-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2023; 13(10): 1456-1461
doi:10.5923/j.ajmms.20231310.20
Received: Sep. 16, 2023; Accepted: Sep. 27, 2023; Published: Oct. 8, 2023

The Effect of Biomayse on the Fatty Acid Composition of Blood in Experimental Atherosclerosis
Azizova D. M.1, Sabirova R. A.1, Tursunov D. H.1, Mavlyanov I. R.2, Soliev A.2
1Tashkent Medical Academy, Republic of Uzbekistan
2Republican Scientific and Practical Center for Sports Medicine
Copyright © 2023 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

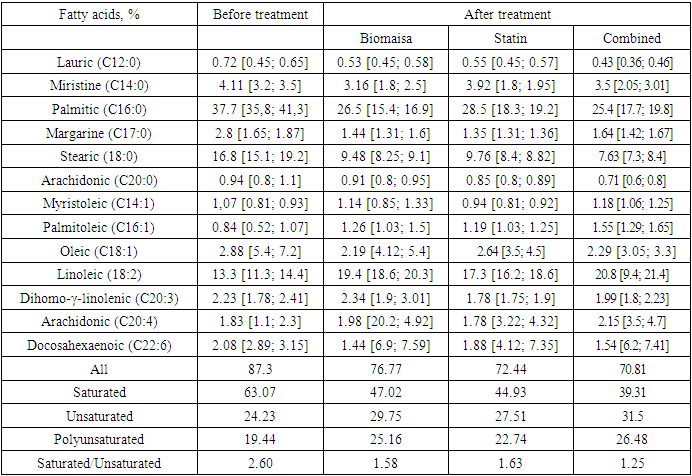
Aim. To study theeffect of biomayse on the fatty acid composition of blood in experimental atherosclerosis. Material and Methods. The experiments were carried out in 30 male rabbits divided into 5 groups. Experimental hypercholesterolaemia was reproduced by daily intragastric administration of cholesterol at 0.2 g/kg of body weight for 2 months. Then, the animals in groups 3, 4 and 5 were treated for 30 days. Ultrox (Nobel Farm) was used as a statin and was administered at 0.6 mg/kg and Biomais at 142 mg/kg. Biomaisa is a wheat germ powder and was provided by ORION-SKORPION Ltd. The fatty acid composition of blood serum was determined on a TRACE 1310 TSQ 8000 triple quadrupole chromatography-mass spectrometer with a gas chromatograph (GC‒MS/MS) and a CTC TriPlus RSH robotic autosampler from Thermo Fisher Scientific (USA). Results. Studies of the fatty acid contents in the blood serum after feeding rabbits for 2 months with cholesterol showed an increase in the total fatty acid content by 13.71% compared with that of intact animals. At the same time, the increase in saturated fatty acids was 65.75% compared with intact animals. In animals with experimental atherosclerosis, the amount of monounsaturated and polyunsaturated acids decreased by 1.58 and 1.67 times, respectively, compared with intact animals. Conclusion. The content of saturated fatty acids with the combined use of biomaysa and statins was normalized (39.31 versus 38.5 for intact animals), and the unsaturated and polyunsaturated fatty acid contents in the treated groups did not reach the values of intact animals, although a decrease in their contents was found when compared to the control group. The ratio of saturated fatty acids to unsaturated fatty acids decreased most pronouncedly in the group of animals treated with biomaysa and a statin simultaneously.
Keywords: Experimental atherosclerosis, Fatty acids, Statins, Wheat germ
Cite this paper: Azizova D. M., Sabirova R. A., Tursunov D. H., Mavlyanov I. R., Soliev A., The Effect of Biomayse on the Fatty Acid Composition of Blood in Experimental Atherosclerosis, American Journal of Medicine and Medical Sciences, Vol. 13 No. 10, 2023, pp. 1456-1461. doi: 10.5923/j.ajmms.20231310.20.
Article Outline
1. Introduction
- Atherosclerosis is a chronic inflammatory syndrome that leads to progressive thickening of the walls of large arteries, that is, the “arteries of the elastic (aorta and its branches) and muscular-elastic (arteries of the heart, brain, etc.) types” [1], as well as decreased blood flow over time, which can lead to stroke, myocardial infarction (MI), and VCID [2].In Uzbekistan, more than half of the deaths among the population aged 30-70 years are related to cardiovascular disease (CVD). Over the past five years, their incidence has increased by 20%, even among young people. Overall, approximately four million people are diagnosed with CVD, which is 12% of the total population.Due to the high mortality of the population from CVD, scientists around the world have been studying the mechanisms of AS development for many years. The result of these studies has been the emergence of a large number of theories of atherogenesis. Dominant among them are theories involving lipoprotein infiltration, proposed by the pathologist N.N. Anichkov in 1913 [3], and endothelial dysfunction, put forward in the mid-1970s by R. Ross.Previously, it was thought that the symptoms of atherosclerosis depended solely on the degree of stenosis, which determines the degree of perfusion of target tissues, such as the brain and heart [4].Although impaired NO production is a major feature of endothelial dysfunction (ED), the production and/or utilization of other vasodilatory agents such as prostacyclin (PGI2) and bradykinin are also impaired in the context of ED. Free fatty acids (FFAs) or nonesterified fatty acids (NEFAs) are well-known risk factors for CVD [5] and are closely associated with metabolic syndrome (MS) events such as obesity and type 2 diabetes mellitus (DM2) [6]. Recent studies have shown that FFAs are not only the main causes of insulin resistance [7,8] but are also responsible for inducing inflammation in tissues targeted by insulin, such as endothelial cells (ECs), the liver and skeletal muscle [9]. Therefore, elevated blood FFA levels are seen as an important link between insulin resistance, inflammation, obesity, T2DM, and arterial hypertension (AH) [Boden G._ Obesity and free fatty acids. Endocrinol Metab Clinic N Am. 2008; 37 (3): 635–46.]. Impaired insulin-mediated glucose uptake correlates with circulating FFA levels, and this insulin resistance may be due to FFA-mediated phosphoinositide 3-kinase (PI3K) inactivation.In addition, a growing body of evidence suggests an important role for FFAs in mediating ED. In particular, insulin resistance, oxidative stress, and inflammation are significant causes of FFA-induced ED [10-12]. Generally, diabetes and other metabolic conditions can lead to increased levels of FFA, which in turn has a direct effect on transcription factors that cause inflammation and increased stress in the body [13]. In addition, FFAs also promote apoptosis/necroptosis of ECs [14-16] and mediate many deleterious effects on endothelial progenitor cells (ECs) [17].Fatty acids are able to participate in metabolism in the human body, and they can undergo elongation, desaturation and oxidation. However, among them, it is very important to highlight the essential ones—those that the body can only obtain with food. Polyunsaturated fatty acids are considered the most important fatty acids. Numerous studies have shown that their increased consumption reduces the risk of developing malignant tumours, weakens insulin resistance, and reduces the severity of cardiovascular pathology [18-19]. The most studied are eicosapentaenoic (EPA, timnodonic) and docosahexaenoic (DHA, cervonic) acids contained in the fat of marine fish, as well as vegetable alpha-linolenic acid (ALA). These are omega-3 acids, which means that the first unsaturated (double) bond between alkyl radicals is at position 3 from the methyl end of the molecule.In connection with the above information, it is very important to consume unsaturated fatty acids with food or to add them to the diet in the form of biologically active additives (BAAs) to stimulate the body to fight atherosclerosis.The purpose of the study was to examine the content of fatty acids in the blood serum with respect to the dynamics of the development of experimental atherosclerosis and correction by Biomaysa.
2. Research Methods
- Experiments were carried out on 30 male rabbits of the Chinchella line divided (depending on the purpose of the study and the method of treatment) into 5 groups (with 6 rabbits each). Experimental hypercholesterolaemia was reproduced by daily intragastric administration of cholesterol at 0.2 g/kg of body weight for 2 months. Then, the animals in groups 3, 4 and 5 were treated for 30 days. Ultrox (Nobel Farm) was used as a statin and was administered at 0.6 mg/kg and Biomais at 142 mg/kg. Biomaisa is a wheat germ powder and was provided by ORION-SKORPION Ltd.The fatty acid composition of blood serum was determined on a TRACE 1310 TSQ 8000 triple quadrupole chromatography-mass spectrometer with a gas chromatograph (GC‒MS/MS) and a CTC TriPlus RSH robotic autosampler from Thermo Fisher Scientific (USA).
3. Statistical Analysis
- The standard Jamovi software package was used for statistical processing. The normality of the distribution of scores was assessed using the Shapiro‒Wilk test and/or kurtosis and skewness analysis. In cases of normal distribution, quantitative data are presented as the mean and standard deviation (M ± 50) and 95% confidence intervals. If the data distribution differed from normal, the data are presented as the median (Me) and percentiles (25%-75%). The level of statistical significance of the indicators was defined as p< 0.05.
4. Research Results and Discussion
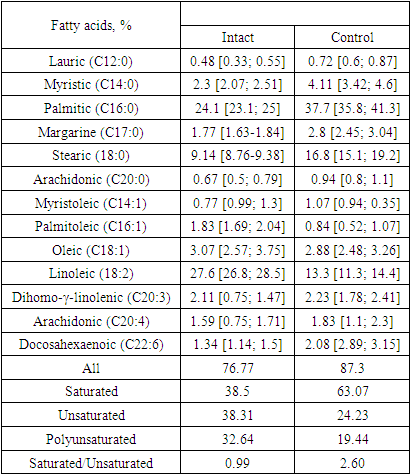
- Free fatty acids promote apoptosis/necroptosis of endothelial cells (ECs) [20-22] and mediate many detrimental effects on EC progenitors [23].The results of the study of the fatty acid contents in the blood serum of rabbits with experimental atherosclerosis are shown in Table 1. As seen from the data in Table 1, when feeding rabbits cholesterol for 2 months, an increase in the content of total fatty acids by 13.71% occurs in comparison with that in intact animals. At the same time, the increase in saturated fatty acids was increased by 65.75% compared with that in intact animals. Such an increase in the content of saturated fatty acids occurs due to increases in lauric, palmitic, margaric, myristic and stearic acids. The content of these acids is increased by 1.5, 1.56, 1.58, 1.78 and 1.83 times, respectively, compared with intact animals. These data indicate the main role of palmitic acid in the development of hypercholesterolaemia, and an increase in its content can serve as a predictor of the development of atherosclerosis [24]. With an increased content of palmitic acid in food, a large amount of palmitic VLDL is formed in the body, which disrupts the synthesis of LDL. Additionally, fatty acid-binding proteins (FABPs) are not able to transfer all of the latter into the cell, or the proteins composing the LDL are not activated. Thus, a large amount of nonligand palmitic LDL is formed, which subsequently turns into “biological garbage” in the blood, but the body cannot absorb it and tries to destroy it by any means. The active components of the complement system “recognize” m-LDL labelled by neutrophils and contribute to the formation of ligands, which help the “biodebris” enter the intima of the arteries [25]. In the intima of the arteries, sedentary macrophages (a type of phagocyte) begin to act on m-LDL. Macrophages perceive m-LDL as a protein macromolecule. In the lysosomes of macrophages, proteolysis (decay) of the apoB protein occurs. However, macrophages cannot hydrolyse cholesterol-esterified essential fatty acids (cholesterol esters), which are found in large quantities in LDL. Nonhydrolysed cholesterol esters first accumulate in macrophage lysosomes; later, they occupy the entire cytoplasm and form foam cells [26], triggering a pathophysiological process called “storage disease”. Violation of the integrity of lysosomes leads to autolysis and the death of macrophages [27]. When the number of dead macrophages formed exceeds the capacity limit of the intima of the arteries, the artery swells and becomes exfoliated. An atherosclerotic plaque forms. In animals with experimental atherosclerosis, the amounts of unsaturated and polyunsaturated acids decreased by 1.58 and 1.67 times, respectively, compared with the amounts in intact animals. The ratio of saturated/unsaturated fatty acids in animals with experimental atherosclerosis increased 2.62 times compared to intact animals. The content of polyunsaturated fatty acids in intact rabbits was significantly higher than that in the control group, and the presence of polyunsaturated fatty acids increased the level of cholesterol absorption in the intestine and its conversion to cholic acids with subsequent excretion from the body. It should also be noted that with an increase in palmitic VLDL in the blood, the intake of essential PUFAs (linoleic and linolenic) decreases simultaneously for cells. These essential PUFAs are structural elements of cell membranes and ensure the normal development and adaptation of the human body to adverse environmental factors. Essential fatty acids composing LDL, together with palmitic acid, also cannot penetrate the cell, and this may be the main factor in the initiation of the cascade of the pathological process.
|
|
5. Conclusions
- The content of saturated fatty acids was normalized with the combined use of biomaysa and statins (39.31 compared with the intact group at 38.5), and the unsaturated and polyunsaturated fatty acids in the treated groups did not reach the values of intact animals, although a decrease in their contents was found compared to the control group. The ratio of saturated fatty acids to unsaturated fatty acids decreased most pronouncedly in the group of animals treated with biomaysa and statin simultaneously.An important cause of the changes in lipid metabolism and the accumulation of lipids in arterial walls may be local hypoxia, which develops under the influence of intimal hyperplasia [34]. The lack of oxygen seems to cause arterial smooth muscle cells to synthesize additional neutral lipids from free fatty acids in the blood instead of using FFAs in the oxidation process to the full extent. When they accumulate in lipid vacuoles (in conditions of oxygen deficiency or high levels of FFA), these lipids will contribute to cell degeneration. This view of atherogenesis may explain the accelerated development of atherosclerosis in a number of conditions accompanied by an increase in plasma free fatty acids, such as obesity, metabolic syndrome, diabetes mellitus, and chronic stress [35-38].It should be especially noted that sprouts are a natural product. All useful substances are found in them in natural, balanced amounts and appropriate combinations. These substances are built into the organic system of living tissue, and their absorption does not adversely affect human health, which can be observed when using certain pharmaceuticals. In addition, enzymes formed in germinating seeds breakdown complex storage substances (proteins, fats, and carbohydrates) into simpler ones (amino acids, fatty acids, and simple sugars).
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML