-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(7): 2110-2113
doi:10.5923/j.ajmms.20251507.05
Received: May 10, 2025; Accepted: Jun. 2, 2025; Published: Jul. 3, 2025

High Prognostic Risk MDS: Comparative Evaluation of Treatment Efficacy
Zulhumor Dzhumabaevna Yunusova
2nd Clinical Department of Hematology, Republican Specialized Scientific-Practical Medical Center of Hematology (RSSPMCH) MoH RUz, Tashkent, Uzbekistan
Correspondence to: Zulhumor Dzhumabaevna Yunusova, 2nd Clinical Department of Hematology, Republican Specialized Scientific-Practical Medical Center of Hematology (RSSPMCH) MoH RUz, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Objective: To comparatively evaluate the effectiveness of using Cytarabine and Azacitidine in patients with high prognostic risk MDS. Material and Methods. The study was based on voluntary participation of 68 adult patients with myelodysplastic syndrome (MDS) (median age 51.0±1.7 years). Patients were selected at admission to the Republican Specialized Scientific and Practical Medical Center of Hematology (RSSPMCH, Tashkent) from 2014 to 2024. Conclusion. More promising as the most effective method of treating patients with high prognostic risk MDS is the use of Azacitidine.
Keywords: Myelodysplastic syndrome, High prognostic risk, Chemotherapy, Azacitidine, Cytarabine
Cite this paper: Zulhumor Dzhumabaevna Yunusova, High Prognostic Risk MDS: Comparative Evaluation of Treatment Efficacy, American Journal of Medicine and Medical Sciences, Vol. 15 No. 7, 2025, pp. 2110-2113. doi: 10.5923/j.ajmms.20251507.05.
Article Outline
1. Introduction
- Myelodysplastic syndrome (MDS) is an unexplored tumor disease of the hematopoietic system that develops at any age, including children. MDS mainly affects the elderly population [1,4,9,13]. According to WHO statistics, MDS is diagnosed in 86% of cases in people aged 60 years and older [2,3,8]. Along with this, the estimated incidence of MDS increases significantly with age: in the fourth decade of life from 0.7 per 100,000 adults, and after 70 years of age to 10.8-36.3 per 100,000 adults [3,9]. The risk of developing the disease increases fivefold between the ages of 60 and 80 years [3]. As the elderly population grows, world statistics estimate that by 2025 there will be 1.2 billion people over 60 years of age, and therefore there will be an increase in the incidence of MDS in the general population [1,3].Patients diagnosed with MDS experience a deterioration in quality of life, jeopardizing the ability of elderly patients to independently perform activities of daily living [5,7,11,14]. Depending on the severity of MDS and the degree of cytopenia, patients present with excessive fatigue, night sweats, bone pain, fever, weight loss, recurrent infections, and hemorrhagic events in the form of bruising, bleeding, and skin rashes [6,10,12,15]. There are data showing the effect of different treatment methods on the course of MDS [16,18,19,21]. Some foreign studies have shown the effect of treatment with erythropoiesis-stimulating drugs and granulocyte colony-stimulating factor on the quality of life of MDS patients before and after treatment [17,20,24,28]. Treatment options for patients with MDS are very limited, and there are many factors that increase the likelihood that they will not receive active treatment for MDS [22,23].Age-related comorbidities, functional impairment, poor tolerability, ineffectiveness of available treatments, and patient preference may influence whether a physician recommends active treatment for a patient [25,27]. Results of large studies regarding the treatment of patients with MDS showed that 27% of high-risk patients with newly diagnosed disease and 24-49% of high-risk patients with established disease received only maintenance replacement therapy [26]. Meanwhile, although the main goal of currently available therapies is to eliminate cytopenia, there is currently conflicting data regarding the use of drugs of the pyrimidine nucleoside cytidine analogs azacytidine (Aza) in the treatment of patients with MDS.Thus, the choice of therapy has a crucial role in improving the quality of life of patients with MDS of high prognostic risk. Timely correct choice of therapy for patients with MDS will contribute to the reduction of complications of the disease, reduce the incidence of MDS transformation into acute leukemia, improve the relapse-free overall survival and quality of life of patients.Aim of the study. To conduct a comparative evaluation of the efficacy of cytarabine and azacitidine in patients with high prognostic risk myelodysplastic syndrome (MDS).
2. Main Body
2.1. Material and Methods of Study
- The study was based on voluntary participation of 68 (1st main group) adult patients with MDS aged 18 to 85 years, with an age median of 51.0±1.7 years. Patients were selected upon admission to the Republican Specialized Scientific and Practical Medical Center of Hematology (RSNPMCH, Tashkent) from 2014 to 2024. The diagnosis was established taking into account clinical manifestations, changes in the clinical blood count, myelogram, taking into account diagnostic standards (Oster H. S., Mittelman M., 2024). Based on the results of the analyses performed in patients with MDS, all subjects were categorized into groups based on the method of treatment (Table 1).
|
2.2. Results of the Study
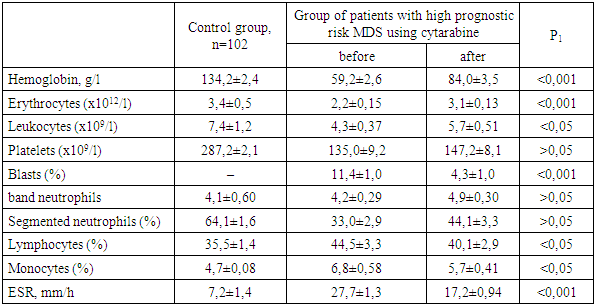
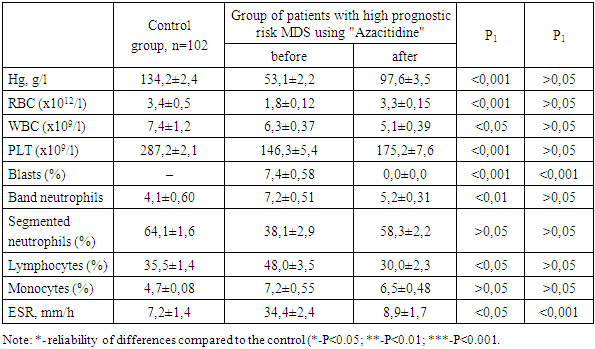
- We carried out a comparative analysis of the effectiveness of the conducted treatment methods with cytarabine (n=32) and azacitidine (n=36) according to the studied indicators of the general blood analysis and myelogram (Table 2).
|
|
3. Conclusions
- The results of the analysis of the dynamics of peripheral blood parameters and myelogram in patients with MDS showed a variety of hematological disorders characterized by erythrocytopenia and thrombocytopenia, accompanied by a decrease in the number of mature neutrophils and simultaneous lymphocytosis.Comparison of treatment methods for patients revealed a decrease in blast cells and an increase in the number of erythrocytes, platelets, and neutrophils with the use of Cytarabine and Azacitidine. In addition, among patients with high-risk MDS, the release of blast cells into the peripheral blood was maintained with the use of Cytarabine due to the excess of the permissible values of blast cells in the myelogram after treatment with Cytarabine. At the same time, the use of Azacitidine treatment courses in patients with high-risk MDS resulted in a more pronounced recovery of blood and myelogram parameters with a decrease in blast cells to normal values in the bone marrow.Thus, from the above results, it can be concluded that the use of "Cytarabine" and "Azacitidine" in patients with high prognostic risk MDS is observed to have positive dynamics in terms of peripheral blood and myelogram indices. However, the preservation of the increased number of blast cells, both in the general blood test and in the myelogram, as well as less pronounced recovery of other indices against the background of the use of "Cytarabine", compared with the effectiveness of changing similar indices after the use of "Azacitidine", which indicates its lower effectiveness. Consequently, the use of "Azacitidine" is more promising as the most effective method of treating patients with high prognostic risk MDS.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML