-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(5): 1387-1393
doi:10.5923/j.ajmms.20251505.15
Received: Apr. 2, 2025; Accepted: Apr. 21, 2025; Published: May 9, 2025

Clinico-Epidemiology and Molecular Detection of Chikungunya Virus in Kassala, Eastern Sudan
Yasir B. Ahmed 1, Alzubair M. Ahmed 2, Doaa Mohammed Elhassan Burei Musa 3, 4, Abubaker A. Ramli 5, Mona M. S. Salama 6, Nadir M. Abuzeid 7, 8
1Department of Microbiology, Albandr Clinic Complex, Altaif City, Kingdom of Saudi Arabia
2Department of Medical Microbiology, Faculty of Medical Laboratory Sciences, University of Kassala, Kassala State, Sudan
3Department of Microbiology, Alnoor Medical Complex, Oman City, Sultanate of Oman
4Department of Medical Microbiology, Postgraduate College, Karrai University, Khartoum, Sudan
5Shamekh Medical Complex, Al Baha, Saudi Arabia
6Department of Haematology, Currently Achieved PHD from Al-Neelain University, Sudan
7Department of Medical Microbiology, Programme Faculty of Medical Laboratory Science, Delta College of Science and Technology, Khartoum, Sudan
8Department of Medical Microbiology, Faculty of Medical Laboratory Science, Omdurman Islamic University, Khartoum, Sudan
Correspondence to: Nadir M. Abuzeid , Department of Medical Microbiology, Programme Faculty of Medical Laboratory Science, Delta College of Science and Technology, Khartoum, Sudan.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

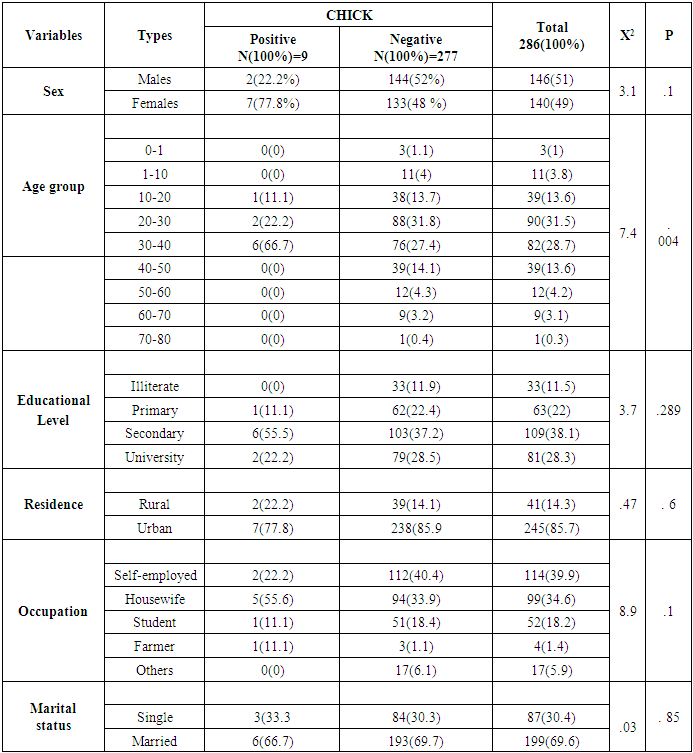
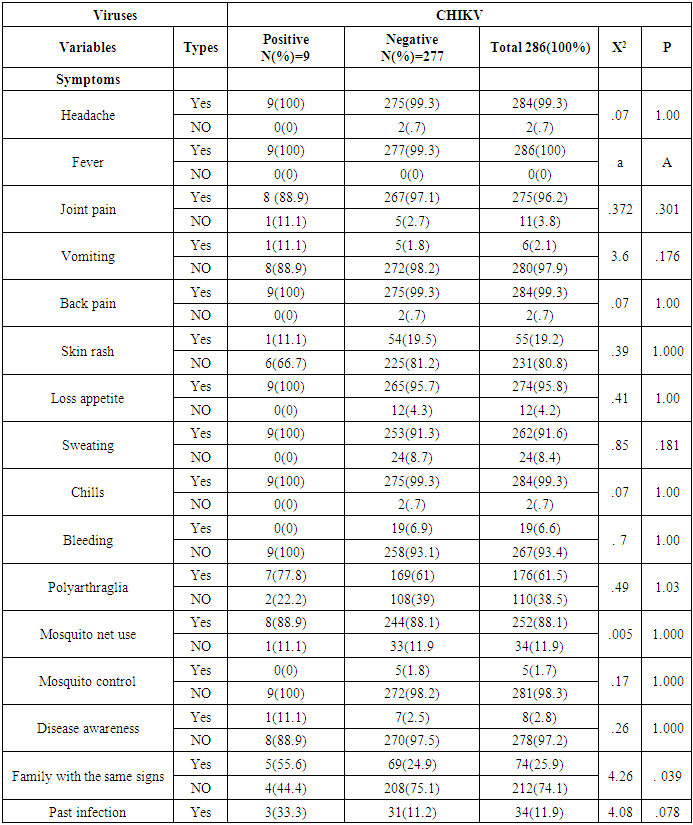
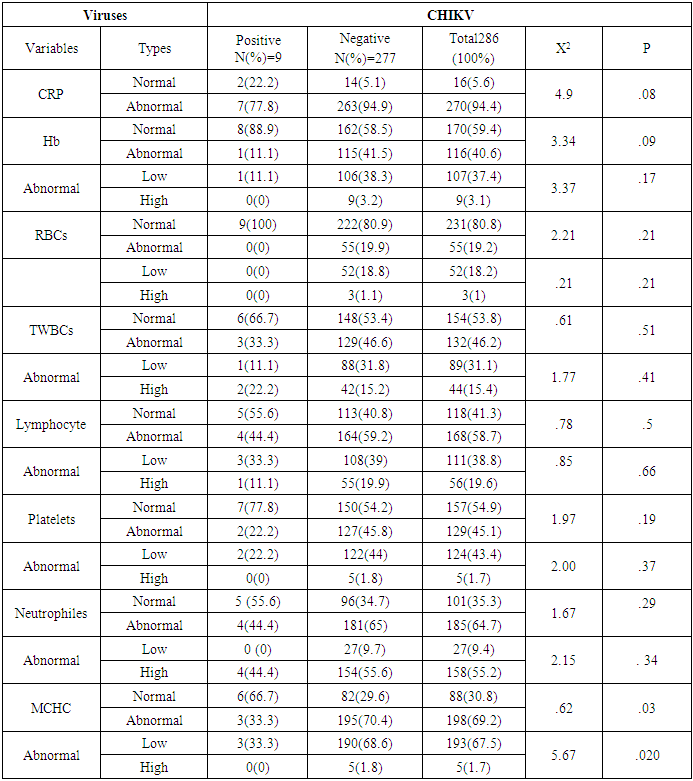
Background: Chikungunya is a mosquito-borne disease that affects humans in Africa, Asia, and America. It is caused by the Chikungunya virus (CHIKV), an RNA virus classified under the genus Alphavirus in the Togaviridae family. Early monitoring and diagnosis with specific molecular techniques will reduce the severity of viruses. Objective: This study aims to evaluate the prevalence and molecular characterization of CHIKV, and relation of age, sex, locality and other biomarkers to CHIKV in Kassala State, Eastern Sudan. Methodology: This cross-sectional study was conducted at Kassala Teaching Hospital in Kassala State, Eastern Sudan in which analyzed 286 samples for CHIKV using Real Time Polymerase chain reaction (RT-PCR), patients data were collected from questionnaire, Ethical approval was obtained from the Health Research Ethics Committee of the Ministry of Health and Social Development, Kassala State, and informed consent was obtained from participants or their guardians, statistical significance was determined using chi-square with a threshold set at P < 0.05. Results: Out of 286 participants, nine tested positive for Chikungunya virus (CHIKV), as confirmed by RT-PCR. Among the positive cases, 2 (22.2%) were male and 7 (77.8%) were female (χ² = 3.1, P = 0.1), there was no a statistically significant association between gender and infection All patients exhibited symptoms, including fever (9 cases, 100%), headache (9 cases, 100%), joint pain (8 cases, 88.9%), back pain (9 cases, 100%), loss of appetite (9 cases, 100%), sweating (9 cases, 100%), chills (9 cases, 100%), vomiting (6 cases, 2.1%), skin rash (1 case, 11.1%), and no cases of bleeding (0%). Additionally, seven patients (77.8%) experienced polyarthralgia. No statistically significant differences were noted between these symptoms and CHIKV infection. There was statistically significant differences family with the same signs (χ² = 4.26, P = 0.039). The haematological parameters revealed both normal and abnormal findings, with thrombocytopenia and lymphopenia being the most common abnormalities. However, no statistically significant differences were observed about CHIKV. Conversely, there was statistically significant differences in mean corpuscular haemoglobin concentration (MCHC) when compared to CHIKV infection (χ² = 5.67, P = 0.02). Conclusion: No statistically significant differences were found among the symptoms of CHIKV-positive patients. Notably, family with the same signs and abnormal MCHC levels were statistically significant (χ² = 4.26, P = 0.039) and (χ² = 5.67, P = 0.02) respectively concerning CHIKV infection.
Keywords: Chikungunya, Clinico, Epidemiology, Kassala, Sudan, RT-PCR, Prevention
Cite this paper: Yasir B. Ahmed , Alzubair M. Ahmed , Doaa Mohammed Elhassan Burei Musa , Abubaker A. Ramli , Mona M. S. Salama , Nadir M. Abuzeid , Clinico-Epidemiology and Molecular Detection of Chikungunya Virus in Kassala, Eastern Sudan, American Journal of Medicine and Medical Sciences, Vol. 15 No. 5, 2025, pp. 1387-1393. doi: 10.5923/j.ajmms.20251505.15.
Article Outline
1. Introduction
- Arthropod-borne viruses (Arboviruses) cause many infectious diseases worldwide. One of these is CHIKV, which is present in tropical and subtropical regions and other parts of the world. It causes infectious diseases and can be clinically diagnosed in endemic countries [1].CHIKV is an RNA virus single strand of the genus Alphavirus of the Togaviridae family. Transmitted by Aedes albopictus. It is enveloped by five structural proteins and four nonstructural proteins [2].The symptoms of CHIKV are muscle and joint pain, headaches, back pain, nausea, and rash in the acute phase; patients develop carpal or tarsal tunnel syndrome [3].The diagnosis of CHIKV involves detecting antibodies to the virus and viral RNA using enzyme-linked immunosorbent assays (ELISA) and RT-PCR, respectively [4].CHIKV is endemic in Sudan, with frequent outbreaks reported. Infections have been identified in seven of the 18 Sudanese states. The first documented case of the disease in Sudan dates back to 1973, revealing a seroprevalence of 13% for CHIKV in Central Sudan. Similar rates were found in the central and northern provinces, with seroprevalences of 10% and 12%, respectively. In contrast, studies in the western region reported significantly higher seroprevalence rates, reaching up to 44%. An epidemiological survey in East Sudan, utilizing quantitative reverse transcription polymerase chain reaction (qRT-PCR), indicated that 24% of pregnant women were infected with CHIKV, and these infections were linked to preterm deliveries. A broader study across the country found a relatively low seroprevalence of CHIKV in East and Central Sudan at 1.8%, while dengue virus seropositivity was at 100%. A significant outbreak of chikungunya is currently ongoing [5]. However, a few studies have been investigated in eastern Sudan by using real-time PCR. Therefore the aims of the study is to investigate the epidemiological and molecular detection of CHKIV, to identify areas with high risk for infection and to associate relative risk age, sex, locality and others biomarkers with infection.
2. Material and Methods
- Study designA cross-sectional study was conducted at Kassala Teaching Hospital, Eastern Sudan. Two hundred eighty-six participants were included in this study according to the formula estimated by proportion and confidence interval 95%.Study duration From October 2021 to December 2023.Inclusion criteriaAll febrile patients attending Kassala Teaching Hospital.Exclusion criteriaThe study excluded all patients who had malaria positive or those from outside the state.Sample size• Samples were calculated by equation• N= Z^2*P(1-P)/D^2• N= Sample size.• Z= level of confidence = 95% = 1.96.• P= Prevalence of disease = 0.23%.• D= The margin of error = 5% = 0.05.• N= (1.96)^2*0.23(1-0.23)/(0.05)^2 = 286 samples.Sample collection5 mL of blood was collected aseptically into EDTA vacutainer tubes. They were then centrifuged at 3000 rpm for 5 minutes. RNA was extracted and stored at -20°C.Data collection Patients were interviewed through a structured questionnaire. Information on age, place, occupation, gender, onset of disease, clinical symptoms, and date and place of sample collection will be recorded during the medical consultation.Data AnalysisData was analyzed by Statistical Package for Social Sciences (SPSS) program version 21 Inc., Chicago, IL, USA)for interpretation of the results and binary regression analyses; a logistic regression analysis test was employed to assess the association between variables. Statistical significance was set at P < 0.05. Frequencies and proportions were drawn for numerical data, and recurrent response was done for quantitative data mean and standard deviation.Ethical considerationThis study was approved by the Ethical in Research Committee of the Ministry of Health, Kassala State, and the University Committee board. Informed consent will be obtained from adult patients, parents, or legal guardians of children.RNA extractionCHIKV RNAs were extracted from plasma samples using a Patho Gene-spinTM DNA/RNA Extraction Kit (Intron Biotechnology, korea) is designed for rapid and sensitive isolation of DNA or RNA from a variety of pathogens, uses advanced silica-gel membrane technology for rapid and effective purification of DNA or RNA without organic extraction or ethanol precipitation. The procedure followed according to the manufacturer's.Real time PCRRT-PCR was carried out to evaluate the extracted RNA, use of the Aridia Zika/Dengue/CHIK Real-Time PCR Test (CTK Biotech, Inc., Poway, California, USA kit), is based on the real-time amplification of the CHIKV NSP1 gene in a one-step format. The RNA is transcribed into complementary DNA (cDNA) by reverse transcription, followed immediately by PCR within the same well.Principle of RT-PCRThe Aridia Zika/Dengue/CHIK Real-Time PCR test comes ready-to-use. All real-time PCR components, including DNA polymerase, reverse transcriptase, primers, probes, and dNTPs, are stabilized within each reaction well. In addition, an Internal Control is included within each well for monitoring PCR reaction inhibition, the assay is based on 5’ nuclease chemistry which utilizes CHIKV-specific primers and hydrolyzable fluorogenic probes to detect the accumulation of amplified target sequences during the PCR reaction. Upon extension of the primers by DNA polymerase, the fluorogenic probes are hydrolyzed by the 5’ to 3’ exonuclease activity of the polymerase, causing spatial separation of the reporter and quencher. The resulting increase in CHIKV-specific fluorescence signal is measured by the real-time PCR thermocycler and is proportional to the amount of amplified product. PCR protocolThe RT-PCR thermocycler protocol was programmed as follows: an initial reverse transcription step was conducted at 50°C for 15 minutes, followed by an initial denaturation at 95°C for 2 minutes. Subsequently, 45 amplification cycles were performed, each consisting of denaturation at 95°C for 10 seconds and annealing/extension at 60°C for 50 seconds. The PCR thermocycler used was a Bio-Rad machine, model NO. CFX96 Optics Module, with serial NO. 785BR32294, Singapore.Positive sample: A sample is assigned as positive for the target if the Ct value is less than 40 and the Internal Control also has an amplification signal.Negative sample: A sample is assigned as negative for the target if there is no evidence of an amplification signal in the detection system, but the Internal Control is amplified.Positive and negative control must show an amplification signal or no respectively.Data AnalysisData were analyzed using version 28 of the Statistical Package for Social Sciences (SPSS). The results were interpreted through cross-tabulation analyses, employing tests to evaluate the relationships between variables. Statistical significance was determined using chi-square with a threshold set at P < 0.05. Numerical data were expressed in terms of frequencies and proportions, while quantitative data were assessed through recurrent responses, calculating both the mean and standard deviation.
3. Results
- A total of 286 participants from Kassala State, Eastern Sudan, were involved in this study. The mean age of the participants, with standard error, was 30.3 ± 0.789 years, with ages ranging from 1 to 73 years. Among the participants, 146 (51%) were male, and 140 (49%) were female. The prevalence of CHIKV was found to be 3.15% (Figure 1).
 | Figure 1. Frequency and percentage of positive and negative CHICKV |
|
|
|
4. Discussion
- Arthropod-borne viruses (arboviruses) are responsible for a multitude of infectious diseases worldwide, including the Chikungunya virus (CHIKV). This virus is prevalent in tropical and subtropical regions, among other areas, and causes various infectious diseases that can be clinically diagnosed in endemic regions [1].CHIKV is an RNA virus that belongs to the genus Alphavirus within the Togaviridae family, primarily transmitted by Aedes albopictus mosquitoes [2].A study by Mota et al. (2021) examined 182 samples and observed that 7 cases (3.8%) were positive for CHIKV analyzed by RT-Qpcr. The frequency of positive CHIKV compared with participants in the study by Mota et al. (2021) was similar to ours [6].A study in Sudan by Aljaily et al. (2024) studied of 773 suspected cases, 63 (8.15%) were confirmed positive for Arbovirus, 49 (77.77%) of the 63 positive were CHIKV positive [7], a study by Marinho et al. (2020) tested of 102 samples for Arbovirus, identified 52 cases with CHIKV [8], Silva et al. (Silva et al., 2019) studied of 948 participants, 247 (26.1%) positive for viruses, of which CHIKV was 159 (16.7%), in the three studies mentioned, the frequency of CHIKV was high unlike our study the frequency was low may be due to the outbreak of CHIKV or rainy season during these studies [9]. In the present study, the typical age group positive for CHICKV was 30-40 years, and CHICKV cases showed a statistically significant association difference between age and infection in the study by Edwards et al. (2015) [10]. Aljaily et al. (2024) observed young participants aged 1-19 years more affected, explained that by young participants stay at home and more exposed to mosquito bite at day [7].In line with our study, Aljaily et al. (2024) noted a higher incidence of CHIKV infection among females with 41 cases (65.08%) compared to 22 cases (34.92%) in males [7]. Similarly, Farias et al. (2023) reported that the majority of CHIKV cases occurred in females and within the age group of 20 years and older. The common frequent females attributed to females spend more time at home than males and exposed to mosquito bite [11].Moreover, factors such as residence, educational level, occupation, and marital status did not exhibit statistically significant differences in relation to CHIKV infections. A meta-analysis conducted by Power et al.(2022) indicated a lack of strong statistical evidence linking education to Arboviral infections, higher risk of infection tended to be among less educated participants and it was also found that marital status whether currently married or previously (e.g., divorced or widowed) was associated with an increased risk of infection. Also unobserved effect of employment due to occupation may be in side or out side [12].The symptoms reported in patients included fever (100%), headache (100%), joint pain (88.9%), back pain (100%), loss of appetite (100%), sweating (100%), chills (100%), vomiting (11.1%), skin rash (11.1%), and bleeding (0%), with polyarthralgia affecting 77.8% of the participants. No statistically significant differences were observed between symptoms and viral infection. Aljaily et al. (2024) highlighted that all cases (100%) presented with fever, with the majority also showing symptoms of headache (86%), joint pain (79%), muscle pain (78%), and back pain (75%) [7]. In contrast, Mota et al. (2021) observed symptoms such as fever (82.85%), exanthema (77.14%), headaches (77.14%), myalgia (71.42%), arthralgia (54.28%), and pruritus (54.28%) among CHIKV-positive patients, differing from the current study's findings [6]. Taraphdar et al. (2012) identified common features in CHIKV-positive cases, including fever, joint pain, rashes, headache, myalgia, and nausea/vomiting, with severe arthralgia and joint swelling being particularly [13]. Marinho et al. (2020) observed The symptoms of CHIKV monoinfected patients were fever (89.3%), headache (78.7%), arthralgia (91.4%) and generalized body pain (93.6%) were more frequent and Exanthema (38%). Our study and other studies from Sudan similar in symptoms and differ slightly from studies from other countries, may attributed to regional or genetic difference [8].The hematological parameters observed in this study revealed both normal and abnormal findings, with the most notable abnormalities being thrombocytopenia and lymphopenia, which align with the observations made by Kularatne et al. (2005) [14].Sample bias due to represent certain participants not all population, lack of prior research studies on the biomarkers and haematological parameters against CHIKV.Vector control measures, especially mosquito control, are essential, habitat control involves draining swamps and removing other pools of stagnant water (such as old tires, large outdoor potted plants, empty cans, etc.) that often serve as breeding grounds for mosquitoes. Use of insecticides, personal protective measures like sleeping under mosquito nets, wearing protective clothing, applying insect repellents.
5. Conclusions
- The prevalence of CHIKV was found to be 3.15%. The number of infected females exceeded that of males (X² = 3.1, P = 0.1), although this difference was not statistically significant.The age group most commonly affected by CHIKV was between 30 and 40 years, with a statistically significant difference observed in age distribution among the cases.Patients reported symptoms including fever, headache, back pain, loss of appetite, sweating, and chills.Analysis of residency, educational level, occupation, and marital status revealed no statistically significant differences in relation to CHIKV infection.Haematological parameters varied, showing both normal and abnormal results; the most notable abnormalities were thrombocytopenia and lymphopenia. However, no statistically significant relationships were found between these abnormalities and the virus.
6. Recommendations
- Further studies with a larger sample size are warranted. A meta-analysis should be conducted to compile data from all previous CHIKV studies in Sudan, encompassing screenings from both eastern and western regions in Sudan.Vector control measures, especially mosquito control, are essential, habitat control involves draining swamps and removing other pools of stagnant water, take personal protective measures seriously.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML