible economic advantages, as evidenced by reductions in healthcare utilization, length of hospitalization, and overall treatment burden.In conclusion, the findings validate the proposed methodology as a clinically and economically sound tool for improving outcomes in pregnant women with myocarditis. Future large-scale, multicenter studies are warranted to confirm these results across broader populations and healthcare systems, as well as to refine the predictive accuracy of the model for wider application in obstetric cardiology.
ACKNOWLEDGEMENTS
- The authors sincerely thank the staff of the Department of Obstetrics and Gynecology at Tashkent Medical Academy and the Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology for their invaluable professional support, cooperation, and contributions during the research process.
DISCLOSURE
- The author declares no conflicts of interest relevant to the content of this article.
FUNDING
- This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML