-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2023; 13(4): 359-363
doi:10.5923/j.ajmms.20231304.04
Received: Mar. 8, 2023; Accepted: Mar. 25, 2023; Published: Apr. 13, 2023

Androgenic Profile of Women with Premature Ovarian Insufficiency
Fakhrutdinova Sevara, Khaidarova Feruza
Republican Specialized Scientific and Practical Medical Center for Endocrinology Named after Academician Y.Kh. Turakulova, Tashkent, the Republic of Uzbekistan
Correspondence to: Fakhrutdinova Sevara, Republican Specialized Scientific and Practical Medical Center for Endocrinology Named after Academician Y.Kh. Turakulova, Tashkent, the Republic of Uzbekistan.
| Email: | 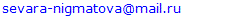 |
Copyright © 2023 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

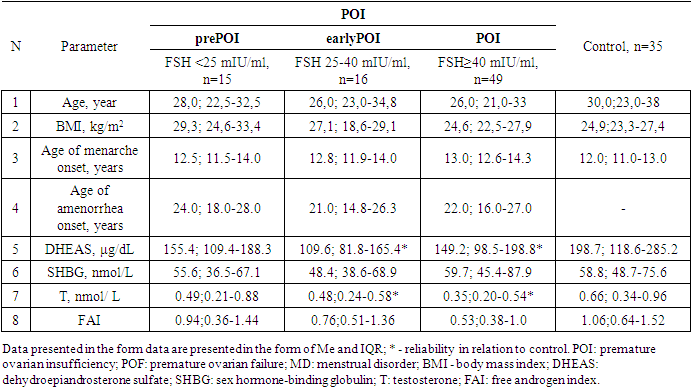
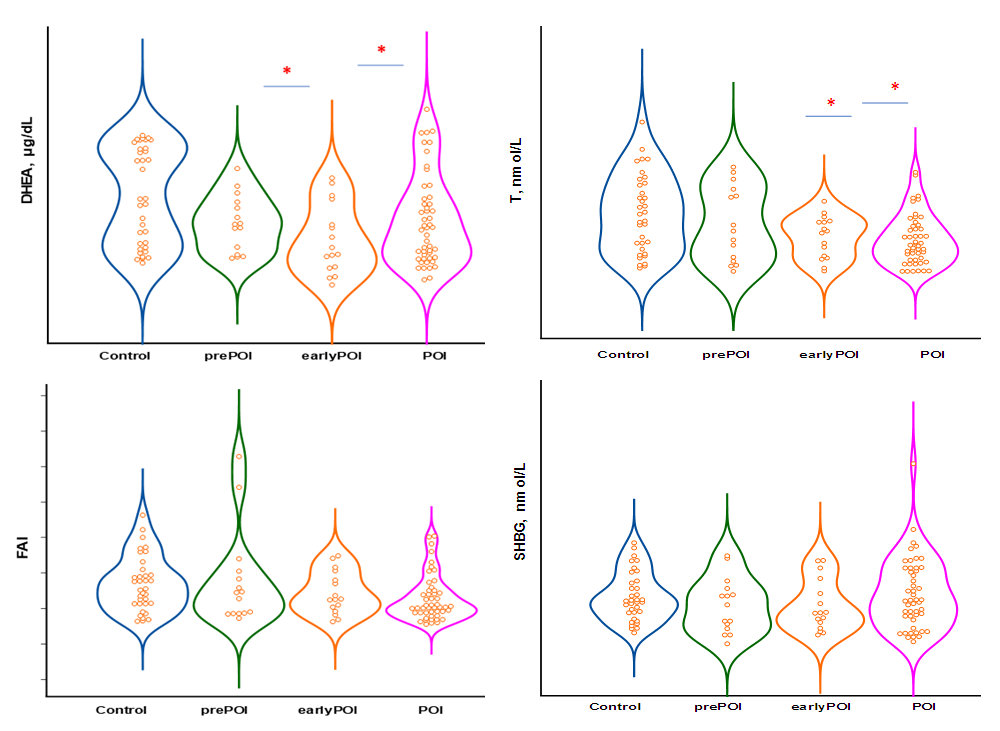
Premature ovarian insufficiency (POI) is a condition characterized by an irreversible decrease in ovarian function outside the normal range for women under 40 years of age. The purpose of the study is to assess androgens level in women with POI with different levels of FSH. Materials and methods. The main group included 80 women (mean age 28.0±7.2 years) with an idiopathic form of POI. The control group included 35 women (mean age 30.6±8.3 years) with regular menstruation and without ascertained endocrine disorders. Results: All women with POI were divided into groups based on ESHRE recommendations, group with FSH level >25 to 40 mIU/mL included 16 women, group with FSH level ≥40 mIU/mL included 49 patients. On average, in patients with POI, the level of DHEAS and T was statistically significantly lower than in the control group, respectively, but within the laboratory reference values. The level of SHBG and FAI did not differ in women with POI and in the control group, respectively. Similarly, no differences were found between POI subgroups. Diagnostically significant cut-off points were determined for DHEAS (187.5 µg/dl), SHBG (51.3 nmol/l), T (0.63 nmol/l) and FAI (0.64). DHEAS (AUC 0.640), T (AUC 0.674) and FAI (AUC 0.632). The level of DHAES (20.0%, 18.8%, and 20.4%) and T (20.0%, 25.0%, and 22.4%) were significantly lower in some patients than markers cut-off points in prePOF, early POF and POI groups, respectively. However, no significant difference was found between the groups. Conclusion: With the help of ROC analysis, diagnostically significant cut-off points for DHEAS, SHBG, T and FAI were determined. It was found that DHEAS (AUC 0.640), T (AUC 0.674) and FAI (AUC 0.632) had an medium diagnostic and prognostic value. Despite the low prognostic significance, androgens shall be taken into account as one of the factors in the development of cardiovascular diseases.
Keywords: Premature ovarian insufficiency, Androgenic profile, Risk groups
Cite this paper: Fakhrutdinova Sevara, Khaidarova Feruza, Androgenic Profile of Women with Premature Ovarian Insufficiency, American Journal of Medicine and Medical Sciences, Vol. 13 No. 4, 2023, pp. 359-363. doi: 10.5923/j.ajmms.20231304.04.
Article Outline
1. Introduction
- Premature ovarian insufficiency (POI) is a condition characterized by an irreversible decrease in ovarian function outside the normal range for women under 40 years of age [1].According to some researchers, POI exists as a continuum of changes in ovarian function, which includes a "latent" state in which women have reduced fertility, but have normal FSH levels and regular menstruation, a "bio-chemical" state with reduced fertility, elevated FSH levels, but regular menstruation and finally a "clear" condition with reduced fertility, elevated FSH levels, and irregular or no menstruation [2,3,4,5].According to the literature, in women with POI, a decrease in ovarian production of estradiol and progesterone is sometimes accompanied by a decrease in the level of circulating androgens compared with women with normal ovarian function [6,7,8,9,10].Androgens play a significant physiological role in women, acting both as precursors to estrogen biosynthesis and directly through androgen receptors. The most significant biologically active androgen is testosterone (T), which circulates in close connection with sex hormone binding globulin (SHBG) synthesized in the liver. Approximately 78-80% of androgens bind to SHBG, about 19-20%- to albumin, and only a small part of testosterone (1-3%) circulates freely in the blood and shows biological activity [10,11]. Some authors have suggested that premature cessation of ovarian function can lead to a hypoandrogenic state. Since hypoandrogenism is associated with a decrease in estrogen concentration (a decrease in the peripheral conversion of testosterone to E2 by fat tissue) and, possibly, with a deterioration in well-being and sexual health, as well as an increased risk of cardiovascular disease [7,12,13]. Androgen deficiency in women is characterized by decreased libido, poor health, depression, decreased muscle mass and prolonged causeless fatigue [14]. Glucocorticoids, androgens, growth hormone, synthetic progestins, insulin contribute to a decrease in the content of SHBG in the blood. SHBG concentration in plasma increases by 5-10 times under the influence of estrogens and decreases by 2 times under the influence of testosterone [11].
2. Purpose of the Study
- The purpose of the study was to assess androgens level in women with POI with different levels of FSH.
3. Materials and Methods
- The main group included 80 women (mean age 28.0±7.2 years) with an idiopathic form of POI. The control group included 35 women (mean age 30.6±8.3 years) with regular menstruation and without ascertained endocrine disorders. All women in the study signed an informed consent to an anonymous analysis of their medical data.According to ESHRE recommendations, the diagnostic criteria for POI are: oligo / amenorrhea for at least 4 months and elevated FSH level >25 IU / l in two cases with an interval of >4 weeks [15].Blood sampling was performed in the follicular phase (3-5th day of the menstrual cycle) in controls and against the background of amenorrhea in women with suspected POI.The levels of hormones in the blood serum were determined (FSH, DHEAS, SHBG and T) by the electrochemiluminescent method on the immunochemical analyzer Elecsys and cobas e. using standard Cobas Roche kits («Roche Diagnostics GmbH», Germany). The normal reference ranges used in our laboratory were as follows: FSH: 3.5–12.5 mIU/ml; DHEAS: 60.9-407.0 μg/dL; SHBG: 32.4-128.0 nmol/L; T: 0.29-1.67 nmol / L. FAI indicator was calculated by the formula: Testosterone x100/SHBG.
4. Statistical Analysis
- Statistical processing of the results was carried out using programs Microsoft Excel, IBM SPSS Statistica 23 and MedCalc version 18.5. The initial data were evaluated for compliance with the normal distribution according to Kolmogorov-Smirnov criterion. Dependences were analyzed using Spearman's rank correlation coefficients. To assess the prognostic significance of markers, ROC analysis (Receiver Operating Characteristic) was used, with the calculation of the area under curve AUC (area under the curve), Se (sensitivity) and Sp (specificity) of the model. Results are presented as median (Me) [interquartile range Q25; Q75]. Differences were considered statistically significant at p<0.05.
5. Results and Discussion
- All women with POI were divided into groups taking into account ESHRE recommendations, the group with FSH level >25 to 40 mIU / ml included 16 women, the group with FSH level ≥40 mIU / ml included 49 patients.Since the maximum reference range of FSH in our laboratory was 12.5 mIU/ ml, the study included women under 40 years of age with menstrual disorders (secondary amenorrhea lasting 6 months or more), with FSH level <25 mIU / ml, high LH level and low E2 level, obtained twice with an interval of 4 weeks, AMH concentration of was also taken into account. We identified this cohort of women (n = 15) as a group of high risk of premature ovarian failure – POF (mean age 28.7±8.0 years), they did not take medications (for 6 months before the examination), which could affect hormonal and biochemical indicators.Thus, 3 groups of patients were identified: group 1of high-risk- POF precursor (prePOF) - FSH <25 mIU / ml (n=15), group 2- early POF (earlyPOF - FSH 25-40 mIU / ml (n=16) and Group 3- POF with FSH ≥40 mIU/ml (n=49). It should be noted that the majority (61.3%) of women with POF had FSH level ≥40 mIU/ml.Kalantaridou S. et al. [8] believe that androgens play an important biological role in women in maintaining bone mass and normal sexual function. According to the authors, androgen deficiency may explain a significant decrease in mineral density of bony tissue even against the background of standard estrogen/progestogen hormone therapy in two-thirds of young women with spontaneous POI. A significant factor affecting T clearance is the level of SHBG, which binds T and reduces its free fraction. The analysis revealed no differences in age and BMI between the three groups. The age of the examined patients and persons from the control group ranged from 18 to 45 years. Age of onset of menarche, age of menstrual disorder, age of onset of amenorrhea did not differ significantly between the groups. Nevertheless, menstrual disorder and amenorrhea were noted in early POF and POI groups approximately 1.5-2 years earlier than among patients with prePOF. (Table 1.).
|
 | Figure 1. Changes in androgenic status at various stages of ovarian insufficiency |
 | Figure 2. ROC-curves for forecasting POI |
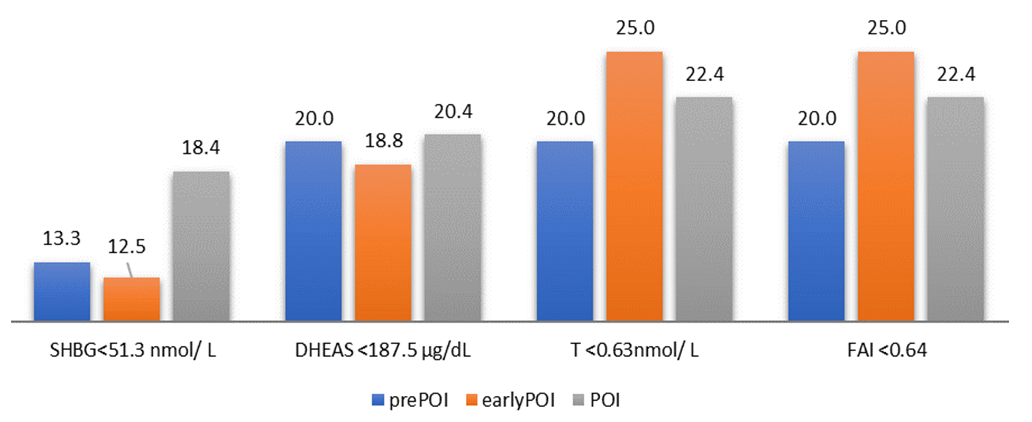 | Figure 3. The occurrence of androgens and FAI below the cut-off point in patients with POI, in % |
6. Conclusions
- With the help of ROC analysis, diagnostically significant cut-off point was determined for DHEAS (187.5 μg / dL), SHBG (51.3 nmol / L), T (0.63 nmol / L) and AAI (0.64). DHEAS (AUC 0.640), T (AUC 0.674) and ISA (AUC 0.632) had average diagnostic and prognostic value. Despite the low prognostic significance, androgens shall be taken into account as one of the factors in the development of cardiovascular diseases.Conflict of interest: The author has no conflict of interest to declare.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML