-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(6): 1844-1850
doi:10.5923/j.ajmms.20251506.44
Received: May 10, 2025; Accepted: Jun. 6, 2025; Published: Jun. 13, 2025

Formation of Urogenital Pathology in Women of Fertil Age with Chronic Brucellosis
Yarmuxamedova N. A., Djuraeva K. S.
Chair of Infectious Diseases, Samarkand State Medical University, Samarkand, Uzbekistan
Correspondence to: Yarmuxamedova N. A., Chair of Infectious Diseases, Samarkand State Medical University, Samarkand, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Brucellosis is a chronic zoonotic infection that significantly affects women of reproductive age, particularly in endemic regions. This study explores the formation of urogenital pathology in women with chronic brucellosis, highlighting its clinical manifestations and reproductive consequences. The infection commonly leads to menstrual dysfunction, infertility, spontaneous abortions, and adverse pregnancy outcomes. Brucella spp., especially Brucella melitensis, can persist in reproductive tissues, causing chronic endometritis, ovarian inflammation, and hormonal imbalance. Histopathological changes include follicular atrophy, luteal persistence, and vascular damage in ovarian tissue.Up to 26% of affected women develop infertility, while spontaneous abortion rates may reach 40%, primarily in early pregnancy. Pregnancy often exacerbates latent infection, increasing the risk of nephropathy, hypotonic bleeding, and placental pathology, including infarctions and vasculitis. Diagnosis is challenging due to nonspecific symptoms and often requires a combination of epidemiological history, serological tests, and immunological assays. Treatment with prolonged antibiotic regimens—such as doxycycline and rifampicin—is essential to prevent chronic complications.The study emphasizes the need for brucellosis screening in women with reproductive issues in endemic areas and supports integrated public health measures, including veterinary control and patient education, to reduce transmission and long-term reproductive harm.
Keywords: Brucellosis, Infertility, Pregnancy, Diagnosis, Screening
Cite this paper: Yarmuxamedova N. A., Djuraeva K. S., Formation of Urogenital Pathology in Women of Fertil Age with Chronic Brucellosis, American Journal of Medicine and Medical Sciences, Vol. 15 No. 6, 2025, pp. 1844-1850. doi: 10.5923/j.ajmms.20251506.44.
Article Outline
1. Introduction
- Brucellosis (Malta fever, Mediterranean fever, Gibraltar fever, Cyprus fever, undulating fever, typhoid fever, Bang's disease) is a particularly dangerous and socially significant infection that causes significant economic damage and causes a high level of disability among patients [1,5].The causative agent of brucellosis in humans and animals belongs to the bacteria of the genus Brucella [2,8].Brucella are very small gram-negative microorganisms, characterized by great polymorphism, having a spherical, ovoid or rod-shaped form. They grow well on nutrient media. There are several forms of Brucella. The S-form is considered normal, but under unfavorable conditions brucellae form R-, M- and MVB-forms. It is possible to form an L-form, characterized by the peculiarities of metabolism and antigen structure. Under the influence of various unfavorable factors, treatment with antibiotics, the morphological, cultural and tinctorial properties of Brucella change, even forms incapable of agglutination arise [5,11,18].Brucella are resistant in the external environment. They survive in tap water for up to 76 days, in raw milk - up to 40, in sheep's milk cheese - 60, in butter, cream, sour milk - throughout the entire period of their nutritional value. In raw meat of sick animals, brucella survive up to 3 months, in wool - up to 3-4 months [3,7,19]. However, brucella are very sensitive to the effects of various disinfectants. They die instantly when boiled. The main role in infecting people is played by Br. Melitensis.Brucellosis is a typical zoonotic disease, humans are a biological dead end – a sick person is not a source of infection. This infection is characterized by multiple routes of infection and a pronounced professional nature of the disease [8].The sources of infection are small and large cattle, pigs, and in some places reindeer. The most important epidemiological significance belongs to small cattle due to the obligate pathogenicity of Br. Melitensis for humans. Epizootics of brucellosis among sheep and goats entail epidemic outbreaks of this disease in humans. Br. Abortus bovis, Br. Abortus Suis are less pathogenic for humans. For this reason, cattle and pigs are of lesser epidemiological importance [11].When several types of sick and healthy animals are kept or grazed together, there is an exchange of different types of brucellae between them. In this process, the migration of Br. Melitensis from small cattle to cattle and other types of animals acquires great epidemiological significance. In most cases, brucellosis in animals during pregnancy leads to abortion, and the fetus, its membranes, amniotic fluid, and placenta contain a large amount of the pathogen [6].In animals, brucellosis is excreted in urine, feces and milk, and in aborted animals, in amniotic fluid.In the transmission of infection from animals to humans, meat and raw dairy products (milk, feta cheese, and cheese) are of great epidemiological importance. In raw dairy products, brucellae survive for 15-60 days, in meat - up to 20 days [3,8].The excretions of sick animals containing brucellosis contaminate wool, feed, bedding in stalls, soil, water, etc. All these infected objects serve as factors for the transmission of the pathogen.Human infection usually occurs through alimentary or contact routes. However, an airborne mechanism of infection is also possible, caused by the penetration of microbes through the upper respiratory tract with dust particles of wool, manure, bedding and soil. In intensive foci, especially sheep and pig brucellosis, the main route of human infection is contact. In this case, microbes penetrate through the skin of people caring for sick animals or processing raw materials of animal origin [6,12].In enzootic foci of brucellosis, the seasonal increase in human morbidity is associated with the period of calving, lambing or farrowing in animals. Immunity in brucellosis is weak and short-lived, on average it lasts 6-9 months, and is not strictly specific [16].
2. Pathogenesis
- Brucella enters the body through the skin or mucous membranes, is captured by macrophages, multiplies in them and is carried by the lymph flow to the regional lymph nodes, and from the latter, through the lymphatic and blood vessels, spreads throughout the body [13].The pathogenesis of brucellosis includes five phases: lymphogenous, hematogenous, the phase of polyfocal localizations, the phase of exofocal seeding and the phase of metamorphosis. The degree of manifestation of individual phases and the sequence of their development depend on the individual characteristics of the organism, the entry gate of infection, the type and dose of the pathogen, and the conditions of infection [9,14].The entry of pathogens through the lymphatic pathways into the regional lymph nodes is the first phase of pathogenesis, the phase of lymphogenous drift and lymphoreceptor irritations corresponds to the incubation period of the disease. Further development of the pathological process is determined by the infecting dose and the state of the body's defenses. Brucella can persist in the lymph nodes for a long time, causing immunological restructuring of the body without any clinical manifestations (primary latency). With a significant accumulation of the pathogen due to incomplete phagocytosis observed in this infection, the lymph nodes become reservoirs of pathogens, from where brucella can enter the blood and spread throughout the body (the phase of hematogenous drift, or primary generalization). Clinically, this corresponds to the acute period of the disease, manifested by fever, chills, sweats, micropolyadenitis and other symptoms [17,22].Brucella are captured from the blood by cells of the mononuclear phagocyte system of various organs (liver, spleen, bone marrow, etc.) with the formation of metastatic foci of infection in them (the phase of polyfocal localizations). With the onset of generalization of the pathogen and the formation of metastatic infectious foci, an immunoallergic restructuring of the body occurs, determining the features of the pathogenesis and pathomorphological picture of the disease [10].Brucellosis is one of those infectious diseases in which the immune response does not provide bacteriological cleansing of the body in a significant proportion of patients. Brucella persist for a long time in metastatic foci, from which repeated multiple dissemination of the pathogen occurs with the development of reactive-allergic changes and chronicity of the process (phase of exofocal seeding and reactive-allergic changes). Superinfection, which often occurs in patients in endemic foci of the disease, is an additional factor contributing to the development of exacerbations, relapses and chronicity of the disease. Chronic course, accompanied by multiple generalization of the pathogen from metastatic foci, gives brucellosis the character of chronic sepsis. Long-term exposure of the pathogen and its waste products to organs and tissues causes immunoallergic restructuring of the body and a complex of manifestations characteristic of subacute and chronic brucellosis. In the clinical picture, along with the symptoms inherent to acute brucellosis, focal lesions of various localizations appear. During this period, the probability of brucellosis seeding from blood decreases, and positive allergic tests are detected (Burne test) [3,7,15,24].However, it should be noted that even in this phase of the disease, it is possible to isolate brucellae from the blood, most often in the form of L-forms [6,14].The next phase – the phase of residual metamorphosis – corresponds to the outcomes of brucellosis, ending either in complete resorption of inflammatory formations or in the formation of persistent irreversible cicatricial changes in the affected organs and tissues [10,16,20].
3. Clinical Manifestations
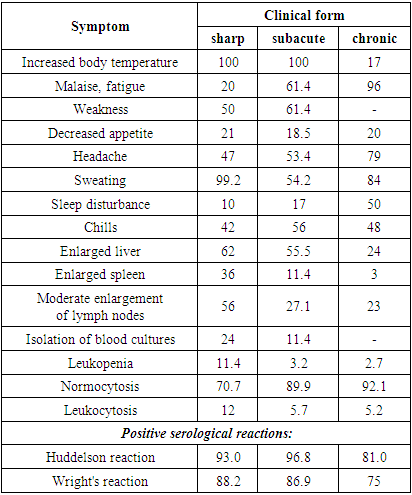
- The clinical manifestations of brucellosis are extremely diverse and its clinical classification is complex due to the polymorphism and lability of clinical symptoms, the duration of the course and the alternation of phases of exacerbation and remission.N. I. Ragoza proposed a simple, easy-to-use classification of clinical forms of brucellosis:Ø primary -latent;Ø acute septic;Ø septic -metastatic (primary and secondary chronic metastatic);Ø secondary latent.Clinical classification of brucellosis according to K.D. Dzhalilov:Ø AcuteØ SubacuteØ Chronic: (primary and secondary)Ø Latent: (primary and secondary)Ø ReinfectionØ Residual Clinical classifications of chronic brucellosis were also developed by G. P. Rudnev:Ø Visceral form: (cardiovascular, pulmonary and hepatosplenic)Ø Osteoarticular formØ Nervous formØ Urogenital formØ Combined formThe clinical course of brucellosis infection is characterized by polymorphism with damage to various organs and systems. An idea of how diverse the clinical manifestations of brucellosis infection are is given in Table 1, which also presents the results of laboratory studies [4,18].
|
4. Diagnostics
- Diagnostics of brucellosis of female genital organs presents significant difficulties due to the polymorphism of clinical manifestations and the presence of latent, atypical forms of the disease. A complete diagnostics of brucellosis is possible only with the use of a complex of research methods, i.e. with a carefully collected epidemiological anamnesis, a comprehensive clinical examination and the setting of specific immunological reactions. Preventive vaccinations in epidemic foci of brucellosis, the use of antibiotics, corticosteroids and other active methods of treatment often significantly change the clinical picture of the disease, the nature of the allergic restructuring of the body and thereby significantly complicate the recognition of brucellosis [15,17,25].During the examination of the patient, the epidemiological anamnesis is carefully collected. It is necessary to take into account the patient's stay in an endemic focus, contact with sick animals, etc. The patient's place of work and her specialty are of no small importance. Infection with brucellosis is possible during the processing and consumption of infected dairy and meat products, as well as during the processing of wool, astrakhan fur, and skin of sick animals. It is much more difficult to establish a diagnosis in people accidentally infected through the alimentary route when consuming infected dairy products [4,9].It is extremely important to correctly evaluate the gynecological anamnesis of patients. Menstrual dysfunction, long-term chronic inflammatory processes without a tendency to suppuration and damage to the peritoneum and pelvic tissue, in which functional disorders prevail over anatomical changes, may be suspicious of brucellosis with the corresponding epidemiological anamnesis. A wave-like long-term course of the disease, damage to the endometrium, cicatricial changes in the area of the uterine appendages, sacrouterine ligaments, primary or secondary infertility, repeated spontaneous abortions, premature births, stillbirths allow us to suspect brucellosis infection. Brucellosis is characterized by an exacerbation of the disease during pregnancy and after childbirth [1,3,9].Laboratory tests play an important role in determining the degree of allergic restructuring of the body in response to the introduction of brucellosis infection. However, the severity of the clinical course does not always correspond to the degree of immunological reactivity of the body, and negative laboratory test results are not always grounds for excluding the diagnosis of brucellosis. At the same time, positive reactions in vaccinated people or those in endemic foci of brucellosis do not always indicate the presence of the disease [26].In the diagnosis of brucellosis, clinical data, epidemiological history and laboratory test results are taken into account.An epidemiological history indicating the possibility of professional or domestic infection is of exceptional importance in the diagnosis of brucellosis.Each case of disease suspected of brucellosis must be confirmed by laboratory tests. Bacteriological, biological, serological and allergological research methods are usually used for this purpose [19].Brucella culture from patients is an undoubted confirmation of the diagnosis. The pathogen can be obtained (using special media) from blood, bone marrow, bile, urine, lymph nodes, cerebrospinal fluid, joint fluid (in arthritis), vaginal discharge, spleen puncture. Brucella grow slowly, over the course of a month. Bacteriological studies, due to their complexity and the need to observe precautions, are carried out in special laboratories [26].For the purposes of immunodiagnostics of brucellosis, a fairly specific and sensitive enzyme-linked immunosorbent assay (ELISA) method has been developed, which has not yet found wide application in our country due to its expense and the low prevalence of the disease.In the serological diagnostics of brucellosis, the Wright reaction remains of great importance, which is often positive already in the first days of the disease; a titer of agglutinins in the blood serum being tested of at least 1:200 is considered diagnostic [17].Heddleson plate agglutination reaction with whole serum and concentrated antigen is widely used. The CSC, RNGA and Coombs reaction are also of diagnostic value. They are distinguished by a higher sensitivity than other serological methods [17].Among the allergological methods for diagnosing brucellosis, the Burnet reaction is used by intradermal administration of brucellin, a filtrate of the broth culture of the pathogen. This reaction is based on the ability of an organism sensitized by a brucellosis antigen to respond by developing a specific process in the skin in the form of redness and swelling. The reaction is taken into account by the size of the swelling: if its diameter is less than 1 cm, the reaction is considered doubtful, from 1 to 3 cm - weakly positive, from 3 to 6 cm - positive, and more than 6 cm - sharply positive. As a rule, the intradermal test becomes positive by the end of the first month of the disease. It can also be positive in individuals vaccinated with a live brucellosis vaccine. This reaction is of particular importance in the diagnosis of chronic brucellosis [7,9,17,21,23].
5. Treatment Strategies
- The objectives of treating patients with brucellosis are to stop the disease process, eliminate its consequences and restore the patient's ability to work. Therapy for the disease depends on the phase, degree of compensation of the process and the nature of the immunoallergic restructuring of the body.Methods of treating brucellosis are varied: depending on the stage and degree of compensation of the process, antibiotic therapy, anti-inflammatory, desensitizing, stimulating, resolving and symptomatic therapy are used. Antibiotics are widely used in the treatment of active forms of brucellosis: acute, subacute, re- and superinfection.Antibiotics are used in the acute period. The most effective against brucellosis are the following groups of antibiotics: fluoroquinolones, tetracycline, and aminoglycosides. To increase the effectiveness, 2 antibiotics from different groups are used, one of which should act on the pathogen at the cellular level. Antibiotics have a pronounced antimicrobial effect, but do not prevent exacerbations, relapses and the formation of a chronic process [6].For the treatment of adults with acute brucellosis, it is recommended to use rifampicin at a dose of 600-900 mg and doxycycline 200 mg daily for at least 6 weeks.Combination therapy is mandatory to reduce the frequency of relapses - doxycycline orally 0.1 g every 12 hours + streptomycin intramuscularly 1 g / day during the first 15 days of therapy. WHO recommends the following regimen: doxycycline orally 0.1 g every 12 hours + rifampicin orally 0.6-0.9 g / day in 1-2 doses for 1.5 months. Alternative drugs: co- trimoxazole [sulfamethoxazole + trimethoprim (co- trimoxazole)] orally 0.96 g + rifampicin orally 10-20 mg / kg per day (but not more than 0.6 g / day ) or streptomycin intramuscularly 1.0 g / day. Duration of therapy is at least 1.5 months [3,7,10,18].In endocarditis, a combination of antibiotics must be used for 6 months. In the presence of complications such as meningoencephalitis or endocarditis, combined therapy with rifampicin, tetracycline, and aminoglycosides is recommended. Tetracycline and aminoglycosides may not reach the cerebrospinal level; doxycycline penetrates the hematoencephalic barrier better than generic tetracycline and is used successfully with trimethoprim + sulfamethoxazole and rifampicin in brucellosis meningitis. Third-generation cephalosporins also reach high concentrations in the cerebrospinal fluid, but the sensitivity of different strains of Brucella to them is not the same [5].Treatment of pregnant women: monotherapy rifampicin or a combination with co- trimoxazole [sulfamethoxazole + trimethoprim (co- trimoxazole)].When treating patients with brucellosis, a typical mistake is to prescribe antibiotics in short courses (7-10 days), prescribed repeatedly at intervals of 5-10 days, which leads to the transition of acute brucellosis to chronic forms in 30-70% of patients. When using the recommended antibiotics in one long course (at least 3 weeks), relapses of brucellosis are observed in 6-12% of patients [4,18].The therapeutic vaccine, which was widely used in the past, is currently used extremely rarely due to the high allergy rate of the population in general and the sensitizing properties of the vaccine [18,21,26].In severe acute brucellosis, as well as in chronic brucellosis, glucocorticosteroids are used, most often prednisolone at a rate of 1-1.5 mg / (kg × day); the course duration is 3-4 weeks. In the chronic form, sometimes courses of hormonal therapy are repeated 2-3 times with a break of 3-4 weeks.Symptomatic treatment is also widely used [ibuprofen (Nurofen), paracetamol, etc.].During persistent remission of the chronic form of the disease and the post-brucellosis period, the following methods of physiotherapy treatment are prescribed:Ø therapeutic exercise;Ø radon baths;Ø balneotherapy;Ø paraffin applications;Ø ultra-high frequency therapy;Ø ultraviolet radiation;Ø electrophoresisIn case of chronic brucellosis, spa treatment is indicated [4,8].The prognosis for life with brucellosis is quite favorable. Fatal cases in our country are very rare. The ability to work of patients may be impaired for a long time, and it is difficult to decide on the establishment of a disability group or employment after suffering brucellosis.
6. Prevention
- The main measures to prevent brucellosis are to eradicate the infection in farm animals, which is determined by strict adherence to veterinary and sanitary rules. To prevent brucellosis in humans, milk is disinfected by boiling and pasteurization. Products made from raw milk are aged for the required time before consumption. Measures should be taken to prevent the spread of the pathogen with meat and meat products. Animal skins and wool are subject to disinfection [18].Brucellin or have been vaccinated against brucellosis are allowed to work with infected animals and at enterprises processing livestock raw materials. Protective clothing and disinfectants must be used during work. Specific prevention of brucellosis is achieved by using a live anti-brucellosis vaccine, which provides immunity for 1-2 years [21].Vaccination is carried out in areas where there is a high incidence of brucellosis among animals. Immunoprophylaxis is subject to persons caring for farm animals and workers at enterprises processing livestock products.Despite the important role of immunoprophylaxis of brucellosis, sanitary-hygienic and veterinary measures are of decisive importance in preventing infection [4,8,9].
7. Conclusions
- Considering the serious disorders that occur in the female reproductive system under the influence of brucellosis infection, it is advisable to carry out a number of special measures in relation to a certain contingent of female workers in endemic foci. Thus, it is important to carry out a medical examination of women with brucellosis by a gynecologist regardless of whether they have complaints. When examining patients with chronic inflammatory processes, menstrual dysfunction, infertility, miscarriage, the epidemiological factor should be taken into account. All pregnant women in endemic foci should be examined for brucellosis, and reactions should be made not only with blood serum, but also with the secretion of the mammary glands. If latent forms of brucellosis are detected in pregnant women, it is recommended to conduct a course of preventive treatment in a hospital setting both during pregnancy and after childbirth.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML