-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(4): 1108-1111
doi:10.5923/j.ajmms.20251504.52
Received: Jan. 28, 2025; Accepted: Feb. 21, 2025; Published: Apr. 16, 2025

Morphofunctional Changes in the Spleen of Offspring Born to Mothers with Thyrotoxicosis
Farrukh Khamroev1, Bobir Khoshimov2
1Assistant, Termez Branch of Tashkent Medical Academy, Termez, Uzbekistan
2Associate Professor, Alfraganus University, Tashkent, Uzbekistan
Correspondence to: Farrukh Khamroev, Assistant, Termez Branch of Tashkent Medical Academy, Termez, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This article explores the morphological and functional changes observed in the spleens of offspring born to mother rats experiencing experimental thyrotoxicosis. Thyrotoxicosis, characterized by increased thyroid hormone levels, accelerates metabolic processes in the body. The study, based on experimental research, compares the spleens of healthy offspring with those born to mothers induced with thyrotoxicosis. Morphological examinations revealed increased spleen size and activated development of lymphoid follicles. Histological analyses identified alterations in the red and white pulp zones and variations in lymphocyte counts. Functional evaluations demonstrated enhanced immune and hematopoietic activities of the spleen, with increased lymphocyte activation. The findings confirm that pathological processes in the maternal organism significantly affect fetal development and the immune system. This study underscores the importance of monitoring and treating thyroid gland pathologies during pregnancy. Under the influence of thyrotoxicosis, the spleen also undergoes morphological changes. An increase in the size of the spleen is observed. Changes within the cells, including an increase in lymphocyte activity, are noted. These changes can affect the process of blood production and the spleen’s immune function. Based on preliminary data, in the first generation, an increase in immune response reactions is observed. In the second generation, however, these indicators differ slightly, and in children, morphological changes in the spleen are observed under the influence of maternal hormones.
Keywords: Morphofunctional changes, Spleen, Offspring, Thyrotoxicosis, Thyroid gland, Mother rats, Lymphocyte activity, Body, Hyperthyroidism, Immune system, White pulp, Red pulp, Maternal hormones, Experimental thyrotoxicosis, Functional changes, Influence
Cite this paper: Farrukh Khamroev, Bobir Khoshimov, Morphofunctional Changes in the Spleen of Offspring Born to Mothers with Thyrotoxicosis, American Journal of Medicine and Medical Sciences, Vol. 15 No. 4, 2025, pp. 1108-1111. doi: 10.5923/j.ajmms.20251504.52.
1. Introduction
- According to the World Health Organization (2023), 10–30% of the global population suffers from thyroid dysfunction and structural abnormalities. The prevalence of thyroid disorders increases by 5% annually [WHO, 2023]. These disorders range from minor structural changes to significant functional and morphological conditions that reduce quality of life and, in some cases, life expectancy [1]. Iodine deficiency elimination has reduced non-toxic goiter and congenital hypothyroidism. However, in populations previously suffering from iodine deficiency, a slight increase in iodine consumption has been observed, leading to a decrease in the prevalence of thyroid gland pathologies such as diffuse and nodular goiter, congenital hypothyroidism, and other iodine deficiency–related diseases. At the same time, the autoimmune pathology of the thyroid gland is increasing. On the other hand, despite the increase in iodine levels, several changes in the structure and function of the thyroid gland continue to occur. This reflects not only the current iodine status but also the previous iodine deficiency (“iodine memory” effect) [2,3,4]. Thyrotoxicosis remains one of the most serious issues in endocrinology, leading to poor prognosis and an increased mortality rate from cardiovascular diseases. As a result, mortality from ischemic heart diseases, heart failure, arrhythmia, valvular defects, and arterial hypertension, in conjunction with thyrotoxicosis, has risen by 1.2 times compared to the general population. The underlying cause of this is the development of changes in the cardiovascular system [5,6,7,8].
2. Materials and Methods
- Our study investigating splenic morphology in experimental thyrotoxicosis utilized the following steps:1. Administration of thyroid hormones in laboratory animals (rats) and modeling experimental thyrotoxicosis in animals. That is, 0.3 mg of L–thyroxine was administered for 21 days, based on a dose of 0.3 mg per 1 kg body weight (Model by Petrova, 2021).2. Organ Collection and Fixation: Rats were euthanized with chloroform, and the organs collected for subsequent studies were fixed in 10% isotonic buffered formalin fixative.3. Histological Sample Preparation: Spleen tissues were dehydrated, embedded in paraffin, sectioned using a microtome, and stained with hematoxylin–eosin.4. Morphometric Analysis: Organ size, cellular areas, lymphoid follicle size, and specific cell types were measured.5. Comparative Analysis: Data from thyrotoxicosis-induced animals were compared with those of the control group.The morphological structures of the spleen were highlighted by staining with Hematoxylin and Eosin. The stained preparations were examined under a trinocular microscope (Nexcop–NE700 model) with LED lighting, and microphotographs were taken. The obtained results were analyzed at ocular magnification of 10x with objective lenses of x4, x10, x20, and x40.
3. Results and Discussions
- In the case of thyrotoxicosis, the increased levels of thyroid hormones enhance the processes occurring in maternal rat, which leads to increased activity and restlessness, indicating that metabolic disturbances are caused. As a result, changes observed in the offspring during the early stages, such as in the suckling period, also manifest in the rat pups. Specifically, during the first 21 days, the pups showed weight loss, hyperactivity, and increased aggression.
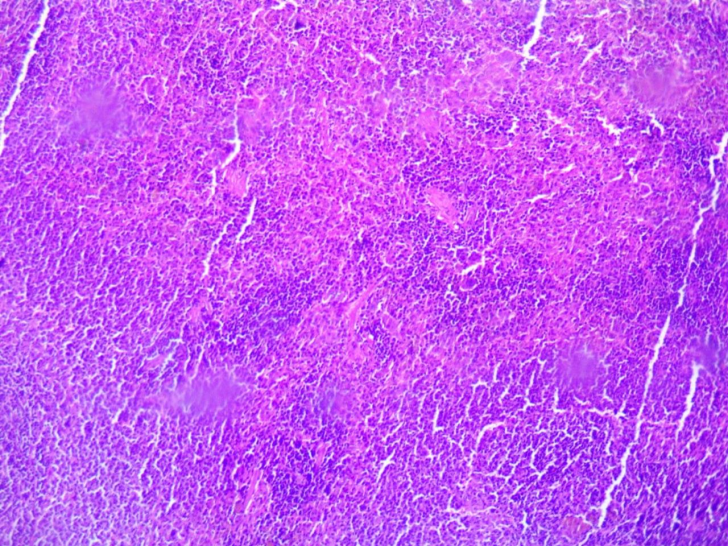 | Figure 1. Spleen of the offspring born from thyrotoxic mothers during the first 3 days of life. Staining was done with Hematoxylin–Eosin. Magnification: ocular 10x, objective 10x |
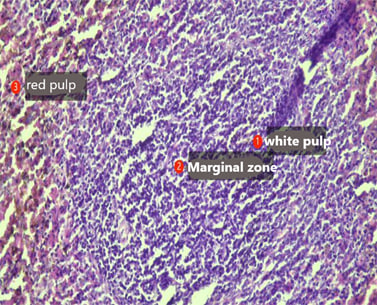 | Figure 2. Spleen of the offspring born from thyrotoxic mothers during the first 3 days of life. Staining was performed with Hematoxylin–Eosin. Magnification: ocular 10x, objective 10x |
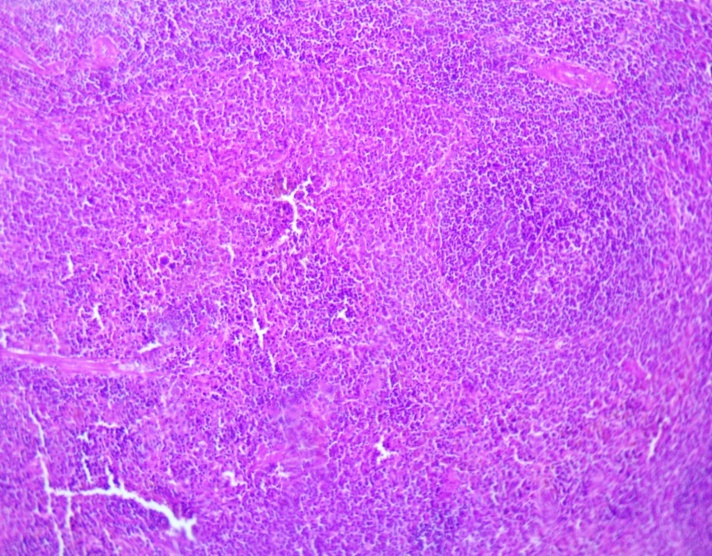 | Figure 3. Spleen of the offspring born from thyrotoxic mothers at the age of 21 days. Staining was performed with Hematoxylin–Eosin. Magnification: ocular 10x, objective 10x |
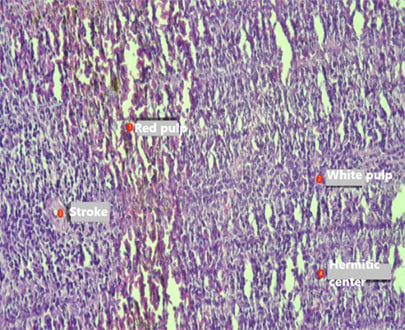 | Figure 4. Spleen of the offspring born from thyrotoxic mothers at the age of 21 days. Staining was performed with Hematoxylin–Eosin. Magnification: ocular 10x, objective 20x |
4. Conclusions
- The damage to cell membranes resulting from oxidative stress leads to structural disruptions in the spleen tissue, which in turn causes a decline in organ function. Another important aspect of the study is that the thyrotoxic condition may have a long–term effect on the immune function of the spleen. These findings further emphasize the importance of maintaining hormonal balance in the maternal organism during pregnancy. Disruptions in the immune system could increase susceptibility to autoimmune diseases and infections in the future [11]. Furthermore, changes in the spleen structure in offspring may also affect the function of other lymphoid organs in the body, such as lymph nodes and bone marrow. Compared to other studies, the uniqueness of this work lies in its focus on the impact of the thyrotoxic condition in maternal rats on their offspring. In the study by Ivanova and colleagues, an increase in oxidative stress in infants was observed due to the elevated thyroid hormone levels in the maternal organism. Our research further confirms these results and sheds light on this mechanism through changes in the immune function of the spleen. However, there are some limitations to the study. First, the level and duration of thyrotoxicosis in the maternal rats and its impact on the immune system of their offspring have not been fully explored. Second, additional research is required to study the effects on other lymphoid organs in the offspring. Third, data on the positive impact of hormonal treatment and other therapeutic approaches on the immune system of the offspring are insufficient. Future research in this direction, including genomic and proteomic analyses, will provide a broader understanding of the pathophysiological mechanisms of thyrotoxicosis and help develop effective strategies to prevent its consequences. The importance of balancing thyroid hormones during pregnancy remains high in clinical practice, as it is crucial not only for maternal health but also for the healthy development of future offspring.This study determines the deep morphofunctional changes in the spleens of pups from thyrotoxic mothers. The findings confirm that maternal thyroid hormone increase during pregnancy leads to remarkable splenic morphological changes, including splenomegaly, hyperactivity of lymphoid follicles, and disruption of the red and white pulp regions. Functional experiments proved an overactive immune response in the first generation, though the long-term effect on subsequent generations is not known.Furthermore, the study indicates the relevance of oxidative stress as a causative process involved in affecting offspring's immune system, evidenced by increased malondialdehyde concentrations and reduced glutathione peroxidase activity. These observations are in accordance with previous work on the outcomes of thyroid disorder on fetal growth, reemphasizing the significance of the maintenance of hormonal equilibrium during gestation to prevent immune dysregulation and potential vulnerability to autoimmune disease in maturity.While this research highlights the effect of maternal thyrotoxicosis on the immune system, there are certain limitations that must be taken into account. Future research shall have to focus on studying the long-term consequences of these changes across generations and testing for potential therapeutic interventions to counteract the deleterious effects of maternal thyroid abnormality. Along with that, a research on its impact on the other lymphoid organs such as the lymph nodes and bone marrow will enable us to learn more about the systemic function of maternal thyrotoxicosis in immunology.Lastly, the role of thyroid function in pregnancy cannot be overstated. The findings of the present study contribute to the growing literature emphasizing the need for early diagnosis and treatment of thyroid disease for the attainment of maximal maternal and fetal health.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML