-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(7): 2104-2109
doi:10.5923/j.ajmms.20251507.04
Received: May 9, 2025; Accepted: Jun. 6, 2025; Published: Jul. 3, 2025

Study of the Relationship Between Detected Cytogenetic Changes and the Severity of the Clinical Course of Myelodysplastic Syndrome in Uzbekistan
Zulkhumor DzhumabaevnaYunusova
2nd Clinical Department of Hematology, Republican Specialized Scientific-Practical Medical Center of Hematology (RSSPMCH) MoH RUz, Tashkent, Uzbekistan
Correspondence to: Zulkhumor DzhumabaevnaYunusova, 2nd Clinical Department of Hematology, Republican Specialized Scientific-Practical Medical Center of Hematology (RSSPMCH) MoH RUz, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Objective: To analyze cytogenetic abnormalities in patients with myelodysplastic syndrome (MDS). Material and methods. The study was based on the voluntary participation of adult patients with MDS (n=72) aged 18 to 85 years (median age 51.0±1.7 years). Patients were selected on admission for examination at the Republican Specialized Scientific and Practical Medical Center of Hematology (RSSPMCH, Tashkent city) during the time period: 2014-2023. The research methods included: clinical, laboratory, cytogenetic and statistical research methods. Conclusion. The results of cytogenetic studies indicate the importance of chromosomal abnormalities for the early detection of prognostically unfavorable variants of MDS, confirming the need for karyotyping in a comprehensive examination of primary patients.
Keywords: Myelodysplastic syndrome, Cytogenetic abnormalities, Chromosome, Deletion, Translocation, Inversion
Cite this paper: Zulkhumor DzhumabaevnaYunusova, Study of the Relationship Between Detected Cytogenetic Changes and the Severity of the Clinical Course of Myelodysplastic Syndrome in Uzbekistan, American Journal of Medicine and Medical Sciences, Vol. 15 No. 7, 2025, pp. 2104-2109. doi: 10.5923/j.ajmms.20251507.04.
Article Outline
1. Introduction
- Myelodysplastic syndromes (MDS) are a poorly understood heterogeneous group of diseases with abnormal genetic information in hematopoietic stem cells (HSCs) [21,27,28,32], characterized by ineffective hematopoiesis, dysplastic processes in the bone marrow, resulting in cytopenia in the peripheral blood [15,18,27]. MDS is characterized by a high predisposition to transformation into acute myeloid leukemia (AML) [3,12,16,26].MDS is usually a primary disease, but secondary cases of MDS are also known to occur as a result of chemotherapy or radiation exposure [5,12].Despite the lack of precise data regarding the causative factor of primary MDS and its pathogenetic mechanisms of formation, it is known that the development of the disease is a multi-stage process, beginning with genetic disorders and associated with chromosomal aberrations, including deletion 5q, deletion 7q or monosomy 7, trisomy 8, etc. [1,31].The epidemiology of MDS also remains poorly studied, although the literature reports that in highly developed countries the incidence rate averages 3–4 cases per 100,000 population per year with a tendency to increase with age [30,33]. According to foreign authors, MDS develops more often in men than in women [4,13,17]. However, cases of the disease accompanied by a 5q deletion are more common in women [15,18].With significant variability in the course of MDS, it can have the character of a sluggish disease or, on the contrary, acquire an aggressive character with an increased risk of transformation into AML and a low life expectancy of patients [2,14,19].The results of recent studies show that cytogenetic abnormalities play a significant role in worsening the prognosis of MDS [7,9,26]. Cytogenetic abnormalities are detected in the form of deletions, translocations, inversions and other rearrangements in the karyotype of approximately 50% of patients with primary MDS, and in 80% of cases with secondary MDS [16,22,29].According to the criteria of the Revised International Prognostic Scoring System (IPSS-R) for Myelodysplastic Syndrome (MDS), the detection of cytogenetic abnormalities is of crucial importance for determining the prognosis of the disease, which allows not only to predict the clinical course of MDS, but also to plan an individual treatment strategy for each patient [6,7,8,10,11,22].It should be noted that the global data obtained to date on cytogenetic abnormalities in patients with MDS are numerous and not systematized [23,25,29].Clinical significance in relation to MDS has been determined only for a small number of cytogenetic abnormalities, including -5/5q-, -7/7q-, i(17q)/t(17p), -13/13q-, 11q-, 12p-/t(12p), 9q-, idic(X)(q13), t(11;16), t(3;21), t(1;3), t(2;11), inv(3)/t(3;3), t(6;9).Karyotype anomalies +8, 20q-, -Y are also often detected in MDS, but without morphological criteria they are not a reason to verify this diagnosis. [2,24]. The role of most non-random karyotype anomalies has not been studied to date due to the low frequency of occurrence of each of them. At the same time, it is known that the frequency of occurrence of certain cytogenetic disorders has population-regional differences [20,31].Thus, karyotype analysis is aimed at identifying chromosomal abnormalities. Evaluation of the significance of each individual cytogenetic abnormality in the clinical course of myelodysplastic syndrome is crucial for early detection of high prognostic risk MDS and selection of an individual strategy for treating patients. Timely and correct choice of treatment tactics for patients with MDS, taking into account the results of karyotyping, will help reduce complications of the disease, decrease the frequency of transformation of MDS into acute leukemia, and increase relapse-free overall survival and quality of life of patients.The aime of this study is to analyze cytogenetic abnormalities in patients with MDS.
2. Main Body
2.1. Material and Methods of Study
- The study was based on the voluntary participation of adult patients with MDS aged 18 to 85 years (age median 51.0±1.7 years). Patients were selected upon admission for examination at the Republican Specialized Scientific and Practical Medical Center of Hematology (RSSPMCH, Tashkent) from 2014 to 2023. The diagnosis was established taking into account diagnostic standards based on clinical manifestations, changes in the clinical blood test and myelogram [18,21].Depending on the prognostic risk according to the R-IPSS system [22], MDS patients were divided into three subgroups.1st subgroup – patients with low risk (very low and low risk);2nd subgroup – patients with intermediate risk (intermediate risk);3rd subgroup – patients with high risk (high and very high risk).As part of diagnostic studies, 72 patients with MDS underwent karyotype testing using standard cytogenetic testing (SCT) using GTG banding technology. The material for karyotyping (SCT) was bone marrow cells obtained by sternal puncture. Karyotyping (SCT) included the following steps: collection of bone marrow in a volume of 2-3 ml, followed by adding the bone marrow to a separate test tube with a culture medium containing: 4.0 ml of RPMI medium with glutamine, 1 ml of fetal calf serum, 0.5 μl of colchicine (0.01% solution). The cultured cells were incubated for 17 to 24 hours at a temperature of +37.5°C in a hermetically sealed test tube in a horizontal position. After incubation, the cells were precipitated by centrifugation, the sediment was broken up by vigorous shaking of the test tube, after which 6-8 ml of hypotonic solution was added in parts, mixed by pipetting and incubated for 20-25 minutes at +37.5°C.After hypotonization - without centrifugation and removal of the supernatant - prefixation of the cell suspension was performed using Carnoy's fixative (ethanol + glacial acetic acid in a ratio of 3:1), adding the fixative in an amount of 0.5 ml per test tube. The contents of the tubes were then mixed and the cells were precipitated by centrifugation (7 min, 1500 rpm). The supernatant was removed with a pipette, the sediment was thoroughly broken up by shaking, while simultaneously adding the fixative dropwise, wall-by-wall. The contents of the test tubes were mixed, after which the test tubes were placed in a freezer (–4 - –5°C) for 10 minutes.After the specified time, the cooled cell suspension was centrifuged and 5-6 ml of freshly cooled fixative was added to the sediment, mixed and placed in a freezer for 15 minutes. This procedure with replacement of the retainer was carried out 2-3 times. The last portion of the fixative added was 1.0–0.5 ml. Preparation of chromosome preparations was carried out by dropping the resulting suspension (150-200 µl) from a height of 30-50 cm onto defatted, cooled glass slides.Slides with chromosome preparations were dried at room temperature for 24 hours. Staining of the preparations was performed using the GTG banding method, for which the slides were treated with 0.25% trypsin solution (the time was selected empirically in the range of 1-60 seconds) and stained (the time was selected empirically in the range of 1.5-2 minutes) with a solution of Romanovsky-Giemsa dye diluted in phosphate buffer (pH = 6.8). The dye was washed off with running water, after which the preparations were dried and examined microscopically. The search for metaphases is carried out at a magnification of X200, and photo archiving and analysis of metaphase plates is carried out at a magnification of X1000.For cytogenetic analysis of archived photographic images of metaphase plates, the computer system for analyzing images of metaphase chromosomes “VideoTestKaryo 3.1” was used. At least 20 metaphase plates were analyzed for each patient. Chromosome classification and karyotype pathology interpretation were performed in accordance with the International Nomenclature of Differentially Segmented Chromosomes (ISCN, 2016).For statistical processing of the obtained digital data, calculations were used using Excel and Statistica programs, while the established differences between the studied indicators were considered reliably significant at p≤0.05.
2.2. Results of the Study
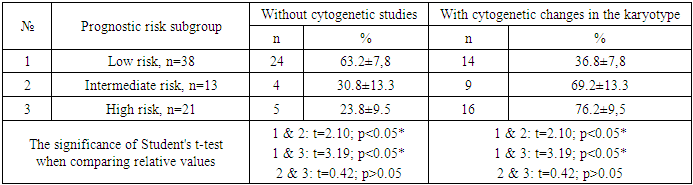
- Karyotyping (SCT) was performed in 72 patients with MDS aged 18 to 82 years, among whom patients aged ≤50 years accounted for n=28 (38.9%), patients aged 51 to 60 years – n=14 (19.4%), and patients older than 60 years – n=30 (41.7%).The study showed that among MDS patients who underwent karyotyping (n=72), chromosomal changes were detected in 39 examined patients (54.2%). When evaluating the results of karyotyping (SCT) depending on the prognostic risk of MDS according to the R-IPSS system, it was found that among the subgroup of MDS patients with low risk (n=38), cytogenetic changes were absent in n=24 (63.2%) patients, while the presence of chromosomal abnormalities was noted in n=14 (36.8%) cases. In the subgroup of MDS patients with intermediate risk (n=13), similar indicators were n=4 (30.8%) and n=9 (69.2%) cases, while among patients with high risk (n=21), cases with and without chromosomal abnormalities were n=5 (23.8%) and n=16 (76.2%), respectively.Comparative analysis of the proportion of MDS patients without cytogenetic changes in the subgroups of low prognostic risk (63.2%) and intermediate prognostic risk (30.8%) revealed a significant difference in the indicator (p<0.05). Similar data were noted when comparing the subgroups of low (63.2%) and high (23.8%) risks (p<0.05). The obtained result indicates the presence of an inverse relationship between the risk and the normal state of the karyotype: as the risk of an unfavorable course of MDS increases, the probability of detecting a normal karyotype without cytogenetic changes decreases (Table 1).
|
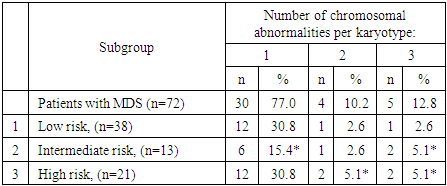
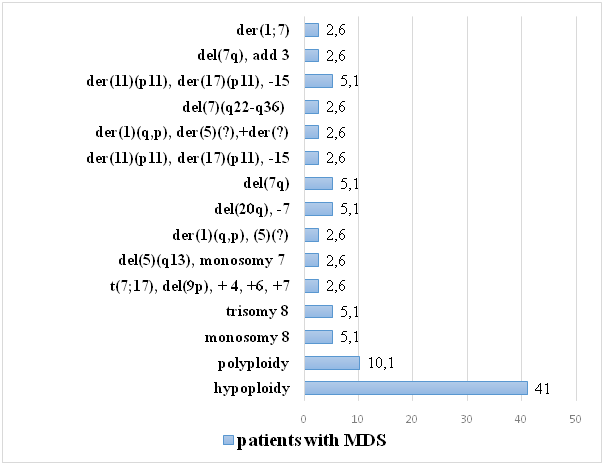 | Figure 1. Spectrum and frequency of chromosomal changes detected in patients with MDS |
|
|
|
|
3. Conclusions
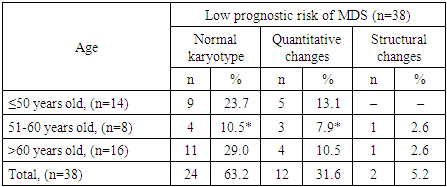
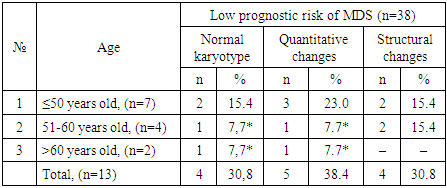
- The results of cytogenetic analyses in patients with MDS show a variety of chromosomal abnormalities that make a pathogenetic contribution to the formation of a prognostically unfavorable high risk of the disease. The study revealed a significantly lower incidence of chromosomal changes among MDS patients with low prognostic risk compared to intermediate and high risk groups. In addition, among the identified chromosomal abnormalities, the most common quantitative chromosomal changes are hypoploidy (41.0%) and polyploidy (10.1%). In addition, it was found that in patients aged 51 to 60 years and over 60 years, normal karyotype and quantitative chromosomal changes are significantly less common compared to patients under 50 years old, indicating a more aggressive course of MDS among patients under 50 years old. A less aggressive course of MDS among patients in the older age group (>60 years) is associated with the absence of cases with known prognostically unfavorable structural chromosomal changes.Thus, the greatest number of MDS cases with normal karyotype were detected in patients from the group with low prognostic risk according to R-IPSS, while various cytogenetic changes were more often detected among patients with intermediate and unfavorable risks. Given the diversity of the picture of chromosomal abnormalities, including individual single anomalies, timely determination of the prognostic risk of the disease and the correct choice of treatment strategy require mandatory SCT in each patient with MDS.The presented results indicate the necessity and expediency of timely standard cytogenetic analysis (karyotyping (SCT)) in primary patients with MDS. The conclusion on the state of the karyotype of a patient with MDS will improve the understanding of the genesis of the disease, will allow to correctly assess the prognostic risk, which is extremely important for determining the strategy of treatment measures.The results of the study will be useful in practical healthcare when implementing measures for the primary diagnosis of MDS in order to early identify prognostically unfavorable high risk of the disease.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML