-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2024; 14(9): 2287-2294
doi:10.5923/j.ajmms.20241409.35
Received: Aug. 25, 2024; Accepted: Sep. 16, 2024; Published: Sep. 20, 2024

Metabolic Status of the Brain in Children and Adolescents with Diabetes, Taking into Account the Presence of or the Absence of Cognitive Deficits
Alidjanova D. A., Madjidova Ya. N.
Tashkent Pediatric Medical Institute, Tashkent, Uzbekistan
Copyright © 2024 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

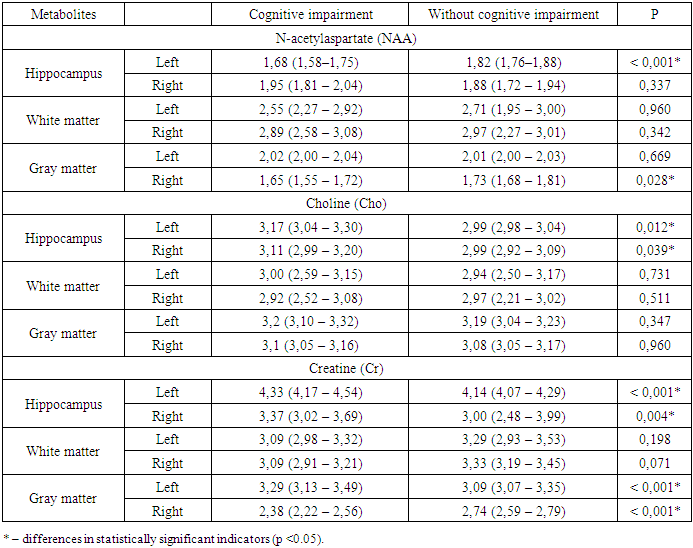
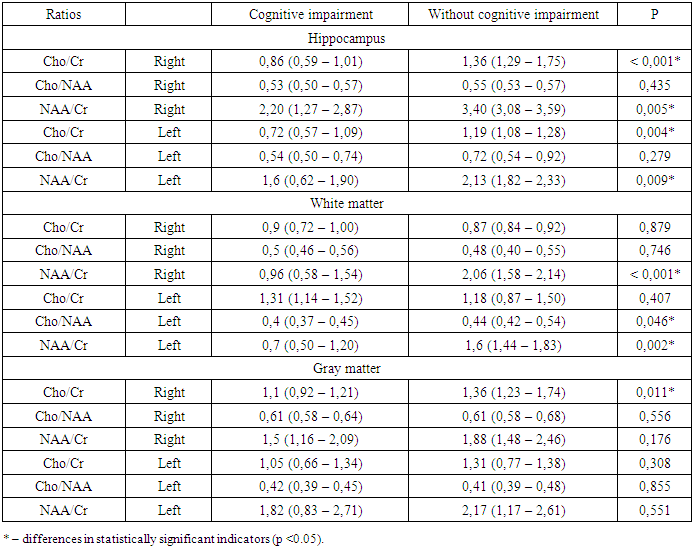
This publication will present an analysis of the results obtained from a study of the metabolic status of the brain using multivoxel proton magnetic resonance spectroscopy in children and adolescents with cognitive dysfunction in type 1 diabetes. According to the results obtained, correlations between the main metabolites of the brain – N-acetylaspartate (NAA), creatine (Cr), choline (Cho) and the results of the MoSA test will be demonstrated. The biochemical shifts discovered in the course of the study may be the primary links in the pathogenetic mechanisms of the formation of cognitive disorders in children and adolescents with type 1 diabetes.
Keywords: Cognitive dysfunction, Children and adolescents, Diabetes mellitus, Proton magnetic resonance spectroscopy, Brain metabolites
Cite this paper: Alidjanova D. A., Madjidova Ya. N., Metabolic Status of the Brain in Children and Adolescents with Diabetes, Taking into Account the Presence of or the Absence of Cognitive Deficits, American Journal of Medicine and Medical Sciences, Vol. 14 No. 9, 2024, pp. 2287-2294. doi: 10.5923/j.ajmms.20241409.35.
Article Outline
1. Introduction
- Diabetes mellitus (DM) refers to autoimmune pathologies of the endocrine system. The formation of diabetes occurs as a result of an absolute or relative lack of insulin induced by the disintegration of beta cells of the pancreas [1]. To date, diabetes is the most serious problem of the entire global medical community, as it does not lose its leading position in the deterioration of the quality of life and the formation of early disability due to the occurrence of long-term complications. According to the International Diabetes Federation, the expected increase in the number of diabetic patients in the world by 2045 is up to 628.6 million people. At the same time, there has been a sharp increase in type 1 diabetes in children and adolescents in recent years. Unfortunately, the Republic of Uzbekistan is no exception to this statistic, so at the end of 2023, more than 260 thousand people were registered with diabetes mellitus in our country, of which almost 3,500 thousand children, more than 1,000 adolescents.Among the entire range of variability of complications of type 1 diabetes in children and adolescents, deviations from the brain occupy a primary place, the fundamental manifestations of which are changes in the processes of cognitive activity. In recent years, many scientific studies have been conducted and published confirming both direct and indirect effects on the occurrence of structural and functional disorders of the brain in diabetes mellitus [2]. Such close attention to cerebral, namely cognitive abnormalities in diabetes is associated not only with the normal daily functioning and social adaptation of the child, but also with regular and clear self-control of glycemia, which has a direct impact on the overall dynamics of the underlying disease.According to epidemiological studies, all kinds of changes on the part of higher brain functions in type 1 diabetes can begin to form within the first 2-8 years after the onset of the disease. And their wide range of manifestations (10-75%) is almost always associated with such factors as the age of onset of the disease, duration of diabetes mellitus, reference values of glycemia, diagnostic methods used, etc. [3,4]. The pathogenetic mechanisms of the formation of cognitive deficits in type 1 diabetes mellitus, for the most part, remain uncertain and poorly understood. To date, hypo- and hyperglycemia, insulin deficiency, malfunction of pro- and antioxidant activity systems, activation of the polyol pathway of glucose metabolism, intensification of non-enzymatic glycation of proteins, hypoxic changes in brain tissues, induction of cell apoptosis, hemodynamic and hemorheological changes are distinguished among the pathogenetic components. In addition, in some research papers, the demonstrated correlations between cognitive dysfunctions and the degree of hyperglycemia gave the authors reason to assert that this main metabolic change may be the cause of cognitive disorders in DM [5,6]. Thus, in the process of cerebral disorders, there is an interest of various pathogenetic links, which may explain the polymorphism of the clinical manifestations of these disorders in type 1 diabetes. However, it is worth noting here that if the picture is more or less clear with the formulation of various pathogenetic factors, then the question of determining the main primary pathogenetic mechanism for the development of cognitive dysfunction in type 1 diabetes in children and adolescents remains open. In order to diagnose cognitive deficits in a variety of pathological processes, researchers widely use methods of neuropsychological testing of patients. Among these numerous methods, the Montreal Cognitive Assessment Scale – the Montreal Cognitive Assessment (MoSA test), developed for screening disorders of higher brain functions in a fairly short time, is the most popular. This technique is characterized by ease of use, reliability and is considered the most rational method of analyzing cognitive activity, the test is characterized by a fairly large range of study areas of the cognitive sphere [7]. The specificity of this method is 90% [8]. However, even with all the positive assessments of this method, it is not necessary to exclude the possible presence of a subjective assessment of indicators, which entails a significant percentage of both false positive and false negative results of neuropsychological testing. In this regard, and in order to maximize the objectification of indicators, it is necessary, in addition to neuropsychological testing, to conduct neuroimaging diagnostic methods, which at the present stage have found their wide application. For example, the proton magnetic resonance spectroscopy (PMRS) method is a lifetime, non-invasive neuroimaging method, which in a short time by means of metabolic state analysis makes it possible to determine biochemical shifts in any pathology in brain tissues, while at the earliest stages of the disease, when there are no structural changes yet. In addition, PMRS allows you to track both the dynamics of these metabolic changes and shows the effectiveness and expediency of prescribed therapy [9,10]. During the application of MR spectroscopy, it is possible to obtain data, that is, to visualize the following main metabolites of the brain: N-acetylaspartate (NAA); choline (Cho); creatine (Cr)/creatine phosphate (PCr); lactate (Lac); glutamate (Glu). If we consider each of the metabolites in more detail, then, for example, N-acetylaspartate can be determined only in nervous tissue and at the same time it is considered a marker of neuronal and axonal integrity. A decrease in its concentration indicates the loss or damage of neurons, which can occur with various brain injuries [11,12]. The level of choline provides information and is a marker of the functioning of cell membranes. An increase in its concentration can be observed with an increase in the number of cells, an increase in membrane synthesis, or with increased membrane destruction [13]. Creatine is a marker of the metabolic state of brain cells and has the greatest stability among other metabolites, however, an increase in its concentration occurs in a hyperosmolar response to any damage, and a decrease indicates degenerative changes in brain tissues [14]. Thus, cognitive disorders, along with type 1 diabetes itself in children, are among the primary problems of the modern medical community. The reasons for this circumstance are the high prevalence of both DM and cognitive dysfunctions with it, as well as their medical, social and socio-economic significance. In the future, the method of MR spectroscopy may make it possible to analyze the dynamics of the metabolic status of children and adolescents with type 1 diabetes, while starting from the earliest stages of the disease, demonstrating the progression of pathology, as well as the effectiveness of selected therapy to correct abnormalities of higher cortical activity.The purpose of the study. Determination of the metabolic status of the brain in children and adolescents with type 1 diabetes, taking into account the presence or absence of cognitive impairment.
2. Material and Methods
- Our study involved 205 children in the age range from 7 to 18 years old suffering from type 1 diabetes. Children from 7 to 11 years old (average age – 9.0±1.6 years) accounted for 81 patients –39.5%), 124 (60.5%) patients from 12 to 18 years old were examined, the average age of which was 14.7±1.8 years. In accordance with the conditions of the study, all patients underwent an objective clinical and neurological examination with a detailed examination of personal data and clarification of anamnesis. Changes in higher brain activity were determined using the Montreal Cognitive Function Assessment Scale (MoSA test) [15]. The level of glucose concentration in the blood was determined using a quantitative method on a Hitachi 912 biochemical tester (Germany). On the DS5 tester (Netherlands), the concentration of glycated hemoglobin (HbA1c) in capillary blood was determined by liquid chromatography. MR spectroscopy of the main metabolites of the brain was performed immediately after the introduction of routine MRI, while not changing either the apparatus or the position of the child's body, with a relaxation time of TE = 135 ms, the volume of one voxel was 1.5 cm3. [16]. The survey was carried out in a multi-pixel mode, which provides an option for placing 64 voxels on one slice simultaneously. Peaks of the main spectra of N-acetylaspartate, choline, and creatine, as well as their ratios, were recorded in the pre-selected areas of greatest interest. The choice of mathematical methods was determined taking into account the tasks set in each specific case and according to the criteria for processing medical data.
3. The Results and Their Discussion
- When evaluating the total score of the MoCA test, it was found that in 156 (76.09%) patients with type 1 diabetes, the result was less than 26 points. Of these, 2 (1.3%) children had 22 points; 44 (28.2%) patients had 23 points; 29 (18.5%) children had 24 points and 81 (51.9%) patients had 25 points. At the same time, the performance of testing tasks by patients was significantly (p < 0.001) worse than in healthy children (Figure 1).
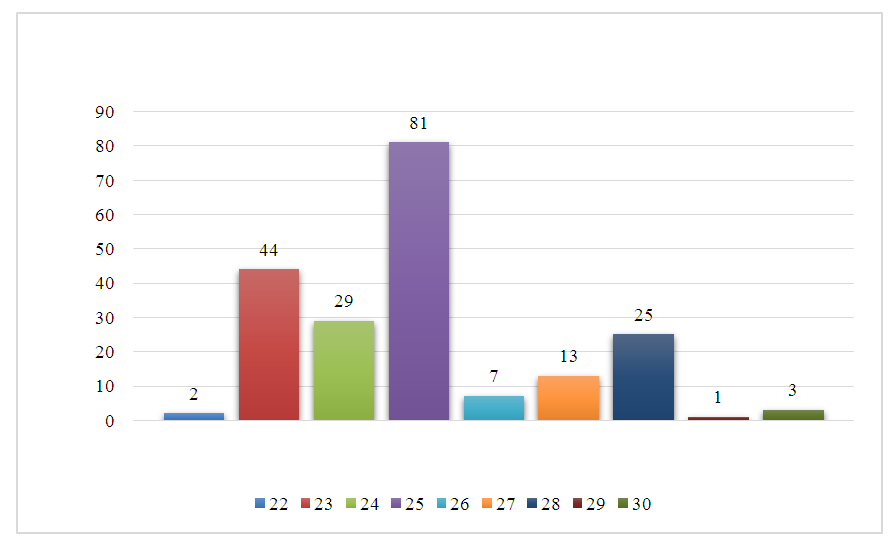 | Figure 1. The average score on the MoCA scale, depending on the presence of diabetes |
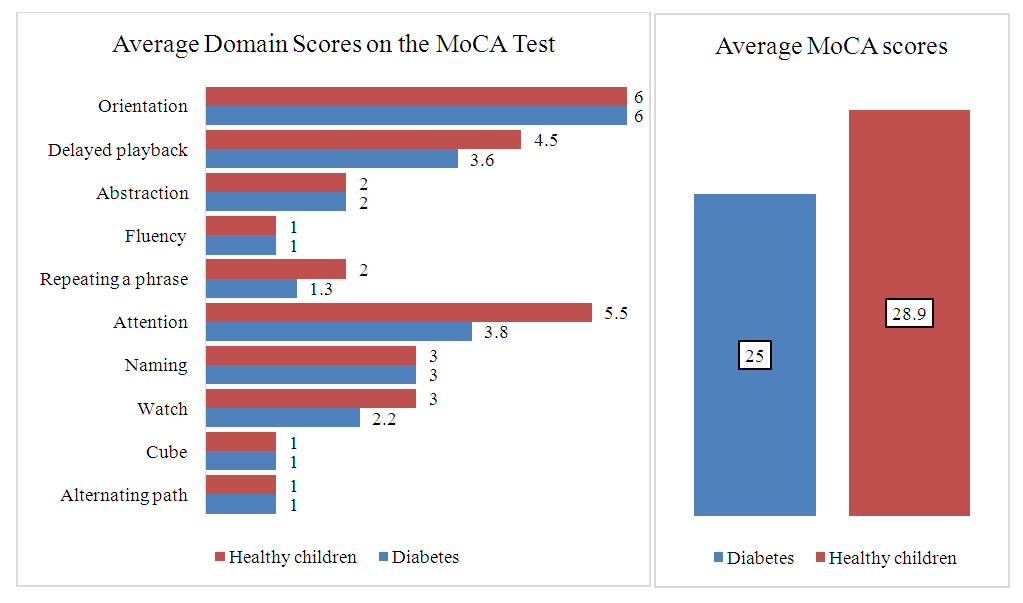 | Figure 2. The average score for the domains of the MoCA scale, depending on the presence of SD |
|
|
4. Conclusions
- Thus, when analyzing MR spectroscopy in children and adolescents with cerebral changes in type 1 diabetes, the main abnormalities of metabolites were determined in areas directly related to the processes of cognitive activity, that is, in the structures of the limbic-reticular complex. The changes we found revealed a multi-vector nature of biochemical abnormalities that occur during the formation of cerebral disorders, which were largely interrelated, complemented and enhanced the pathological effect of each other. At the same time, as mentioned above, one of the key factors was the energy deficit of nerve cells, which causes the activation of catabolic processes, which in turn contributes to the disruption of the synaptic apparatus, leading to a decrease in the efficiency of the information dissemination process in neurons. In addition, changes in the cerebral metabolic status are manifested by a decrease in the neuronal activity of the hippocampus, as well as the white and gray matter of the brain, and with an increase in the length of the disease and depletion of energy resources, these disorders tend only to progress with the occurrence of neuronal degenerations and demyelination processes in these areas of the brain. Given the importance of this circumstance for the optimal functioning of higher cortical activity, the detected biochemical shifts should be considered within the framework of the main links in the pathogenesis of cognitive disorders in children and adolescents with type 1 diabetes. And the use of PMRS as a method of the earliest diagnosis of cerebral changes in patients with type 1 diabetes is considered appropriate and justified. The PMRS study, in addition to being able to localize the areas of the brain responsible for cognitive decline in children and adolescents with type 1 diabetes, provided more accurate information about metabolic changes in these areas of the brain. In general, this method allows you to determine the dynamics of the condition of patients with type 1 diabetes mellitus, the presence of cerebral changes in them, while at a stage when there are no clinical manifestations yet, in addition, to monitor the possible progression of cognitive impairment, as well as monitor the adequacy of selected therapy to correct abnormalities of higher cortical activity, in addition PMRS it makes it possible to detect not only metabolic changes in the brain at the early stages of the formation of cognitive deficits, but even to predict the development of atrophic changes in the cerebral cortex, taking into account the length of the disease, since the level of NAA, Cr and Cho decreases 3.2 times faster than the normalized volume of the brain, which indicates the preceding loss of neurons and axons to the formation of atrophic changes.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML