-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(7): 2125-2128
doi:10.5923/j.ajmms.20251507.08
Received: May 29, 2025; Accepted: Jun. 21, 2025; Published: Jul. 3, 2025

Cytokine Background of Peripheral Blood - A Predictor of the Development of Herpes-Associated Erythema Multiforme Exudative
Indira Nurullaevna Abduvakhitova
PhD, Tashkent Regional Branch of the Republican Specialized Scientific and Practical Medical Center of Dermatovenereology and Cosmetology, Uzbekistan
Correspondence to: Indira Nurullaevna Abduvakhitova, PhD, Tashkent Regional Branch of the Republican Specialized Scientific and Practical Medical Center of Dermatovenereology and Cosmetology, Uzbekistan.
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

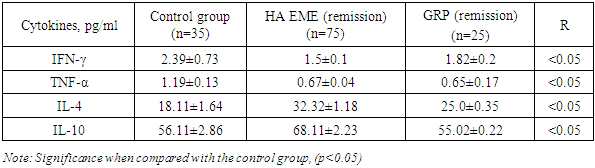
Background: Herpes-associated erythema multiforme exudative is a recurrent inflammatory disease of the skin and mucous membranes that develops predominantly in response to herpes simplex virus reactivation. The severity and recurrence of the disease are closely linked to the nature of the immune response, particularly the cytokine balance between T-helper type 1 and type 2 cells. However, data on cytokine-based prognostic markers remain scarce. Objective: To identify the peripheral blood cytokine profile in patients with herpes-associated erythema multiforme exudative and generalized recurrent herpes during relapse, and to assess their prognostic significance for mucocutaneous involvement. Methods: The study included 132 participants: 75 with herpes-associated erythema multiforme, 22 with generalized recurrent herpes infection, and 35 healthy controls. Serum levels of interferon-gamma, tumor necrosis factor-alpha, interleukin-4, and interleukin-10 were measured using enzyme-linked immunosorbent assay. Statistical analysis was performed using the Student’s t-test or Mann–Whitney test (p < 0.05). Results: During relapse, patients with herpes-associated erythema multiforme had significantly increased levels of interleukin-4 (64.65 ± 1.84 pg/ml) and interleukin-10 (78.11 ± 2.13 pg/ml), and markedly decreased levels of interferon-gamma (0.43 ± 0.14 pg/ml) and tumor necrosis factor-alpha (0.15 ± 0.06 pg/ml), compared to controls. These deviations persisted to a lesser extent during remission. Cytokine patterns in generalized recurrent herpes were less pronounced. Conclusions: A Th2-dominant cytokine profile characterizes herpes-associated erythema multiforme and may predict disease relapse. Cytokine monitoring could improve early diagnosis and guide personalized immunotherapy.
Keywords: Herpes-associated erythema multiforme, Cytokine profile, IFN-γ, TNF-α, IL-4, IL-10, Th1/Th2 balance, Herpes simplex virus, Relapse, Immunological predictor
Cite this paper: Indira Nurullaevna Abduvakhitova, Cytokine Background of Peripheral Blood - A Predictor of the Development of Herpes-Associated Erythema Multiforme Exudative, American Journal of Medicine and Medical Sciences, Vol. 15 No. 7, 2025, pp. 2125-2128. doi: 10.5923/j.ajmms.20251507.08.
Article Outline
1. Introduction
- Herpes-associated erythema multiforme exudative (HAEM) is an inflammatory disease of the skin and mucous membranes that occurs mainly against the background of reactivation of the herpes simplex virus (HSV). The disease often has a recurrent course and can significantly reduce the quality of life of patients [1,3,6,14]. The trigger is the viral load, but clinical manifestations are determined by the nature and severity of the immune response. Of particular interest is the cytokine background as a reflection of the pathogenetic processes underlying the exacerbation and chronicity of the disease [2,5,7,15].It was found that GRP (generalized recurrent herpes), preceding the development of HA MEE, is characterized by a staged course with alternating phases of remission and relapse. Immunological analysis showed that in the early stages of inflammation, cytokines of the Th2 response dominate, while the late phase and remission are associated with the activation of Th1 cells. This switch in the immune response is accompanied by a change in the production of cytokines and can be reflected in an increase in the IgG level, which, in turn, is associated with the transition to the manifest form of MEE [3,4,8,16].In this context, the cytokine profile of peripheral blood is considered not only as a reflection of the stage of inflammation, but also as a potential predictor of the transition of herpesvirus infection to a cutaneous form with epithelial damage. Proinflammatory cytokines (IFN-γ, TNF-α) and anti-inflammatory (IL-4, IL-10) play a key role in the balance between tolerance and aggression of the immune response, forming an individual risk of developing HA MEE [6,8,13,18].To date, systemic data on immune predictors of the development of HA EME are limited, which complicates targeted prevention and early immunomodulatory therapy. Studying the cytokine background in the peripheral blood of patients with GERD and HA EME will help clarify the pathogenetic mechanisms of the transition of latent herpes infection to an inflammatory skin process [3,7,11,17].Thus, the study of the cytokine profile in patients with herpes infection acquires practical significance both for the prognosis of relapses and for the development of personalized treatment regimens and prevention of HA EME. This topic is relevant from the point of view of clinical immunology, dermatology and translational medicine.Purpose of the study: To identify the features of the cytokine profile of peripheral blood in patients with herpes-associated erythema multiforme exudative (HAEME) and generalized recurrent herpes infection (GRH) in the relapse phase in order to assess their prognostic significance in the formation of cutaneous and mucosal manifestations of the disease.
2. Material and Methods
- The study included 132 patients stratified into three groups: the main group consisted of 75 patients with herpes-associated erythema multiforme exudative (HAEM) in the relapse phase; the comparative group included 22 patients with generalized recurrent herpes infection (HRI) without mucocutaneous manifestations; the control group consisted of 35 practically healthy volunteers. All participants underwent determination of the levels of key proinflammatory and anti-inflammatory cytokines in the blood serum: interferon gamma (IFN-γ), tumor necrosis factor alpha (TNF-α), interleukin-4 (IL-4) and interleukin-10 (IL-10). The study was performed by enzyme-linked immunosorbent assay (ELISA) using certified commercial kits in accordance with the manufacturer's instructions. The measurement results are expressed in pg/ml. Statistical processing of the obtained data was performed using the Statistica 10.0 software package. To assess differences between groups, Student's t-test (for normal distribution) and Mann-Whitney test (if not available) were used. Differences were considered statistically significant at p<0.05.
3. Results and Disscusion
- The above serves as the basis for determining the nature of disturbances in the state of the cytokine network of peripheral blood in patients with GA MEE during relapse and remission in comparison with the group of patients with GR.The results of the study of the cytokine profile of peripheral blood of patients with HA EME are presented in Fig. 1.
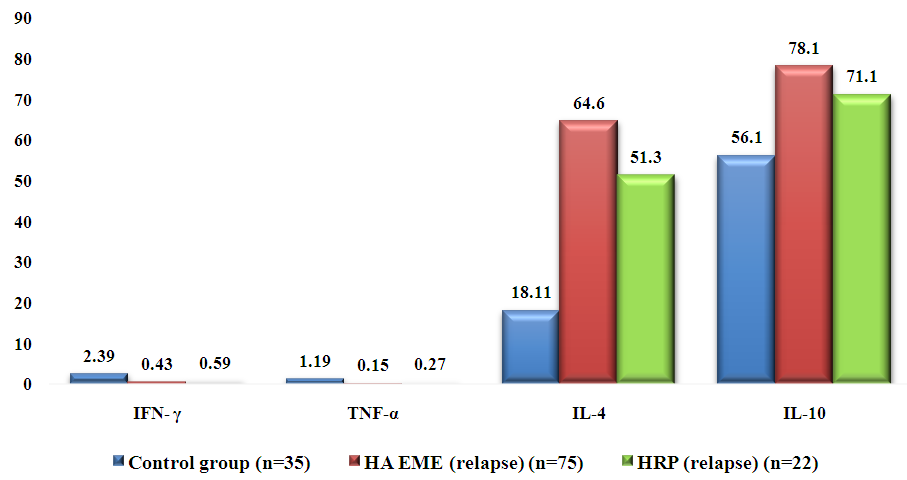 | Figure 1. Th1 and Th2 cytokine profile indices in serum of patients with HA EME during relapse pg/ml |
|
4. Conclusions
- - During the relapse of HA EME, there is an increase in the concentration of cytokines IL-4 by 3.57 times and IL-10 by 1.39 times and a decrease in the level of IFN-γ by 5.56 times, TNF-α by 7.93 times in the blood serum of patients compared to the control group;- During remission of the disease, a reliable decrease in the concentration of IFN-γ by 1.59 times, TNF-α by 1.77 times in comparison with the control group and an increase in the level of IL-4 by 1.78 times and IL-10 by 1.21 times is observed;- A clear difference in the cytokine profile of patients with HA EME from patients with GRP was revealed, which may indicate the role of cytokines in the formation of HA EME.
Study Limitations
- No significant limitations were identified in the present study. All procedures were conducted under standard conditions, and the study sample was representative of the target population. However, further research with larger and more diverse samples is recommended to validate and expand upon these findings.
ACKNOWLEDGMENTS
- The authors declare that there are no acknowledgments for this study.
Conflicts of Interest
- The authors declare no conflicts of interest.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML