-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(4): 1099-1103
doi:10.5923/j.ajmms.20251504.50
Received: Feb. 20, 2025; Accepted: Mar. 19, 2025; Published: Apr. 16, 2025

Assessment of the Association of Val16Ala Polymorphism of the SOD2 Gene with Stroke Development in a Sample of Patients with Cerebrovascular Diseases
Inoyatova S. O.1, Adambaev Z. I.2, Madzhidova Ya. N.3, Boboev Q. T.4, Abdukodirov E. I.1
1Tashkent State Dental Institute, Uzbekistan
2Urgench Branch of the Tashkent Medical Academy, Uzbekistan
3Tashkent Pediatric Medical Institute, Uzbekistan
4Republican Specialized Scientific and Practical Medical Center of Hematology of the Ministry of Health of the Republic of Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

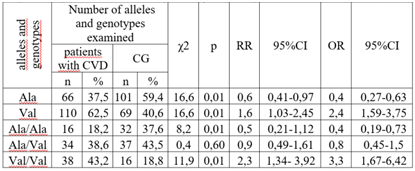
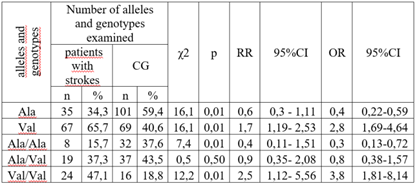
Relevance. Several studies have shown a significant association between the Val16Ala polymorphism of the SOD2 gene with the development of atherosclerosis and type 2 diabetes mellitus; however, this association has not been studied in the development of stroke. Material and Methods. We evaluated the association between the Val16Ala polymorphism of the SOD2 gene (rs4880) and stroke development in a sample of 176 patients with cerebrovascular disease. Genotyping of the Val16Ala of the SOD2 gene was performed by real-time polymerase chain reaction. Results. A significant difference was found when comparing the genotype and allele frequencies of the Val16Ala polymorphism of the SOD2 gene Val16Ala among patients with CVD and in the control group. The number of ancestral genotype Ala/Ala among the patients was statistically significantly less frequent compared to the CG representatives, which indicates the protective effect of this genotype in relation to the formation of CVD. Also, a significant increase in the proportion of carriers of the unfavourable Val/Val genotype was found among patients with CVD compared to the CG (43.2% vs. 18.8%, respectively). According to the odds ratio, the risk of CVD development in the presence of this genotype is significantly increased by 3.3 times (χ2=11.9; p=0.01; OR=3.3; 95% CI 1.67 - 6.42). Conclusions. A significant association was found between carriage of the SOD2 Val16Ala polymorphism and stroke development.
Keywords: Val16Ala polymorphism of SOD2 gene, Cerebrovascular diseases, Pre-stroke cerebrovascular diseases, Strokes
Cite this paper: Inoyatova S. O., Adambaev Z. I., Madzhidova Ya. N., Boboev Q. T., Abdukodirov E. I., Assessment of the Association of Val16Ala Polymorphism of the SOD2 Gene with Stroke Development in a Sample of Patients with Cerebrovascular Diseases, American Journal of Medicine and Medical Sciences, Vol. 15 No. 4, 2025, pp. 1099-1103. doi: 10.5923/j.ajmms.20251504.50.
Article Outline
1. Introduction
- Each year, 15 million people worldwide suffer a stroke, and the number of deaths from this pathology is increasing annually [11]. Stroke is the 2nd leading cause of death worldwide and is the leading cause of dementia and age-related cognitive decline. Most patients have impaired ability to work, making stroke the leading cause of disability in the adult population [3,5]. Stroke occurs as a result of sudden occlusion or rupture of a blood vessel in the brain, and ischaemic (IS) or haemorrhagic (HI) stroke is distinguished accordingly. IS is observed in approximately 85% of cases.Although well-known risk factors for stroke are arterial hypertension, atrial fibrillation, diabetes mellitus and smoking [5,7], most cases of stroke are due to unknown causes. Genetic predisposition is probably one such cause [1-4]. Identification of genetic variants predisposing to stroke is a key step towards improving diagnosis and developing innovative therapies to help reduce the burden of the disease [14,15].Numerous studies have shown that altered gene expression plays an important pathophysiological role in the development and progression of cardiovascular disease (CVD). Superoxide dismutase 2 (SOD2), also known as manganese superoxide dismutase (MnSOD), is one of the major antioxidant defence systems against mitochondrial superoxide radicals [6]. The rs4880 A>G mutation is located in the SOD2 gene and is a single nucleotide substitution of ‘A’ for ‘G’ at position 159,692,840 in chromosome 6. Currently, the clinical significance of this variation is considered as the effect has not been studied. The probable association of this mutation with the diseases ‘Superoxide dismutase polymorphism 2’, ‘Microvascular complications of diabetes, susceptibility to, 6’, ‘Non-alcoholic fatty liver disease’, ‘Mesothelioma’ and ‘Prostate cancer’ is being investigated. The worldwide incidence is 47 per 100 people (according to the TOPMED project).’ rs4880 A>G: [Electronic resource] // GENOCART Genetic Encyclopaedia. 2021. - URL: https://www.genokarta.ru/snps/rs4880_AG]. The Val16Ala polymorphism (rs4880) in the SOD2 gene has been predicted to cause a sequence change of the alpha helix target sequence in the Ala variant (encoded by the C allele) to a beta-sheet in the Val variant (encoded by the T allele), which causes a 30-40% decrease in SOD2 activity due to less efficient protein transport into the mitochondrial matrix [8,9]. Several studies in patients with type 2 diabetes mellitus (T2DM) [13,19], have shown a significant association between the Val16Ala polymorphism of the SOD2 gene and albuminuria, as well as an imbalance between the pro- and antioxidant systems of the body leads to the development of oxidative stress, which in combination with the presence of hyperlipidaemia creates favourable conditions for the development of atherosclerosis with the formation of genetically determined predisposition to its various clinical forms (angina pectoris, acute myocardial infarction, cerebral stroke). The aim of the study was to evaluate the association of the Val16Ala polymorphism of the SOD2 gene with stroke development in a sample of patients with cerebrovascular disease.
2. Material and Research Methodology
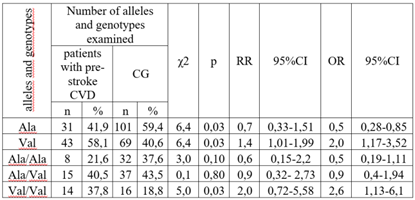
- We examined 176 patients with CVD, aged 30 to 75 years, unrelated and comparable in terms of socioeconomic and ethnic status. Patients were included in the study after obtaining informed consent for the study. The study protocol was approved by the local ethical committee (Institute of Haematology, TashPMI) of the Ministry of Health of the Republic of Uzbekistan. Depending on the degree of severity of clinical and morphological manifestations of CVD, patients were divided into 3 groups: 1) 102 patients with confirmed diagnosis of stroke; 2) 74 patients with pre-stroke CVD; 3) the control group was represented by a population sample of 101 Uzbek residents without hereditary aggravation, risk factors and clinical manifestations of CVD. The examination of the control group included: BP measurement, anthropometry (height, weight), socio-demographic characteristics, interview about smoking, alcohol consumption (frequency and typical dose), level of physical activity, assessment of lipid profile (total cholesterol (TC), triglycerides (TG), low-density lipoproteins (LDL) and high-density lipoproteins (HDL)). All patients were analyzed for SOD2 gene expression in peripheral blood. Exclusion criteria were: severe comorbidities, autoimmune diseases, diagnosed tumors, psychiatric diseases, refusal of genetic testing. A specially designed clinical record was filled out for each patient.DNA was isolated from peripheral blood using the Ampli Prime RIBO-prep kit (AmpliSens, Russia) according to the manufacturer's recommendations. Genomic DNA concentration was measured using a NanoDrop 2000 instrument (NanoDropTechnologies, USA) at A260/280 nm wavelength. Genotyping of Val16Ala polymorphism of SOD2 gene (rs4880)The Val16Ala polymorphism of the SOD2 gene (NPO Syntol, Moscow) was genotyped by RT-PCR using a RotorGeneQ thermocycler (QUAGEN Germany).Statistical analysisAssociation analysis of the Val16Ala polymorphism of the SOD2 gene was performed using a case-control model (case-control, two-sample comparisons).Deviations from Hardy-Weinberg proportions were assessed using GenePop (Genetics of Population), available on the Internet (http://wbiomed.curtin.edu.au/genepop).The application software package ‘OpenEpi 2009, Version 2.3’ was used as a tool for processing the obtained results.
3. Research Results
- Table 1 shows the allele and genotype frequencies of the Val16Ala polymorphism of the SOD2 gene in the compared groups. There were no significant deviations from the Hardy-Weinberg proportions in the CG and in the group of CVD patients. The frequencies of the Ala allele and Val allele were: 0.375 and 0.625 in the group of CVD patients and 0.594 and 0.406 in the CG.
|
|
|
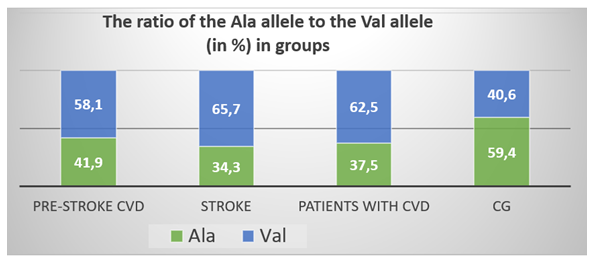 | Figure 1. Ratio of the Ala allele and the Val allele (in %) in groups |
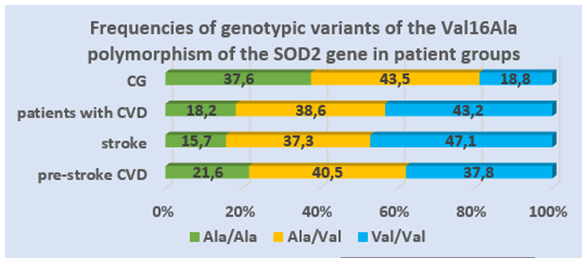 | Figure 2. Frequencies of genotypic variants of the Ala16Val polymorphism of the SOD2 gene in patient groups |
4. Discussion
- Cerebrovascular disease with the development of stroke is an important problem worldwide. Stroke is a common and fatal neurovascular disease with high morbidity and mortality rates, accounting for approximately 15 million new cases annually [11].Oxidative stress plays an important role in the pathogenesis of a significant number of diseases. The antioxidant enzyme, superoxide dismutase, is an important component of cellular defence against oxidative stress. In the cell, superoxide dismutase 2 is localised in the mitochondrial matrix. The SOD2 gene encoding this enzyme is located on chromosome 6q25.3 and consists of 5 exons [12].Superoxide dismutases (SOD) are important antioxidant enzymes that protect against superoxide toxicity. A variety of SOD enzymes have been characterized that use the cofactor copper, manganese, iron, or nickel to effect superoxide disproportionation. Copper and manganese SODs are highly divergent in sequence as well as in the process of metal insertion. Intracellular copper SODs of eukaryotes (SOD1) can acquire copper posttranslationally by interacting with the copper chaperone CCS. Disulphide oxidation can lead to gross misfolding of immature forms of SOD1, especially with SOD1 mutants associated with amyotrophic lateral sclerosis. In the case of eukaryotic mitochondrial MnSOD (SOD2), metal insertion cannot occur posttranslationally, but requires new synthesis and mitochondrial import of the SOD2 polypeptide. SOD2 can also bind iron in vivo but is inactive with iron. Such misincorporation of metal ions into SOD2 may become common when mitochondrial metal homeostasis is impaired. Accurate and regulated metallation of copper and manganese molecules by SOD is vital for cell survival in oxygenated environments [16,20].The SOD2 gene is a member of the iron/manganese superoxide dismutase family. It encodes a mitochondrial protein that forms a homotetramer and binds one manganese ion per subunit. This protein binds to superoxide by-products of oxidative phosphorylation and converts them to hydrogen peroxide and divalent oxygen. Mutations in the SOD2 gene are thought to play a causal role in the pathogenesis of idiopathic cardiomyopathy, premature aging, sporadic motor neuron disease, and cancer. It has been reported that SOD2 protein can be acetylated on a lysine residue, promoting stem cell reprogramming [16,20].Russian researchers analysed the distribution of genotypes and alleles of the polymorphic variant Ala16Val (rs 4880) of the SOD2 gene in a sample of men with coronary heart disease (CHD). We found a statistically significantly higher frequency of the T allele in the group of male CHD patients, 54.84%, compared to the male population sample, 44.14% (χ2=5.998; p=0.014; OR=1.54). The frequency of TT genotype in male CHD patients was 27.10%, which was also significantly higher than in the control group, 17.97% (χ2=7.265; p=0.026; OR=1.70). Thus, the authors revealed that the T allele and TT genotype are associated with CHD [4].Another study genotyped the Val16Ala polymorphism of the SOD2 gene in 994 patients with DM2: 119 individuals with macroalbuminuria and 875 patients with normoalbuminuria. In a logistic regression model including other relevant covariates, CT and CC genotypes had lower odds ratios than the reference TT genotype (CT: OR 0.67, p= 0.308; CC: OR=0.42, p= 0.034). Given the different odd ratios observed for CT and CC genotypes, the codominant model (CC, CT, TT) appears to best fit the data. Using the dominant model instead of the unrestricted model, the CC + CT genotypes had OR=0.55 (p= 0.026) relative to the TT genotype [10].In a subsequent study, the authors found that the expression of REL and superoxide dismutase 2 (SOD2) was significantly increased in the ischemic stroke group of a clinical cohort (P < 0.05). In addition, REL expression was increased in endothelial cells and fibroblasts at the single-cell level, indicating that REL is a cell-specific regulon [21].It has been established that the activity of superoxide dismutase 2 is affected by the Ala16Val polymorphism (401T>C) of the SOD 2 gene. The Ala16Val polymorphism, which results in the replacement of alanine with valine, is localized in the region responsible for binding to the mitochondria for transporting the enzyme into the mitochondrial matrix. In the mitochondrial matrix, this region is cleaved and superoxide dismutase passes into the active form [17]. It has been noted that the enzyme containing Valine (Val) in this position is transformed into the mitochondrial matrix more slowly than the protein containing alanine (Ala). Accordingly, carriers of the Val allele and the Val/Val genotype accumulate superoxide in the matrix, which leads to greater severity of oxidized damage to mtDNA [18].
5. Conclusions
- A significant association was found between the Val16Ala polymorphism of the SOD2 gene and the development of stroke. According to the odds ratio, the risk of developing CVD in the presence of the Val/Val genotype increases by 3.3 times (χ2=11.9; p=0.01; OR=3.3; 95% CI 1.67 - 6.42). The Ala16Val polymorphism in the SOD2 gene is one of the causes of CVD development with sensitivity, specificity and prognostic efficiency of the Val locus (SE=0.59, SP=0.63, AUC=0.61, OR=2.44, p=0.39).
ACKNOWLEDGEMENTS
- The authors would like to thank the staff of the neurology department of the Tashkent State Dental Institute for their help in selecting patients for diagnosis and treatment; Professor Boboev K.T. for assistance in laboratory and genetic studies; Professor Adambaev Z.I. of the Urgench branch of the Tashkent Medical Academy for writing the article; Professor Madzhidova Y.N. of the Tashkent Paediatric Medical Institute for editing the article. The authors declare the absence of conflicts of interest in writing this article. The work was carried out within the framework of the dissertation work. The authors used their own funds in the fulfilment of the scientific work.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML