-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2023; 13(8): 1068-1072
doi:10.5923/j.ajmms.20231308.08
Received: Jul. 17, 2023; Accepted: Aug. 7, 2023; Published: Aug. 12, 2023

Concentric Myocardial Remodeling in Patients with Chronic Kidney Disease
Maksud Atabayevich Sabirov, Feruza Erkinovna Salyamova, Maksud Begmatovich Bobokulov
Tashkent State Dental Institute, Republican Specialized Scientific and Practical Medical Center for Nephrology and Kidney Transplantation, Tashkent, Uzbekistan
Copyright © 2023 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The aim of the research was to study the mechanisms of early remodeling of the myocardium in different stages of chronic kidney disease. Background. An increase of the Left ventricular mass and a change in its geometric model increase the risk of cardiovascular diseases and worsen the prognosis of patients with chronic kidney disease. Concentric myocardial remodeling (C-remodeling) was established by echocardiography in the presence of normal left ventricular myocardium and an increase in the relative thickness of the left ventricle walls. Material and methods. The study included 106 patients (50 men and 56 women) observed at the Republican Specialized Scientific and Practical Medical Center for Nephrology and Kidney Transplantation at the age of 44.9±13.6 years with normal left ventricular mass, without verified or possible coronary artery disease, hemodynamically significant valvular disorders and heart failure II-IV FC (by NYHA). Predialysis stage of chronic kidney disease occurred in 74 (69.8%), dialysis - in 32 (30.2%) patients. The duration of dialysis was 18.5 (3.0; 41.3) months. Results. Concentric myocardial remodeling represents the initial stage of developing of the left ventricle hypertrophy. This conclusion is confirmed by a significant increase in Left ventricular mass index during Concentric myocardial remodeling. As the results of our studies have shown, risk factors for Concentric myocardial remodeling in patients with chronic kidney disease are age, duration of previous hypertension, higher values of clinical blood pressure, which is consistent with previous findings about a similar relationship in patients with essential hypertension. Conclusion. Leading risk factors for Concentric myocardial remodeling in patients with chronic kidney disease include age, female gender, duration of previous hypertension, systolic blood pressure, diastolic blood pressure and hemoglobin levels, and more severe renal impairment. An additional risk factor for Concentric myocardial remodeling in hemodialysis patients is its volume overload.
Keywords: Chronic kidney disease, Cardiovascular diseases, Myocardial remodeling
Cite this paper: Maksud Atabayevich Sabirov, Feruza Erkinovna Salyamova, Maksud Begmatovich Bobokulov, Concentric Myocardial Remodeling in Patients with Chronic Kidney Disease, American Journal of Medicine and Medical Sciences, Vol. 13 No. 8, 2023, pp. 1068-1072. doi: 10.5923/j.ajmms.20231308.08.
1. Introduction
- Cardiovascular disorders (CVD), according to the world's leading dialysis centers, are the leading cause of morbidity and mortality in patients with chronic kidney disease (CKD) [1-4]. Mortality among dialysis patients from CVD is 40-60% of all deaths, which is 20-30 times higher than mortality in the general population. It was found that the predictor of a high risk of CVD and an unfavorable prognosis is an increase in the Left ventricular mass (LVM), regardless of arterial hypertension (AH) and coronary heart disease [5-6,1,7-9]. Modern studies have demonstrated that not only an increase of LVM, but also a change in the geometric LV model of the heart, which is identified as preclinical cardiovascular pathology (especially in asymptomatic patients), has a significant prognostic value at CKD [10-13]. It was found that the risk of developing cardiovascular complications is highest at concentric hypertrophy (CVD develops 4 times more often than at unchanged LV geometry), less with eccentric hypertrophy and concentric remodeling of the myocardium. The issue of risk factors and mechanisms of early changes in LV geometry in CKD remains unclear [5-6,11].The aim of the research was to study the mechanisms of early remodeling of the myocardium in different stages of chronic kidney disease.
2. Material and Methods
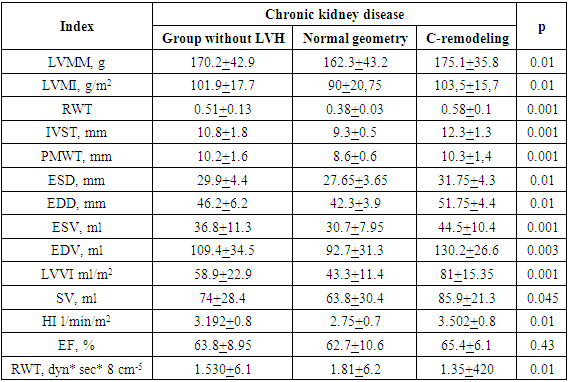
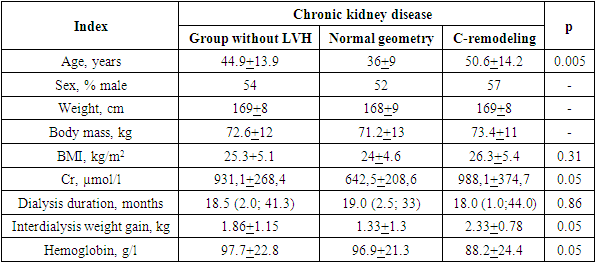
- The study included 106 patients (50 men and 56 women) observed at the Republican Specialized Scientific and Practical Medical Center for Nephrology and Kidney Transplantation at the age of 44.9±13.6 years with normal left ventricular mass, without verified or possible coronary artery disease, hemodynamically significant valvular disorders and heart failure II-IV FC (by NYHA). Predialysis stage of chronic kidney disease occurred in 74 (69.8%), dialysis - in 32 (30.2%) patients. The duration of dialysis was 18.5 (3.0; 41.3) months. The causes of CKD were: chronic glomerulonephritis (n=52), pyelonephritis (n=15), polycystic kidney disease (n=12), obstructive nephropathy (n=9), hypertensive nephroangiosclerosis (n=7), tubulointerstitial nephritis (n=3), other nephropathies (n=5). The diagnosis was not clear in 3 patients, due to the detection of the disease in the CKD stage. All patients were performed a standard clinical, laboratory and instrumental examination for patients with CKD. Predialysis CKD was classified according to creatinine clearance (Сcr) by the Cockcroft-Gault calculation method: Ccr (male)= (140-age) х weight (kg)/ Сr (mmol/l) х 0,81; Ccr (female)= Ccr (male) х 0.85. Clinical and laboratory parameters were taken into account as average monthly. The dry weight of dialysis patients was defined as the lowest post-dialysis weight without symptoms of hypovolemia in two months prior to the study and was used as the basis for establishing interdialysis weight. Blood pressure (BP) was measured with a mercury sphygmomanometer using the Riva-Rocci-Korotkov auscultatory method. The cuff was applied to the contralateral arm without an arteriovenous fistula or shunt in hemodialysis patients. BP was established as the mean of two measurements observed in two dialysis sessions, one pre-dialysis and one post-dialysis BP and presented as pre-dialysis and post-dialysis BP. Arterial hypertension was diagnosed at systolic BP (SBP) ≥140 mm Hg. and/or diastolic BP (DBP) ≥90 mmHg. Echocardiographic investigation was carried out on the Aloca SSD-2000 ultrasound system (Japan) in accordance with the recommendations of the American Echocardiographic Society. All hemodialysis patients were examined the day after routine dialysis in the middle of the week. Measurements of the LV end diastolic (EDD) and end systolic dimensions (ESD), LV posterior myocardial wall thickness (PMWT) and Interventricular septal thickness (IVST) were performed in the M-mode from the parasternal access. The relative wall thickness (RWT) of the LV was calculated using the following formula: RWT= IVST + PMWT / EDD. LV volume parameters were calculated by the L.Teichgolz method. The following indicators were also calculated: stroke volume (SV, ml), stroke index (SI, ml), ejection fraction (EF,%). The value of the total peripheral resistance (TPR) was calculated by the formula: TPR =80*BPav/MV, dyn * sek * cm-5, where BPav – average BP, which was calculated as BPav = DBP+SBP-DBP/3, mmHg. LV myocardial mass was calculated by the R.B.Devereux formula [4]: LVM=0.8 * 1.04{( IVST + PMWT +EDD) 3 – (EDD) 3 } + 0.6 g. LV mass (LVMI) and the LV volume indices (LVVI) were defined as the ratio of LVM and LVV to body surface area (BSA). To calculate the BSA, the formula was used: BSA (m2) = (0,0001) * (71.84) * (wt 0,425) * (ht 0,725), where wt - weight (kg), ht - height (cm). LV hypertrophy (LVH) is stated in absolute values LVMI ≥134 g/m2 in males and ≥110 g/m2 in females [3]. LV geometry was characterized based on LVMI and Relative Wall Thickness (RWT). It was considered normal if LVMI was not increased and RWT was less than 0.45. Concentric myocardial remodeling was diagnosed in cases when at unchanged LVMI, the RWT increased to 0.45 or more. Standard methods of descriptive statistics were used to analyze and evaluate the data obtained: calculation of means and their standard deviations (M±SD) for a normal distribution, median and interquartile range for a non-normal distribution. The Kolmogorov-Smirnov criterion was used to test the hypothesis of the normality of the distribution. The Kruskal-Wallis criteria and the Mann-Whitney U-test were used to analyze the differences in quantitative characteristics. The significance of quantitative differences was determined using Student's t-test for independent samples. Differences were considered significant at p<0.05. The SPSS 8.0 statistical software package from SPSS Inc (USA) was used for data processing.
3. Results
- According to the selection of patients with unchanged LVMI, two groups of patients were distinguished based on RWT: group I consisted of 59 (55.7%) patients with normal geometry of the left ventricle, group II - 47 (44.3%) patients with myocardial C-remodeling.When analyzing the relationship between the frequency of C-remodeling and the stage of CKD, it turned out that normal LV geometry occurred in 48 (64.8%) cases of pre-dialysis and only in 13 (41%) cases of dialysis CKD (р<0.05). At the same time, the frequency of myocardial C-remodeling was 26 (35.2%) cases in the predialysis and 19 (59%) cases in the dialysis stages of CKD (p<0.001) (Figure 1).
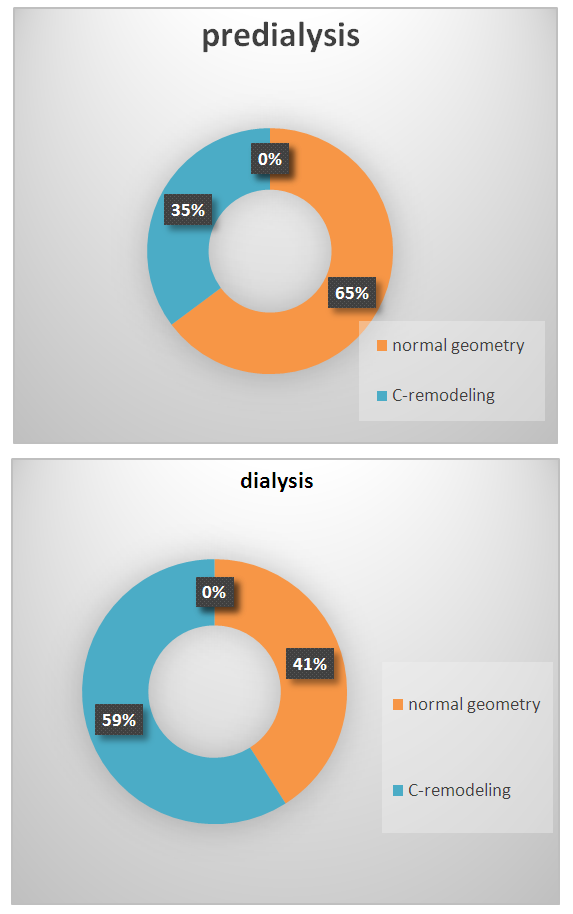 | Figure 1. The frequency of myocardial С-remodeling at different stages of CKD |
|
|
4. Conclusions
- In patients with CKD, C-remodeling of the myocardium is widespread. The frequency of myocardial C-remodeling increases as renal function decreases.C-remodeling of the myocardium represents the initial stage of the development of left ventricular myocardial hypertrophy. In turn, high systolic AH increases LV afterload, and a decrease in diastolic AH leads to a decrease in coronary perfusion, contributing to myocardial ischemia. Our studies have shown that, in patients with CKD grade 3-4, there is an association between systolic and diastolic arterial hypertension and left ventricular hypertrophy.A highly significant correlation was found between mean arterial pressure and left ventricular myocardial mass index, which did not depend on age and creatinine clearance.AH is the main trigger in the occurrence of left ventricular hypertrophy only at the initial stage of CKD [10]. CKD progression, the value of AH as the main risk factor for left ventricular hypertrophy decreases and the main role passes to other risk factors, as evidenced by the lack of relationship between the left ventricular myocardial mass index and AH in the predialysis period.There is often no nocturnal decrease of AH in patients with uremia, which correlates with an increase of left ventricular myocardial mass [14]. At the same time, according to other authors, the correlation between the value of diastolic pressure and the mass index of the left ventricular myocardium [8,17], the thickness of the interventricular septum is weak.The leading risk factors for myocardial C-remodeling in patients with CKD include age, female gender, duration of previous hypertension, SBP, DBP and hemoglobin levels in the blood, more severe renal dysfunction. An additional risk factor for myocardial C-remodeling in hemodialysis patients is its volume overload.Adequate blood pressure control, timely correction of anemia at the predialysis stage of chronic renal failure will slow down the process of myocardial remodeling, the development of left ventricular hypertrophy and improve the prognosis of patients with CKD. The authors declare no conflict of interest. This study does not include the involvement of any budgetary, grant or other funds. The article is published for the first time and is part of a scientific work.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML