-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2024; 14(10): 2649-2654
doi:10.5923/j.ajmms.20241410.42
Received: Sep. 22, 2024; Accepted: Oct. 20, 2024; Published: Oct. 25, 2024

Features of FTO Gene Polymorphicm (rs9939609) in Children with Abdominal Obesity and Metabolic Disorders
Mardonkul R. Rustamov1, Lilya M. Garifulina2
1Doctor of Medical Sciences, Professor, Samarkand State Medical University, Samarkand, Uzbekistan
2Doctor of Philosophy, Associate Professor, Samarkand State Medical University, Samarkand, Uzbekistan
Copyright © 2024 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

76 children with varying degrees of obesity and 40 children with normal body weight were examined. The frequency distribution of the FTO gene polymorphicm (rs9939609) was determined. It was revealed that the FTO gene polymorphicm (rs9939609) is one of the factors of genetic predisposition to abdominal obesity with a pronounced increase in BMI, while allele A increased the risk of accumulation of excess visceral adipose tissue in abdominal obesity. A significant increase in blood triglycerides and a decrease in high-density lipoprotein cholesterol in children with the AA genotype were determined. Carriers of the A/A and T/A genotypes had a significant disturbance in the level of hunger hormones and saturation of leptin, ghrelin and YY3-36 peptide.
Keywords: Abdominal obesity, FTO gene, Leptin, Ghrelin, YY3-36 peptide
Cite this paper: Mardonkul R. Rustamov, Lilya M. Garifulina, Features of FTO Gene Polymorphicm (rs9939609) in Children with Abdominal Obesity and Metabolic Disorders, American Journal of Medicine and Medical Sciences, Vol. 14 No. 10, 2024, pp. 2649-2654. doi: 10.5923/j.ajmms.20241410.42.
1. Introduction
- Over the past decades, the prevalence of obesity has increased several times in all countries in the world. At the same time, the most unfavorable dynamics are observed among children and adolescents, since in these age groups, the growth rate of obesity incidence is 8 times faster than its growth rate among the adult population of the planet [1,2]. Obesity has a tendency towards rejuvenation [3]. In the Republic of Uzbekistan, there is also an increase in the primary and general incidence of obesity among children and adolescents, and its growth rate is significant [4].The need to study risk factors for the development of obesity in children with its complications is determined by the continuing trend of increasing abdominal obesity in the population. An increase in body mass index (BMI) in children and adolescents is recognized as an independent predictor of the development of comorbid conditions that make up the metabolic syndrome [5]. Thus, abdominal obesity in adolescents is recognized as an independent risk factor for stroke at a young age, the development of cardiovascular pathology, diseases of the gastrointestinal tract, and kidney pathology [6].Genetic factors occupy a leading place in the structure of risk factors for the development of abdominal obesity, amounting to 25 to 70% [7]. The 12th version of the Human Obesity Gene Map includes more than 600 genes, genetic markers and chromosomal regions directly or indirectly associated with the obesity phenotype. One of these genes is the FTO gene (Fat Mass and Obesity Associated), which is associated with fat mass and obesity [8].The FTO gene is localized on chromosome 16q12.2 and encodes the synthesis of the FTO protein [3]. The expression product of the FTO Gene is predominantly concentrated in the hypothalamus, namely in the centers of hunger and satiety. The FTO protein is involved in energy metabolism and the metabolism of body cells [7,9]. The existence of a single nucleotide polymorphicm (SNP) T/A (rs9939609) in the FTO gene was revealed. The A allele of this gene is associated with the risk of developing primary obesity, while owners of the AA genotype (16% of the population) are more at risk of accumulating fat mass than owners of the TT genotype (37% of the population) [7,8,9].Currently, special importance is attached to the study of genetic predictors of obesity and its main complications, in connection with which many studies have been conducted. However, studies devoted to the analysis of the influence of genetic factors on the development of obesity and, especially, its comorbid pathology in children of the Uzbek population are incomplete and sporadic, which requires a close study of the problem.This is especially important since the healthcare system of our country is entrusted with a number of tasks aimed at improving the quality of medical services provided to the population and adapting them to international standards, including preserving the health of young children, early diagnosis of diseases that arise in them, and reducing complications.In connection with the above, we were given a goal: to study the features of the FTO gene polymorphicm (rs9939609) in children with abdominal obesity and find a relationship with metabolic disorders.
2. Material and Methods
- Studies were conducted on the basis of family clinics in the city of Samarkand, as well as Samarkand regional branch of the Republican Specialized Endocrinological Scientific and Practical Medical Center named after Academician Y.H. Turakulova (Uzbekistan). The study involved 76 children aged 7 to 18 years with exogenous constitutional obesity, with the average age of the children being 12.02±0.46 years. The control group consisted of 40 children with a normal body weight without the presence of chronic and acute diseases at the time of examination. Children in the control group had a similar age as in the main group, the average of which was 12.14±0.27 years.Anthropometric studies were carried out using standard measuring instruments (floor stadiometer and medical scales). Anthropometric measurements include: height, body weight, waist and hip circumference. Comparison of the obtained data and assescment of physical development were carried out using cumulative centile tables of WHO age and gender distribution of height and body weight for children 5-19 years old [10]. Body mass index (BMI) was calculated from the measurements.The results of anthropometric studies were assessed using standard deviations of body mass index (SDS) in accordance with the recommendations of the World Health Organization (WHO) [10]. The basis for the diagnosis of Obesity was the determination of the intersection point of age and BMI, above +2.0 SDS BMI, overweight was diagnosed with indicators ranging from +1.0 to +2.0 SDS BMI, and underweight from -1.0 to -2.0 SDS BMI.76 children of the main group had a BMI of +2.0 to ≥+3 SDS, i.e. children had a BMI characterizing obesity from degrees I-III, with the average BMI being 32.18±0.5 kg/m2, the average SDS BMI was in the range of 2.56±0.11. At the same time, in the control group, BMI was in the range from +1.0 to -1 SDS, with an average BMI of 19.28 ± 0.26 kg/m2 and an average SDS BMI of 1.10 ± 0.08, which was statistically significantly lower compared with the main group (P<0,001). The anthropometric data of the children in the main group determined the WV, which had an average level of 102.06±1.51 cm, which was significantly higher compared to the control group 66.74±0.81 cm (p<0,001). Wherein, HV at higher rates in the main group (88.14±1.85 cm) did not statistically differ from the indicators of children in the control group (77.21±1.06 cm; р>0,05). An important indicator was the ratio of WV/HV characterizing the presence of abdominal obesity, which were statistically higher values in children from the main group with exogenous constitutional obesity (0.98±0.00 compared to the control 0.79±0.01; р<0,001).The presented data characterize significant differences in body weight in the study groups, while age, divided by gender, had no statistical differences (41 (53.9%) boys and 35 (46.1%) girls in the main group, and 21 (52.5%) boys and 19 (47.5%) girls in the control group).The lipid profile was studied on an automatic biochemical analyzer- Cobas Integra 400 Plus (Roche, Germany) using original test systems (Roche, Germany) with determination of the concentrations of total triglycerides, high-density lipoprotein cholesterol using absorption photometry.Determination of leptin, ghrelin and YY3-36 was carried out by the enzyme immunoassay method on the Human Reader HS apparatus, using the Human LEPTIN ELISA Kit, Human GHRL (Ghrelin) ELISA Kit, ELISA DSL-10-33600 test systems (manufactured by Elabscience USA).The study of the FTO gene polymorphicm (rs9939609) was carried out using a polymerase chain reaction using the method of allelic discrimination. Reverse transcription and PCR reactions were carried out using commercial kits LLC RPC "Litex" (Russian Federation). DNA samples were isolated from the patients' blood using phenol-chloroform extraction.The polymerase chain reaction was carried out in a 25 µl volume in two tubes, one of which contained forward primer 1 and reverse primer, the second - forward primer 2 and reverse primer. After amplification and subsequent electrophoresis in a 3% agarose gel in the presence of ethidium bromide, the amplification results were detected in ultraviolet light. The statistical processing of the obtained data was carried out on a personal computer using the Statistica 10 program. Methods of variational parametric and non-parametric statistics were used to determine the arithmetic mean (M), standard deviation (σ), standard error of the mean (m) relative values (frequency, %). The statistical significance of the obtained measurements was determined by Student's test (t) with a calculation of the probability of error (P). During genetic studies, allele frequencies and frequencies of allelic combinations were calculated and their correspondence to the Hardy-Weinberg equilibrium using the χ2 criterion with the calculated ones, rejecting the null hypothesis at P<0.05.
3. Results and Discussion
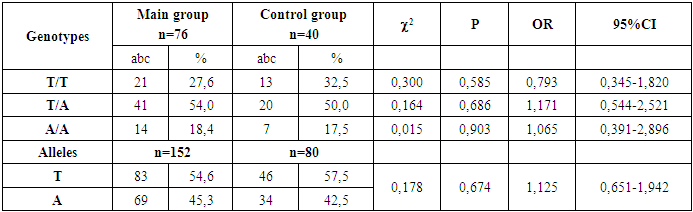
- The search for genetic risk factors for the development of obesity has great practical importance, since hereditary factors remain unchanged throughout a person’s life. Because obesity is polygenic in nature, the search for those genetic markers that are most responsible for the development of excess weight is especially relevant. Literary sources in recent years indicate that the AA genotype of the FTO gene polymorphicm (rs9939609) plays a significant role in the development of obesity in adults, while in children, in regional conditions, these studies have not been conducted.In the course of our research, we determined the frequency of occurrence of the FTO gene polymorphicm (rs9939609) in children with exogenous constitutional obesity. As a control, the blood of apparently healthy children with normal body weight without the presence of chronic pathology was presented.During the study, the distribution of allele and genotype frequencies for the FTO gene polymorphicm (rs9939609) in apparently healthy children did not differ from world literature data and amounted to the frequency of the T/T genotype in 40.4% of cases, the T/A genotype in 53.8% of cases, and in the cmallest percentage the A/A genotype in 5.8% of cases (Table 1).
|
|
|
|
4. Conclusions
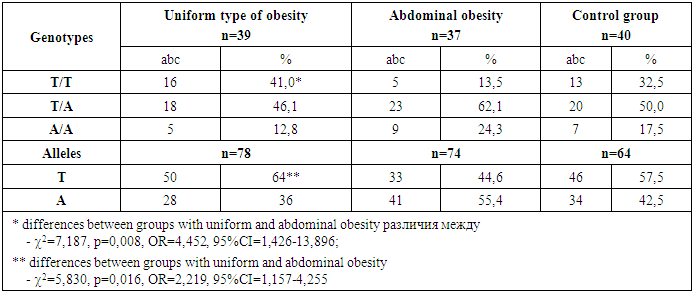
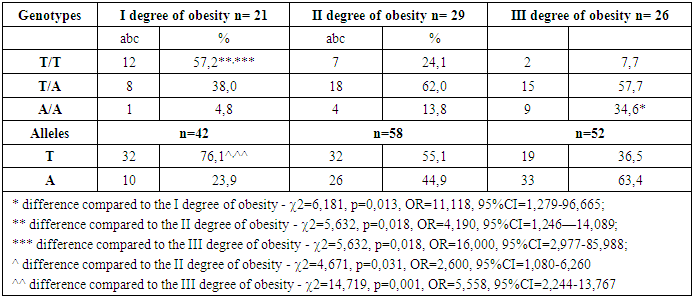
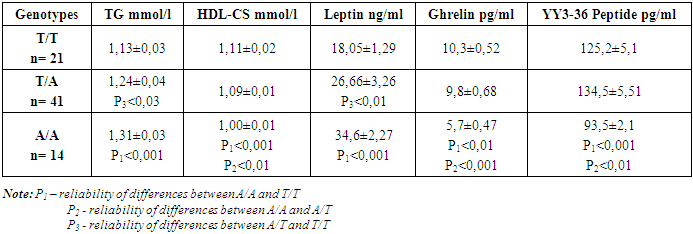
- Polymorphicm of the FTO gene (rs9939609) is one of the factors of genetic predisposition to abdominal obesity with a pronounced increase in BMI.The presence of the A allele increases the risk of accumulation of excess visceral adipose tissue in abdominal obesity. This is explained by the fact that a gene containing nucleotide A is subject to greater expression than a gene containing nucleotide T.Carriers of the AA genotype are more susceptible to the development of metabolic disorders in obesity, which was reflected in significant pathology of blood triglyceride and HDL cholesterol in the children of this sample.Expression of the A/A and T/A genotype disrupts the functioning of the hunger center and stimulates an increase in food consumption, which was manifested in a significant, significant increase in leptin, ghrelin and YY3-36 peptide in children with the mutant A allele.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML