-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(7): 2114-2117
doi:10.5923/j.ajmms.20251507.06
Received: May 12, 2025; Accepted: Jun. 9, 2025; Published: Jul. 3, 2025

Comparative Analysis of Iron Metabolism Parameters in Children with Chronic Kidney Disease with and without Anemia
Ashurova Noila Shuxratovna
Zarmed University, Samarkand, Uzbekistan
Correspondence to: Ashurova Noila Shuxratovna, Zarmed University, Samarkand, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Anemia is among the most common and clinically significant complications of chronic kidney disease (CKD) in children. It substantially worsens quality of life and accelerates disease progression. This study aims to compare laboratory markers of iron metabolism in children with CKD, both with and without anemia, to identify early predictors and to better understand the relationship between inflammation and iron homeostasis.
Keywords: Chronic kidney disease, Anemia, Children, Ferritin, Transferrin, Hepcidin
Cite this paper: Ashurova Noila Shuxratovna, Comparative Analysis of Iron Metabolism Parameters in Children with Chronic Kidney Disease with and without Anemia, American Journal of Medicine and Medical Sciences, Vol. 15 No. 7, 2025, pp. 2114-2117. doi: 10.5923/j.ajmms.20251507.06.
1. Introduction
- Anemia is a common complication in children with chronic kidney disease (CKD), associated with increased morbidity and decreased quality of life [1]. Diagnosing anemia in CKD is challenging due to the effects of inflammation on iron metabolism, which leads to a distinct manifestation of iron deficiency. Unlike classical iron deficiency, anemia in CKD often develops in the context of chronic inflammation, which is reflected in alterations in laboratory markers such as ferritin and hepcidin.According to international and regional studies, anemia occurs in more than 50–70% of pediatric patients with CKD, and its prevalence increases with disease progression [2,3,10,14]. This high incidence, combined with the multifactorial nature of anemia in CKD — including impaired erythropoietin production, iron dysregulation, and persistent inflammation — necessitates a deeper understanding of its pathophysiology.The importance of comparative analysis lies in distinguishing anemia caused by absolute iron deficiency from that associated with functional iron deficiency in CKD, where iron is sequestered despite normal or elevated ferritin levels [12,13]. By evaluating iron metabolism markers in both CKD-related and non-renal anemia, this study aims to enhance diagnostic precision and guide more effective treatment strategies in pediatric patients.The objective of this study is to perform a comparative analysis of laboratory markers of iron metabolism in children with CKD with and without anemia. We examine parameters such as hemoglobin, serum iron, ferritin, transferrin, and hepcidin levels to identify differences between the two patient groups and to better understand the mechanisms underlying anemia in CKD.It should also be noted that the analysis of iron status in children with CKD is essential for assessing not only iron deficiency but also the degree of inflammation, which may obscure its detection. Therefore, the identification and interpretation of laboratory parameters within the context of CKD are crucial for accurate diagnosis and the selection of optimal therapeutic approaches [4,5].
2. Materials and Methods
- The study included children with anemia diagnosed with stage 3 CKD (n = 30) and children with anemia without signs of renal pathology (n = 30). The following laboratory parameters were evaluated: hemoglobin, serum iron, ferritin, total iron-binding capacity (TIBC), transferrin and its iron saturation (TSAT), and hepcidin. Statistical analysis was performed using the Student's t-test and the Mann–Whitney test. A p-value < 0.05 was considered statistically significant.
3. Results
- The results of the laboratory analysis revealed marked differences between children with anemia and CKD, children with anemia without renal pathology, and healthy controls (Table 1). These differences provide a more detailed understanding of the characteristics of anemia in the context of CKD.
|
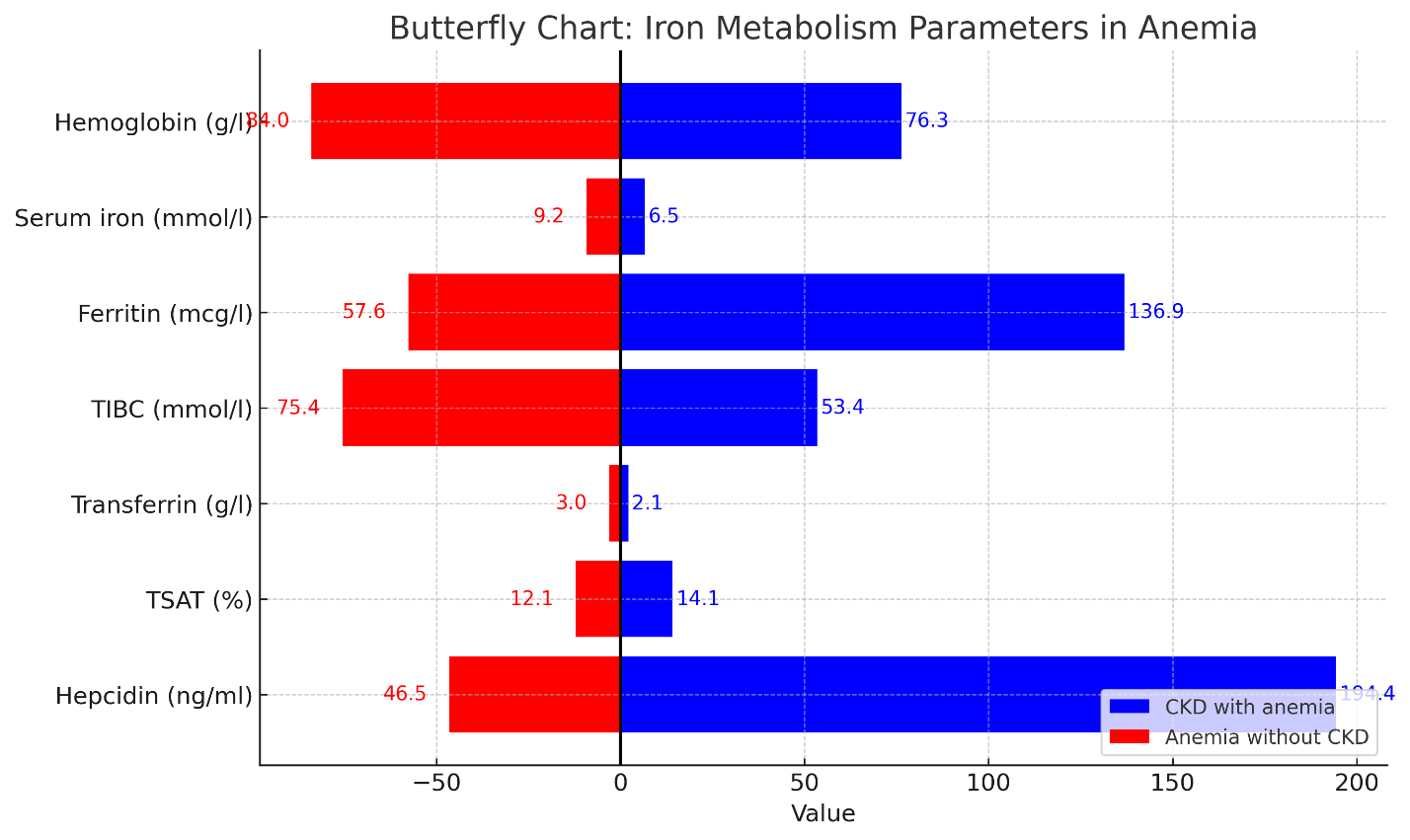 | Figure 1. Butterfly chart showing comparative values of iron metabolism markers in children with anemia associated with CKD and without renal pathology |
4. Discussion
- Children with anemia associated with chronic kidney disease (CKD) exhibit a significantly more pronounced reduction in hemoglobin and serum iron levels compared to children with anemia without renal pathology, indicating a more severe and complex form of anemia related to CKD. The presence of such pronounced anemia in children at stage 3 CKD underscores the fact that hematologic complications begin to intensify even before the advanced stages. Although anemia typically worsens in stages IV–V, the disturbances in iron regulation and elevated hepcidin levels observed in this study already manifest at stage 3, supporting the need for early detection and targeted management.These findings suggest the involvement of additional pathogenic mechanisms beyond simple iron deficiency. Elevated ferritin levels observed in children with CKD do not necessarily reflect adequate iron stores but instead point to an underlying chronic inflammatory state, leading to a condition known as functional iron deficiency. In this state, iron becomes sequestered and unavailable for effective erythropoiesis. The markedly increased hepcidin concentrations further support this mechanism, as hepcidin functions as a negative regulator of iron absorption and release, thereby exacerbating anemia by limiting iron bioavailability [11,13,15].Furthermore, the reduction in transferrin levels in the CKD group may be attributed to suppressed hepatic protein synthesis, a common consequence of systemic inflammation. Finally, the observed decline in transferrin saturation in both CKD-related anemia and classical iron deficiency anemia indicates a restricted iron supply for erythropoiesis, with a more profound impact in the presence of inflammatory dysregulation of iron metabolism.
5. Conclusions
- Anemia in pediatric chronic kidney disease presents a complex, mixed etiology and arises in the context of chronic inflammation, as well as impaired iron transport and utilization. This distinguishes it fundamentally from classical iron deficiency anemia. While both study groups demonstrated reduced levels of hemoglobin, serum iron, and transferrin saturation, the underlying mechanisms were notably different.In children with stage 3 CKD, anemia was predominantly functional, as indicated by elevated ferritin and markedly increased hepcidin levels, along with decreased transferrin — all of which point to inflammation-driven iron sequestration and reduced iron bioavailability. In contrast, children with anemia without CKD displayed laboratory features consistent with absolute iron deficiency, including low ferritin, high TIBC, and normal hepcidin levels.These findings emphasize the importance of distinguishing between absolute and functional iron deficiency in clinical practice. Accurate identification of the underlying mechanism is essential for selecting an appropriate therapeutic strategy. Although anemia typically progresses with advancing CKD stages, the current data demonstrate that significant disruptions in iron regulation and the onset of functional iron deficiency already occur at stage 3. This underscores the importance of early diagnostic screening and timely intervention.A comprehensive diagnostic approach—including the assessment of ferritin, transferrin, TSAT, and especially hepcidin—should be integrated into routine evaluation of pediatric anemia, particularly in children with CKD, to ensure individualized and effective treatment.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML