-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2023; 13(8): 1087-1090
doi:10.5923/j.ajmms.20231308.12
Received: Jul. 27, 2023; Accepted: Aug. 11, 2023; Published: Aug. 12, 2023

Assessment of the Functional State of the Gastric Mucosal Gland Structure in Steroid-Dependent Nephrotic Syndrome in Children
Bobomuratov Turdiqul Akramovich1, Samadov Abduqakhkhor Abdurakhmonovich2, Mamadiyorov Abdibullo Milyevich3
1Head of the Department of Propaedeutic of Children Diseases, Tashkent Medical Academy, Tashkent, Uzbekistan
2Assistant Department of Propaedtutics of Children Diseases, Tashkent Medical Academy, Tashkent, Uzbekistan
3PhD in Medical Sciences, Deputy Chief Physician of the Multidisciplinary Clinic of the Tashkent Medical Academy, Tashkent, Uzbekistan
Copyright © 2023 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

In recent years, there has been a sharp increase in chronic kidney disease among children. In the USA, Europe, Australia, Asian countries, it is noted that every ten of the world's population suffers from kidney dysfunction. Nephrotic syndrome is found in 90% of children with chronic maturation until the age of 10, and approximately 50% of adult children. The main pathogenetic treatment for nephrotic syndrome in children is glucocorticosteroids, which causes gastric and duodenal mucosal lesions in children. For non-invasive endoscopic screening testing in children with nephrotic syndrome, the determination of the value of PG I and PG II s and the PG I/PG II ratio makes it possible to assess the condition of the gastric wall mucosa and the functional state of the glandular structure.
Keywords: Children, Nephrotic syndrome, Glucocorticosteroids, Pepsinogens
Cite this paper: Bobomuratov Turdiqul Akramovich, Samadov Abduqakhkhor Abdurakhmonovich, Mamadiyorov Abdibullo Milyevich, Assessment of the Functional State of the Gastric Mucosal Gland Structure in Steroid-Dependent Nephrotic Syndrome in Children, American Journal of Medicine and Medical Sciences, Vol. 13 No. 8, 2023, pp. 1087-1090. doi: 10.5923/j.ajmms.20231308.12.
Article Outline
1. Introduction
- In recent years, there has been a sharp increase in chronic kidney disease (CKD) among children. In the USA, Europe, Australia, Asian countries, it is noted that every ten of the world's population suffers from kidney dysfunction. [1,3,5,7] S is found in 90% of children with chronic maturation until the age of 10 and about 50% of adult children.The diagnosis of nephrotic syndrome was based on ISKDC, APN [(1974-2002)] by jaxon standards(proteinuria 1g/m2/milk, hypoalbunemia less than 25 g/l, dysproteinemia, hypercholesterinemia, tumors from peripheral edema to anasarca). According to APN (Arbeitsgemeinchaft fur Paediatrisce Nephrology), ISKDC, APN (1974-2009), nephrotic syndrome in children is administered without biopsy in the case of renal function sacking. [1,2,8]Depending on the response to the standard course of treatment with steroids, steroid-sensitive and steroid-resistant forms of nephrotic syndrome differ. 1. Steroid-sensitive nephrotic syndrome is observed in children with minimal - change nephrotic syndrome (MCNS), remission is achieved within 2-4 weeks, in some patients - 6-8 weeks, and only in 4% of cases-12 weeks after the start of treatment: in SSNs-complete long-term, remission is achieved, which is not repeated after one course of steroid treatment.2. After frequent relapsing NS - remission is achieved, relapses-more than 2 times in 6 months (FRNS) are observed.3. Steroid-dependent NS-prednisolone is a steroid-dependent nephrotic syndrome (SDNS) that has not achieved remission even after an 8-week course of treatment and 3 Solu-Medrol courses.The main pathogenetic treatment for nephrotic syndrome in children is glucocorticosteroids. [3,4,5,7]According to the recommendations of international and Russian scientists, for the treatment of nephrotic syndrome, prednisolone is prescribed to drink at a dose of 60 mg/m2 per day or 2 mg/kg per day (dose 60 mg per day). at a dose of no more than) for a regular 4-6 weeks, then a day-to-day alternating regimen of the drug is offered at a dose of 40 mg/m2 or 1.5 mg/kg ( up to a maximum of a day-to-40 mg dose) for 4-6 weeks, a step-by-step reduction and a drug cancellation scheme is recommended [3,4,7]. The total duration of GKS therapy is 4-5 months [5,7]. The alternating mode of GKS intake increases the duration of remission compared to the intermittent intake mode (continuous) [4-6,16].As a result of long-term treatment of NS with prednisolone in tqns and SDNS clinical variants in children, it causes cases of oitis (various changes in the mucous membrane of the gastroduodenal zone, gastroduodenitis, Esophagitis and erosive-ulcerative changes), lagging behind growth and obesity. [1,6,7,8,9]. Children with frequent relapse (SDNS) and steroid - dependent nephrotic syndrome (SDNS), which are considered clinical variants of SDNS, need long-term GKS therapy, [3,5,8]. this causes gastric and duodenal mucosal lesions in children [1,2,3]. Based on the above, in children with nephrotic syndrome, an assessment of the functional state of the mucous membranes of the upper parts of the digestive tract and the gland structure of the stomach wall is considered relevant.Currently, the detection of PG I and PG II levels for non-invasive endoscopic screening testing in the Americas, Europe, Russia, Japan and CIS countries has been widely introduced. Pepsinogens are the proferments of pepsin that provide an early and important step in the digestive cascade, in the process of converting proteins into amino acids. [12,13,14]However, the level of pepsinogens in the blood is a reliable diagnostic indicator of the morphological and functional state of the Meda mucosa, allowing non-invasive biopsy. Indicators of the volume of pepsinogens, which vary from a wide range to a minimum to a maximum value, in many ways allow you to assess the risk of developing not only stomach diseases, but also various diseases associated with reflux esophagitis, duodenal ulcers. A reduced incidence of PG I and PG II is one of the criteria that determine the risk factor for atrophic gastritis and stomach cancer. [10,11,14] for this reason, 92 patients with NS who were included in the task of our study were examined in the serum laboratory immunoferment analysis (IFT) method, PGI, Rdii volume and PgI/Rdii ratio TMA multi functional clinical laboratory, with the aim of assessing the functional state of the gastric mucosal gland structure, as a result of EFGDS examination treated with GKS for more than 6 months.
2. The Purpose of the Study
- To study the functional changes of children with nephrotic syndrome and who have been taking GKS drugs for a long time, the mucous membrane of the gastrointestinal tract and the glandular structure of the stomach wall.
3. Research Materials and Styles
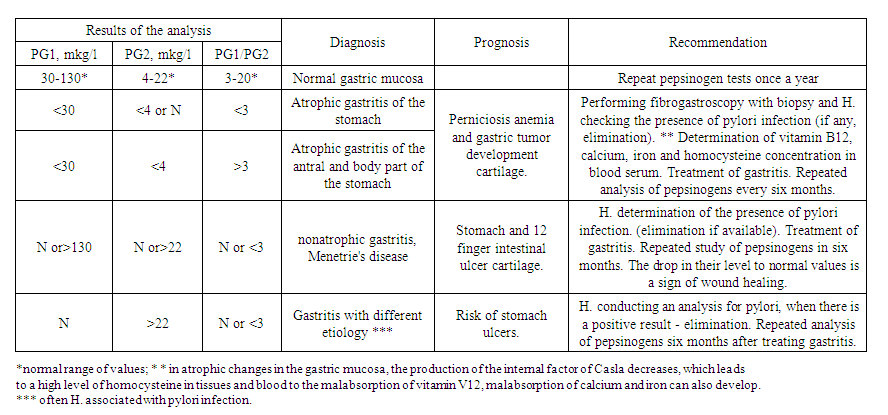
- In the 7-17pcs range, over 6 months with nephrotic syndrome, a GKS-treated EFGDS examination revealed changes in the stomach and duodenum in 92 patients tested in the serum laboratory immunoferment analysis (IFT) method, PGI, rdii volume, and PGI/Rdii ratio TMA multidimensional clinical laboratory.For IFT, the reagent kits PgI (catalog number D-3762) and Rdii (catalog number D-3764), developed on the Russian side of Vektor-Best CJSC, were used. Reference indicator PG1 - 30-130 mcg/l, PG2 - 4-22 mcg/l, PG1/PG2 ratio-more than 3.
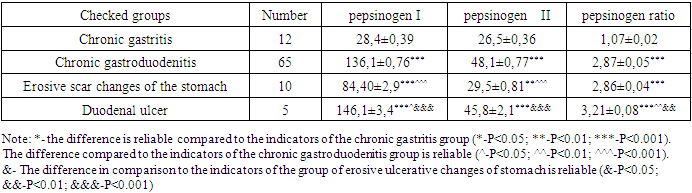
 | Table 1. Analysis of the results of the study of pepsinogens |
4. Results of the Study
- The result of Examinations is an EFGDS examination in the digestive tract, infected with nephrotic syndrome. In 9 children from the CDNS group and 3 from the SDNS Group, signs of hyperplastic and atrophic inflammation of the mucous membranes of the antral, body and fundal part were added together, in patient children who showed signs of pangastitis, PG I and PG II showed a uniform decrease in the rate of PG I 28.41 mcgr/l PG II volume average of 26.53 mcg/L, pepsinogen ratio PGI/PG II PG i <30, PG II <4, pg I/ PG II >3 (gastric antral and atrophic gastritis of the body part, Stomach and 12 fingers corresponded with the result of intestinal ulcer cartilage).As a result of the EFGDS investigation, the highest diagnosis of chronic gastroduodenitis in our study groups was 65 (70.6%) TDNS 15, while in children with SQNS 12 cases, local form of SGD accompanied by damage to the antral and body part was determined, with PG I found to be at a mean of 106.40 mcgr/l, PG II increased to 31.84 mcgr/l, PGI/PG II was found at a value of 2.98. 21 individuals from the TQNS group and 17 individuals from the SQNS group have a diffuse form of SGD inflammation of all parts of the gastric mucosa of the body-antral and bottom in the form of pangastrite and 12 fingers have been added together to the intestine in children who have been diagnosed with a licorice jar. While PG I 136.2 mcgr/l, PG II volume increased by more than 2 times the myory rate to an average of 48.36 mcg/L, pepsinogen ratio was found to have decreased by PGI/PGII-2.84, in quantity.In children with 10 medani erosive ulcerative changes detected by EFGDS testing in the TQNS and CRNS groups, the inflammatory process in most cases was observed to damage the mucous membranes of the body and bottom of the stomach. In these children, PG I was found to be 84.40 mcgr/L with a significant decrease compared to SGD pediatric coursework, while PG II was conversely increased compared to pepsinogen study results analysis coursework-the ratio of pepsinogens to 29.51 mcgr/l was reversed at a value of PGI/PGII-2.85. 12-finger intestinal ulcer disease was observed in 5 children PG I 146.1 mcgr/l, PG II 45.7 mcg/l was found to have a significant increase in the rate of analysis of pepsinogen studies, and reduced the ratio of pepsinogens to 3.21.In children who have taken predizolone more than 6 months with nephrotic syndrome, the average serum volume of PG I in various changes in the stomach and 12 fingers of the intestine is 3.21 mcg/L. The average volume of PG II was 1.16 mcg/l and the average ratio of PG I and PG II was 2.76 and a significant decrease in indicators was observed.
|
5. Conclusions
- In children with nephrotic syndrome, changes in the mucous membrane of the stomach and duodenum were observed in edema, hyperemia as well as various pathological signs of the gastroduodenal zone, accompanied by erosion, and in most cases damage to the mucous membrane of the esophagogastroduodenal area at the same time. In children with nephrotic syndrome, changes observed in the intestinal mucosa of the stomach were found to be in excess of the clinical forms of nephrotic syndrome and the duration of treatment with glucocorticosteroids.Based on Imunoferment analysis, the method of determining and analyzing the level of PG I and PG II in blood serum can be widely enslaved for the purpose of non-invasive endoscopic examination, to assess functional changes in the mucous membrane of the gastrointestinal tract and the gland structure of the gastrointestinal tract, children with nephrotic syndrome and long-term glucocorticosteroid drug treatment.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML