Jasur Alimdjanovich Rizaev1, Otabek Sunnatovich Imamov2, Indira Nurullaevna Abduvakhitova3
1DSc, Professor, Samarkand State Medical University, Samarkand, Uzbekistan
2DSc, Project Office "Center for Healthcare Projects", Uzbekistan
3PhD, Tashkent Regional Branch of the Republican Specialized Scientific and Practical Medical Center of Dermatovenereology and Cosmetology, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
Background: Herpes-associated erythema multiforme exudative is a recurrent inflammatory condition of the skin and mucous membranes associated with herpes simplex virus reactivation. It differs from idiopathic erythema multiforme by immune system dysregulation, often leading to chronic relapses and resistance to standard antiviral therapy. Objective: To assess the immune status of patients with herpes-associated erythema multiforme exudative and compare it with those having idiopathic erythema multiforme, generalized recurrent herpes infection, and healthy individuals. Methods: A total of 190 individuals were examined: patients with herpes-associated erythema multiforme exudative (n = 75), idiopathic erythema multiforme (n = 55), recurrent herpes infection (n = 25), and healthy controls (n = 35). Laboratory evaluation included serum levels of immunoglobulin A, immunoglobulin M, immunoglobulin G, circulating immune complexes, and subpopulations of lymphocytes (T-helper, cytotoxic T, B, and natural killer cells). The cytokine profile included interferon gamma, tumor necrosis factor alpha, interleukin-4, and interleukin-10. Methods included flow cytometry and immunoassays. Statistical significance was set at p < 0.05. Results: Patients with herpes-associated erythema multiforme exudative demonstrated reduced levels of immunoglobulin A and M, elevated immunoglobulin G and circulating immune complexes, decreased T-helper and natural killer cells, and increased cytotoxic T and B cells. The T-helper to cytotoxic T-cell ratio was significantly decreased. Partial normalization of immune parameters occurred during remission. Conclusions: Herpes-associated erythema multiforme exudative is marked by distinct immune dysfunctions. Immune profiling can assist in diagnosis, monitoring, and individualized immunomodulatory treatment strategies.
Keywords:
Herpes simplex virus, Erythema multiforme, Immunoglobulins, T-lymphocytes, Cytokines, Immune complexes, CD markers, HAEM, Relapse, Immune dysregulation
Cite this paper: Jasur Alimdjanovich Rizaev, Otabek Sunnatovich Imamov, Indira Nurullaevna Abduvakhitova, Features of Immune Reactivity in Patients with Herpes-Associated Erythema Multiforme, American Journal of Medicine and Medical Sciences, Vol. 15 No. 7, 2025, pp. 2118-2124. doi: 10.5923/j.ajmms.20251507.07.
1. Introduction
Erythema multiforme exudative (EME) is an acute recurrent inflammatory disease of the skin and mucous membranes, characterized by a polymorphic rash, mainly on the skin of the extremities and face, often accompanied by disturbances in general health [1,5,22,26]. In the structure of acute dermatoses, it occupies a special place, requiring a differentiated approach to both diagnostics and therapy. According to modern data, about 70% of cases of recurrent EME are associated with reactivation of the herpes simplex virus (HSV), in connection with which the concept of herpes-associated EME (HA EME) is increasingly used in clinical practice [2,6,8,12,17].The pathogenesis of herpes-associated EME differs from other forms of the disease and is caused not by the direct action of the virus, but by immune disorders, including delayed-type hypersensitivity, the formation of autoimmune reactions, and cytokine imbalances [11,12,16,18,21,27]. Patients with HA MEE often have a tendency to a chronic relapsing course, a low response to standard antiviral therapy, and the involvement of both innate and adaptive immunity in the pathological process. Despite the availability of clinical guidelines for the treatment of EME, pathogenetic therapy often does not take into account the patient's immune profile, which reduces the effectiveness of the measures taken [3,7,9,13,15,24].A special role in the pathogenesis of herpes-associated EM is played by T-cell subpopulations, imbalance between CD4+ and CD8+ lymphocytes, as well as the activity of NK cells (CD16+) involved in the control of latent viral infection. Changes in the humoral component of immunity, including fluctuations in the levels of IgA, IgM and IgG, as well as an increase in the concentration of circulating immune complexes, can contribute to both the maintenance of chronic inflammation and tissue damage [4,10,14,23,25]. Dysregulation of proinflammatory and anti-inflammatory cytokines, such as TNF-α, IFN-γ, IL-4 and IL-10, reflects a shift in the immune response and can serve as a biomarker of the severity and frequency of relapses [19,21,26].At the same time, most studies of the immune status in EME are fragmentary, limited to individual indicators or small samples. To date, there are no general clinical and laboratory studies comparing the immune profile of patients with herpes-associated and non-herpetic forms of EME, as well as patients with recurrent herpes without signs of erythema. This significantly limits the possibilities of differential diagnosis and personalization of treatment, especially in the case of atypical or latent forms of the disease [1,6,18,24].Given the role of HSV in the development of EME, as well as the high prevalence of herpes infection in the population, the study of the immunological basis of HA EME is particularly relevant in the context of increasing immunodeficiency states, chronic stress and drug load. In addition, understanding the immune mechanisms of the disease opens up prospects for the introduction of pathogenetically substantiated approaches to therapy using immunomodulators, correction of cytokine imbalance and monitoring of treatment effectiveness using immunological markers [2,8,19,23].Thus, the study of the immune status in patients with herpes-associated erythema multiforme exudative is an urgent task of modern dermatoimmunology aimed at deepening the understanding of pathogenesis, clarifying immunological diagnostics and optimizing individualized treatment regimens.Purpose of the study: To assess the characteristics of the immune status in patients with herpes-associated erythema multiforme exudative in order to identify key immunological disorders associated with the clinical course and recurrence of the disease.
2. Patients and Methods
The study included 190 people divided into four groups: patients with herpes-associated erythema multiforme exudative (n = 75), with idiopathic EM (n = 55), with generalized recurrent herpes infection (n = 25), and a control group of practically healthy individuals (n = 35). All participants underwent a comprehensive assessment of the immune status with determination of the levels of immunoglobulins of classes A, M and G, circulating immune complexes (CIC), lymphocyte subpopulations (CD3, CD4, CD8, CD19, CD16), and the CD4/CD8 ratio. The cytokine profile included an assessment of the concentrations of IFN-γ, TNF-α, IL-4 and IL-10 in the blood serum using the ELISA method. Immunophenotyping was performed using flow cytometry, and humoral parameters were determined using enzyme immunoassay and immunoturbidimetric methods. The diagnosis of herpes-associated EME was confirmed based on clinical and anamnestic data and the results of HSV PCR testing. Statistical processing included distribution assessment using the Shapiro–Wilk test and intergroup comparison using the Student t-test or Mann–Whitney test. The level of statistical significance was taken to be p < 0.05. The data obtained allowed us to identify specific immunological features in patients with herpes-associated EME compared to other groups.
3. Results and Disscusion
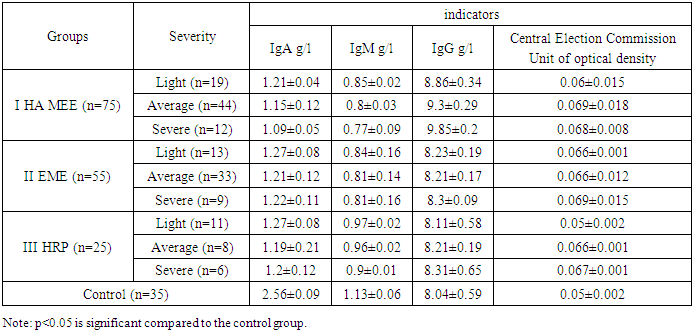
To determine the most characteristic pathological changes in the immune status in patients with GA MEE, a comparative analysis of serum immunoglobulin values was performed depending on the severity of the disease, with a comparison of the obtained values with similar values in comparison groups II and III, as well as the control group (Table 1). The results of studies conducted during a relapse are presented. Analysis of humoral immunity indicators showed that in all experimental groups there is a reliable (p<0.5) deviation from the control values according to the selected criteria. Intergroup comparison revealed a number of features.Table 1. Humoral immunity indicators in patients with HA EME depending on the severity
 |
| |
|
All patients showed changes in the parameters depending on the severity. The IgA level in patients with HA EME was the lowest in severe cases (1.09±0.05 g/l), while in mild and moderate cases the IgA concentration in the blood serum was significantly higher - 1.21±0.04 g/l and 1.15±0.12 g/l, respectively, and in the control group 2.56±0.09 g/l (p<0.05). In the comparison groups this parameter fluctuated depending on the severity of EME and HRP, and was significantly different from the control. It is interesting to note that no reliable difference was found between these groups (p>0.05).The IgM level in patients with GA MEE is significantly lower than the control values - from 0.85 ± 0.02 g / l in the mild form to 0.77 ± 0.09 g / l in the severe form compared to the control 1.13 ± 0.06 g / l (p<0.05). However, no significant difference was found between the values in HA EME and EME (p>0.05). The values of IgM concentration in the serum of patients with HRP (group III) are as close as possible to the control, although they differ significantly from them (p<0.05) - from 0.97 ± 0.02 g / l in the mild form to 0.9 ± 0.01 g / l in the severe form.Regarding the level of IgG in the blood serum of patients in all groups, they were the highest in patients with HA EME with a severe degree of the disease - 9.85 ± 0.2 g / l, significantly decreased in mild and moderate degrees of HA EME - 8.86 ± 0.34 and 9.3 ± 0.29 g / l, but still significantly exceeded the control values - 8.04 ± 0.59 g / l.In patients with EME (group II) and HRP (group III), this indicator of humoral immunity fluctuated depending on the severity, but no significant intergroup difference was found.The assessment of the CIC level showed a reliable increase in the indicator in patients with HA EME (from 0.06±0.015 units of optical density in the mild stage to 0.068±0.008 units of optical density in the severe stage compared to the control 0.05±0.002 units of density (p<0.05) and other groups, however, the dependence on the severity within the group of patients with HA EME was not determined (p>0.05).Thus, the results of the conducted studies showed that in patients with HA EME, with an increase in the severity of the pathological process, the indicators of humoral immunity change, namely, disimmunoglobulinemia develops due to a decrease in the concentration of IgA and an increase in the level of IgG in the blood serum, as well as an increase in the level of CIC.In patients with EME and HRP included in the comparison groups, similar processes also occur, but the degree of their manifestations is significantly lower.Next, the dynamics of the concentration of the main classes of immunoglobulins in patients with HA EME during relapse and remission of the disease was studied (Fig. 1).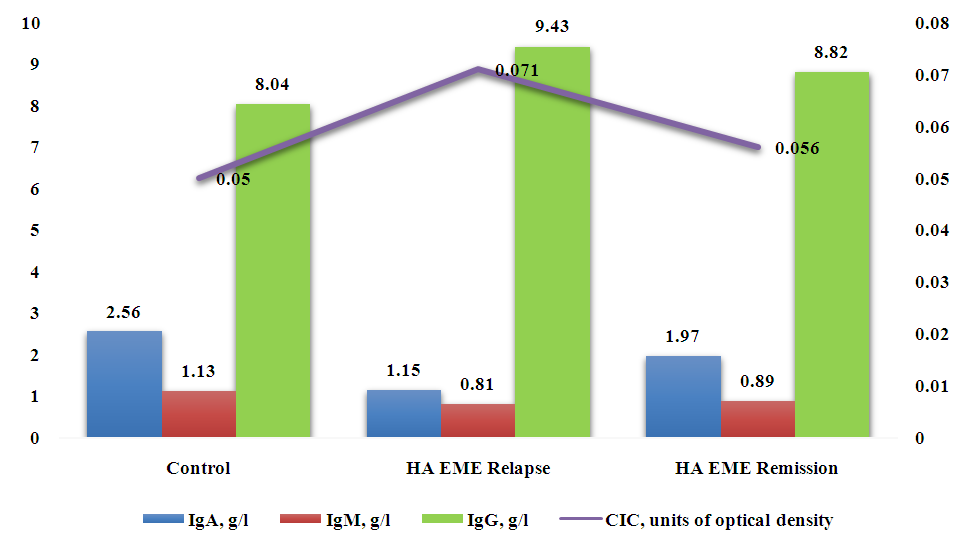 | Figure 1. Humoral immunity indicators in patients with HA EME during periods of relapse and remission of the disease (g/l) |
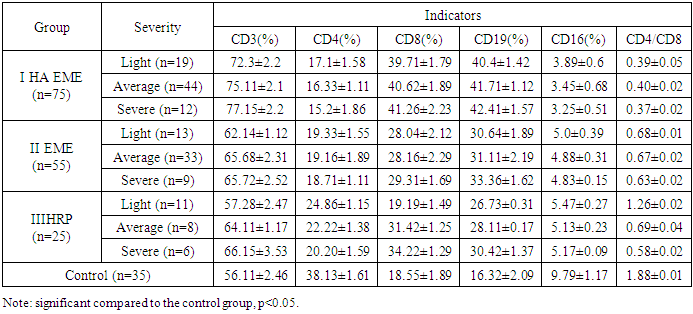
The tendency to normalize the level of the main classes of immunoglobulins in the blood serum during periods of relapse and remission is indicative. First of all, this concerns the level of IgA, when a significant decrease in the indicator during relapse - to 1.15±0.07 g/l compared to the control (2.56±0.09 g/l) increases to the level of 1.97±0.11 g/l, i.e. increases by 41.62%.The IgM level during remission did not differ significantly from the control - 0.81±0.01 g/l with control 1.13±0.06, and the differences between the levels during relapse and remission were 8.98%.The smallest fluctuations were in IgG, where the difference between relapse and remission was 6.91%, although the discrepancy in values in relapse and remission was significant (p<0.05) both when compared with each other and with the control: 9.43±0.11 g/l during relapse, 8.82±0.12 g/l during remission with a control value of 8.04±0.59 g/l.Fluctuations in the CIC level during the periods of relapse and remission reached 26.78% with absolute values of 0.071±0.014 and 0.056±0.005 with a control value of 0.05±0.002 units of optical density. In practice, it can be considered that this indicator revealed the maximum tendency to normalization during remission, since the difference with the control is not reliable (p>0.05).Consequently, during the relapse of HA EME, compared to the period of remission, there is a reliable increase in the IgG level by 1.07 times on average, although even during the period of remission the values do not reach the control values.The dynamics of IgA content in the blood serum during relapse and remission is reliable, but even during remission this indicator is significantly, 1.29 times, lower than the control. In severe cases of HA EME, the IgA content decreases by 2.22 times, the IgM content by 1.4 times, and IgG increases by 1.17 times.Compared with EME and HRP, the dynamics of IgM and IgG in HA EME from relapse to remission is less pronounced and amounts to 8.98% and 6.91%, respectively. At the same time, a comparison of the IgM level indicators in severe EME in relapse shows a decrease compared to the control by 1.5 times and, similarly, by 1.2 times in HRP, and IgA also by 2.2 times.Comparison of the average values of different classes of serum immunoglobulins in peripheral blood of patients with HA EME during the relapse of the disease with the corresponding study values in the remission stage and in the control group gives grounds to assert the development of disimunoglobulinemia due to a decrease in the concentration of IgA and an increase in the level of IgG in the blood serum, the level of the CIC. It should be noted that the CIC value is the most labile in terms of the tendency to normalize values during the remission period.To assess the state of the cellular link of immunity in patients with HA EME, a study of peripheral blood was used using the laser flow cytometry method.Comparative analysis of lymphocyte population indices depending on the severity of the disease revealed a number of characteristic pathological changes in cellular immunity in patients with HA EME. The most significant changes in the indices under study were observed in severe cases of the disease. The data are presented in Table 2.Table 2. Indicators of cellular immunity in patients with HA EME depending on the severity
 |
| |
|
In patients with HA EME, the average values of CD3+ cells differed significantly from the control and similar indicators in the comparison groups. Thus, the proportion of CD3+ cells in HA EME was 72.3±2.2% in the mild form and 77.15±2.2% in the severe form, which exceeded the control indicators by 1.28 and 1.37 times, respectively.In patients of the comparison groups, the number of CD3+ cells was also significantly higher than in the control, but less significant compared to HA EME. Fluctuations in CD3+ cells in EME and HRP differed to a lesser extent from the control: in severe EME, the number of CD3+ cells was 1.17 times higher, and in HRP of the same degree, 1.18 times higher.The average values of CD4+ cells in patients of all studied groups were significantly lower than the values in the control group, but in the group of patients with HA EME, their content was more than twice lower than the control values. Thus, the fluctuations of CD4+ cells in patients of group I were from 17.1±1.58% in the mild degree to 15.2±1.86% in the severe degree, which is 2.22 and 2.5 times lower than the control values, respectively. It should be noted that in patients with GP (group III), the content of CD4+ cells deviated less from the control and was 24.86±1.15% in the mild degree and 20.20±1.59% in the severe degree, which was close to a similar indicator in patients with EME (19.33±1.55% and 18.71±1.11%, respectively).The average values of CD8+ cells in the peripheral blood in all groups were significantly increased compared to the control, and the maximum increase was observed in HA EME - from 39.71±1.79% to 41.26±2.23% depending on the severity, i.e. in severe HA EME the CD8+ cell count exceeded the control by 2.22 times. Deviations from the control values of CD8+ cells in the group of patients with EME were the smallest, and with values from 28.04±2.12% to 29.31±1.69% (the difference in the severity of the process is insignificant, p>0.05) on average exceeded the control by 1.54 times.In parallel with this, a reliable increase in the number of CD19+ cells was revealed in each experimental group, most pronounced in HA EME with a reliable difference in severity (from 40.4±1.42% for mild to 42.41±1.57% for severe) and a reliable excess compared to groups I and II and the control.The CD16+ cell count showed a marked tendency to decrease in HA EME - 3 times in severe cases compared to the control (3.25±0.51% compared to 9.79±1.17%, respectively). The results obtained in patients with EME rank average - from 4.83±0.15% in severe cases to 5.0±0.39% in mild cases (the difference in the count by severity is not significant, p>0.05).The average value of the immunoregulatory index, T-helper/suppressor ratio (CD4+/CD8+), in patients with HA EME was recorded at a low level compared to the control and other groups; in patients in the relapse period with a severe course of the process, its value reaches 0.37±0.01. In patients with moderate and mild severity, this indicator is higher - 0.4±0.01 and 0.43±0.04, respectively, but is significantly reduced compared to the control (2.05±0.01). The same trend was observed in patients from other study groups, but the deviation from the control values is smaller. The immunoregulatory index values that are maximally close to the control in patients with HA EME range from 0.59±0.01 for a severe degree to 1.29±0.02 for a mild one. In patients with EME, the range of fluctuations of this indicator is close to that in patients with moderate and severe HRP, from 0.64±0.03 to 0.69±0.03, p>0.05.Changes in the parameters of the cellular link of immunity of patients with HA EME during relapse and remission of the disease are presented in Fig. 2.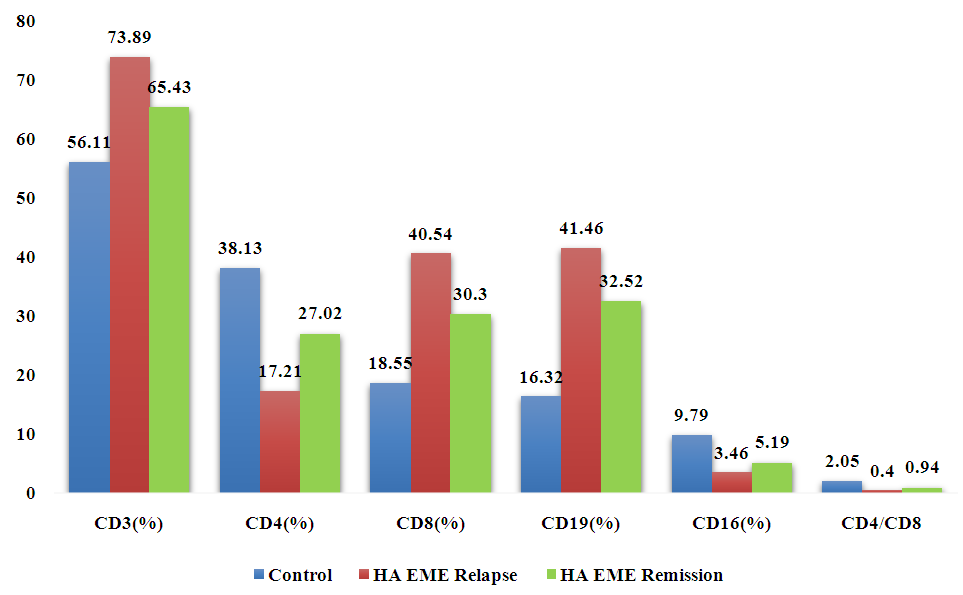 | Figure 2. Indicators of cellular immunity in patients with HA EME during periods of relapse and remission of the disease (%) |
During remission, the indices of cellular immunity in patients with HA EME changed towards normalization, but the intensity of these changes and relative results were different. Improvement was registered in terms of the CD4+-cell index - the difference between the relapse period and the state of remission is 57.0%, in terms of CD16+-cells - 50.0%, the number of CD8+-cells changes less - 33.8% and CD19+-cells - 27.5%. The immunoregulatory index increases by 2.35 times compared to the relapse, but does not reliably approach the control value. At the same time, there is data on a wide range of normal values of this index - from 0.75 to 2.39. Therefore, it is possible to assume a pronounced tendency towards normalization of the T-helper/suppressor ratio (CD4+/CD8+) in patients with HA EME in remission.The average value of the total number of T-lymphocytes, defined as CD3+ cells in the examined patients with HA EME during a relapse, was at the level of 73.89±1.41%, which is significantly higher than similar indicators in practically healthy people 56.11±2.46%. During remission, a slight decrease in this indicator is observed - 65.43±2.09%. Quantitative indices of CD4+ cells, known as inducer T lymphocytes and CD16+ population of natural killers (NK cells) in patients during relapse had reduced indices (17.21±2.59% and 3.46±0.14%) compared to remission of the disease (27.02±1.29% and 5.19±0.71%), and especially with the control group (38.13±1.61% and 9.79±1.17%).The average index of CD8+ cells (T-lymphocytes) in the peripheral blood of patients with relapse of HA EME was at the level of 40.54±1.71%, which is significantly higher than the average index of healthy individuals in the control group (18.55±1.89%) and higher than the index of the remission period (30.3±2.15%).A significant decrease in the average values of CD4+ cells during the relapse period and a decrease during the remission period and an increase in the number of CD8+ cells are reflected in a corresponding decrease in the ratio index (CD4+/ CD8+). In patients with HA EME, a reliable increase in the average total number of CD19+ lymphocytes was observed during a relapse (41.46±2.1%) and to a lesser extent during remission (32.52±2.53%) compared to the control group (16.32±2.09%).
4. Conclusions
In patients during a relapse of HA EME, in contrast to the period of remission of the disease, a reliable increase in the number of CD3+, CD8+ and CD19+ lymphocytes in the blood is observed; the value during a relapse is 73.89±1.41%, 40.54±1.71%, 41.46±2,1% andin case of disease remission - 65.43±2.09%, 30.3±2.15%, 32.52±2.53%, respectively;The period of disease relapse, in contrast to remission, is characterized by a persistent increase in the number of CD19+ B lymphocytes by 1.27 times (41.46±2.1%), a decrease in CD16+ killer cells by 1.5 times (3.46±0.14%) and CD4+ T helpers by 1.57 times (17.21±2.59%);In patients with a severe course of HA EME, during a relapse of the disease, a significant increase in the blood count of CD3+ by 1.02 times, CD8+ by 1.02 times, CD19+ by 1.03 times and a decrease in CD16+ lymphocyte cells by 1.12 times is observed compared to a mild course.
References
| [1] | Adaskevich VP. Emergency Dermatology. Moscow: Triada-Pharm; 2021: 66-69. |
| [2] | Aizyatulova RF. Viral Diseases of the Skin and Mucous Membranes: Illustrative Guide. Kyiv; 2015: 98-107. |
| [3] | Andreichin MA, Chopyak VV, Gospodarsky II. Combined Therapy for Lichen Operculum. Ternopil: Ukrmedkniga; 2017. |
| [4] | Askhakov MS, Chebotarev VV. Erythema of infectious-allergic nature. Vestn Molod Uchen. 2017; (4): 33-38. |
| [5] | Bulgakova AI, Khismatullina ZR, Zatsepina MV. Study of the incidence and clinical manifestations of erythema multiforme in the oral cavity. Stomatol Dlya Vsekh. 2017; (4): 16-18. |
| [6] | Garbuzov DA, Fedotov VP. Influence of complex therapy on immune status in women with herpesvirus and candidal infection. Ukr J Dermatol Venereol Cosmetol. 2018; (4): 73-75. |
| [7] | Gusarenko LA. A case of Lyell's syndrome following abortive erythema multiforme. Russ J Skin Venereal Dis. 2018; (3): 63-67. |
| [8] | Demyanov AV, Kotov AY, Simbirtsev AS. Diagnostic value of cytokine level studies in clinical practice. Cytokines Inflamm. 2015; 2(3): 20-35. |
| [9] | Dubossarskaya ZM, Dubossarskaya YA. Herpes infection in women: solving diagnostic difficulties. Med Asp Zhen Zdorovya. 2017; (5): 38-39. |
| [10] | Zapolsky ME. Herpesvirus Diseases (α-, β-, γ-subgroups). Odessa: Fotosintetika; 2016. |
| [11] | Zapolsky ME. Herpesvirus-associated erythema multiforme exudative: epidemiology and therapy. Klin Immunol Allergol Infektol. 2021; (8): 52-56. |
| [12] | Zapolsky ME. Therapy features of herpes-associated multiform exudative erythema. Dermatol Venereol. 2021; (4): 70-75. |
| [13] | Zapolsky ME. Immunocytokine status in herpes-associated multiform exudative erythema. Dermatol Venereol. 2018; (4): 25-33. |
| [14] | Isakov VA, Arkhipova EI, Isakov DV. Human Herpesvirus Infections: Guide for Physicians. St. Petersburg: SpetsLit; 2016: 63-75. |
| [15] | Mavrov GI, Zapolsky ME. Herpetic-associated multiform exudative erythema: therapy features. In: Clinical Settings and Unified Diagnostic Protocols in Dermatovenereology: Abstracts of the Scientific-Practical Conference, March 21–22, 2013. Kyiv; 2021: 53-54. |
| [16] | Mavrov II. Herpes Virus Infection: Clinical Forms, Pathogenesis, Treatment. Kharkiv: Fakt; 2018. |
| [17] | Mavrov II, Bolotnaya LA, Serbina IM. Fundamentals of Diagnostics and Treatment in Dermatology and Venereology: Guide for Doctors. Kharkiv: Fakt; 2017. |
| [18] | Sergeev AY. Adverse allergic reactions to drugs in dermatology. Immunopatol Allergol Infektol. 2017; (3): 45-67. |
| [19] | Ayangco L, Rogers RS. Mucosal manifestations of erythema multiforme. Dermatol Clin. 2018; 21: 195-205. |
| [20] | Dames S, Bromley LK, Herrmann M, et al. Single-tube nucleic acid extraction and detection using aluminum oxide. J Mol Diagn. 2016; 8: 16-21. |
| [21] | Kats J, Livneh A, Shemer J, Danon Y. Herpes simplex virus-associated erythema multiforme: clinical and therapeutic dilemma. Pediatr Dent. 2019; 21(6): 359-362. |
| [22] | Kenneth S. Oral manifestations of erythema multiforme. Oral Surg Oral Med Oral Pathol. 2018; 25: 366-373. |
| [23] | Kokuba H, Aurelian L, Burnett JW. Herpes simplex virus-associated erythema multiforme is distinct from drug-induced erythema: IFN-γ and TNF-α expression patterns. J Invest Dermatol. 2019; 113: 808-815. |
| [24] | Kokuba H, Imafuku S, Burnett J, Aurelian L. Longitudinal study in a patient with herpes-associated erythema multiforme. Dermatology. 2019; 198(3): 233-242. |
| [25] | Kokuba H, Kauffman CL, Burnett JW, Aurelian L. Clinical and virologic comparison of erythema multiforme cases. Acta Derm Venereol. 2019; 79: 247-248. |
| [26] | Novikov DK, Sergeev YV, Novikov PD, Al-Ubaydi SS, Nalli FF. Erythema multiforme: review of 26 cases. Oral Surg Oral Med Oral Pathol. 2016; 41: 601-606. |
| [27] | Zohdi-Mofid M, Horn TD. Acrosyringeal concentration of necrotic keratinocytes in erythema multiforme: clue to drug etiology. J Cutan Pathol. 2017; 24: 235-240. |




 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML
