-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2022; 12(9): 975-982
doi:10.5923/j.ajmms.20221209.26
Received: Sep. 7, 2022; Accepted: Sep. 21, 2022; Published: Sep. 28, 2022

Description of the Immediate Results of a Multimodal Optimized Approach in the Treatment of Breast Cancer in Combination with Conventional Therapy
Sabirov Jahongir Ruzievich1, Gafur-Akhunov Mirza Aliyarovich2
1Associate Professor of the Department of the Tashkent Medical Academy, Uzbekistan
2Head of the Department of Oncology of the Center for the Development of Professional Qualification of Medical Workers of the Ministry of Health of the Republic of Uzbekistan
Copyright © 2022 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The aim of the study is to conduct an in-depth analysis of the immediate results of a multimodal optimized approach in the treatment of breast cancer with conventional therapy. In this work, we used combinations of some well-known therapeutic methods and an algorithm for their application. Materials and methods. An analysis was made of the immediate results of treatment of 118 patients with breast cancer in the initial stages who were treated in a hospital. The patients were divided into two groups: the control group (64 patients) who received traditional therapy and the main group (54 patients) who received traditional therapy with modulated accompanying immunocorrective drug therapy with preliminary exposure to medium frequency radio waves (350+15 kHz). The age of the studied women ranged from 31 to 73 years: 30-40 years-16 (13.5%); 41-50 years old - 32 (27.2%), 51-60 years old - 41 (34.7%), 61-70 years old -25 (21.2%), 71 and older - 4 (3.4%). 83 (70.3%) patients were young and of working age. Results. The evaluation of the effectiveness of the proposed method of the multimodal approach was carried out depending on the time of onset of partial lysis, a decrease in the volume of tumor formation and regional lymph nodes, changes in immunological parameters, and improvement in the quality of life of patients. One of the evaluation criteria was the time of onset of tumor lysis. A statistically significant difference was shown during the proposed EMRWT (electromagnetic radio wave therapy) and its combinations in comparison with the traditional one, in the control group, the onset of lysis was 21 days, in the main group - 7 days. In visual diagnostics (ultrasound was preferred, except for MSCT and mammography), the size of the tumor and regional lymph nodes, regression of the perfocal inflammatory process, echocardiography of tumor lysis were determined. There was a difference in the onset time and a decrease in the volume of tumor formation, enlarged regional lymph nodes of the proposed multimodal approach compared to the traditional one (p<0.05): in the control group - 7 days, the main one - 21 days. Conclusions: To activate antitumor immunity, both physical and medicinal agents can be used, which enhance the cytotoxic reactions of the cellular immune response. Immunity correction prior to oncolytic therapy makes it possible to effectively carry out measures for the destruction of tumor tissue. Degraded tumor antigens can already be perceived by the immune system and an immune response can be developed against them. At the same time, the tumor becomes more “recognizable” for the immune system. It is very important that under conditions of immunosuppressive effects of chemotherapy or radiotherapy, the functional activity of the immune system remains within the physiological norm.
Keywords: Breast cancer, Accompanying immunocorrective therapy, Medium and low frequency radio waves, Cellular and humoral factors of immunity
Cite this paper: Sabirov Jahongir Ruzievich, Gafur-Akhunov Mirza Aliyarovich, Description of the Immediate Results of a Multimodal Optimized Approach in the Treatment of Breast Cancer in Combination with Conventional Therapy, American Journal of Medicine and Medical Sciences, Vol. 12 No. 9, 2022, pp. 975-982. doi: 10.5923/j.ajmms.20221209.26.
- Breast cancer is a heterogeneous disease, the pathogenesis of which is caused by a complex interaction of genetic, hormonal, metabolic, exogenous and other factors and is the most common malignant neoplasm among women in the world [1-9]. Of the 184 countries covered by the World Health Organization's GLOBOCAN database, breast cancer (BC) is the most common cancer in women in 140 countries (76%) and the most common cause of cancer death in 101 countries (55%). Approximately one in twelfth women will develop breast cancer during their lifetime. In 2020, approximately 685,000 women die from this disease. Most breast cancer cases and deaths occur in low- and middle-income countries (LMICs) [10-13]. Five-year life expectancy for breast cancer exceeds 90% in HIC, but is only 66% in India and 40% in South Africa [14-16].Significant progress has been made in the treatment of breast cancer since the 1980y. till 2020 y. in HIC, age-standardized mortality from this type of cancer was reduced by 40%. But LMICs have yet to achieve comparable performance. Improving outcomes and survival is possible with a combination of early detection and subsequent effective treatment using existing methods - surgery, radiation therapy and drug treatment. In order to increase and enhance the effectiveness of treatment, there are different approaches and techniques. The treatment of this disease requires an integrated approach that combines local (surgical and / or radiation) and systemic drug treatment at stages I-III, [17-19].Immunotherapy is one of the most promising and rapidly developing areas in the treatment of cancer. However, it is constrained by a number of factors that do not allow this method to be completely successful. First, the tumor successfully masks or eliminates antigens that might be recognized by the immune system. Secondly, the tumor not only masks itself, but also actively affects the immune system, enhancing the tolerogenic and suppressive properties of its cells, reprogramming the functions of tumor-associated macrophages and suppressing cell killing reactions. Thirdly, the tumor behaves like a parasitic, alien organism, using all the mechanisms of Darwinian evolution. That is, it actively changes and adapts to adverse conditions arising from chemotherapy, radiotherapy or other methods of exposure. The most important issue is to increase the effectiveness of the methods and approaches used in treatment of breast cancer [20-24]. Despite the introduction of modern high information technologies and IT solutions into clinical practice, screening programs to improve methods of early diagnosis and effective systemic treatment, high rates of morbidity and mortality from oncological diseases remain. Therefore, the problem of increasing the effectiveness of cancer treatment is one of the most important and topical issues in modern medicine. Moreover, it is known that disturbances in various regulatory systems of the body and disruption of homeostasis mechanisms associated with the tumor process can be aggravated during intensive courses of chemotherapy and radiation therapy, and at the same time, the principle of a "vicious circle" can develop, which can cause even more decrease in antitumor resistance. In this regard, recently in oncology and immunology, more and more attention is paid to areas that are focused on the need to mobilize the body's natural defense mechanisms against a tumor, i.e., antitumor immunity [25-29]. Thus, studies of the influence of electromagnetic fields on living organisms have been conducted for more than a decade. It is known that electromagnetic fields are an environmentally significant environmental factor, since all living organisms on the planet are under the influence of the natural geomagnetic field and man-made fields [30,31]. This information served as an impetus for the development of magnetotherapy, especially in oncology. Various authors provide data on the benefits of magnetic therapy in the rehabilitation treatment of cancer patients, which is expressed in the normalization of blood counts, the elimination of postoperative complications, the acceleration of reparative processes, and the removal of severe pain. Thus, the widespread use of electromagnetic fields, especially in oncology, contributes to the accumulation of information about the mechanisms of action of this physical factor on the course of the oncological process. Actively studied are the mechanisms of antitumor immunity under the influence of electromagnetic fields, both at the cellular and molecular levels [32-34]. It was previously known that magnetic fields increase blood circulation in tissues and stimulate the body's metabolism. Also, it has been proven that low and medium frequency electromagnetic waves inhibit the growth of malignant tumors, thereby stopping the neoangiogenesis necessary for tumor growth. Magnetic therapy induces weak electrical currents in tissues, which increases the surface potential of cells, which leads to increased blood circulation, oxygenation, nutrient supply and better removal of metabolic waste from the body. Also, magnetic fields have been used as natural pain relievers, promoting recovery and healing, reducing swelling, stiffness and acidity of wounds. As regards research on the immune system, it should be said that we are now in a state of active accumulation of material on the study of the effect of magnetic radio waves in tumor processes.
1. The Aim of the Study
- To conduct an in-depth analysis of the immediate results of a multimodal optimized approach in the treatment of breast cancer with conventional therapy. In this work, we used combinations of some well-known therapeutic methods and an algorithm for their application. When carrying out complex treatment of breast cancer, a differentiated and selective combination was used. Treatment fits well into the system of traditional therapy and allows you to gradually influence the processes of tumor elimination:a) the course of therapy begins with a non-specific correction of the immune system. As a device for non-specific immunocorrection, the “TOR” technology (technology of operational rehabilitation) was used with a non-localized point of influence using a copper coil on the patient’s body, which affects the immune system with an alternating magnetic field in the medium frequency range of 350 + 15 kHz, at a wavelength of 909-857 meters and with an impact energy of 20V. During preliminary immunological experiments, it was found that when exposed to a given alternating magnetic field, the number of T-lymphocytes and CD16 NK cells significantly increases. At the same time, the patient is taking oral immunomodulatory drug "RCV" (Uzbekistan). It has been shown that this drug activates the cytotoxic functions of macrophages (i.e., it is likely that reprogramming from M2 to M1), intensively enhances the synthesis of interferons (IFN) and increases the number of active CD4+ and CD8+ subpopulations of T-lymphocytes. According to the indications of the immunogram and the data of biochemical blood tests, additional immunotropic drugs, macro- and microelements, as well as vitamins and other drugs can be prescribed.b) After such a preparatory course, antitumor treatment begins within 2-3 weeks. Low-dose chemotherapy or radiotherapy can be carried out for quite a long time (1-2 months) and even more. The main thing is to periodically carry out clinical and laboratory monitoring of the patient's condition, preventing deterioration of his condition. At the same time, one should not strive at all costs to destroy the entire tumor at once. The main thing here is not to suppress immune responses. The processes of tumor lysis, in conditions of full-fledged work of the immune system, will continue even after the end of the course of chemotherapy. Lysis can be carried out by activated cells of innate (M1 and NK cells) and acquired (CD8+ - T - cytotoxic lymphocytes) immunity. Even if small doses of chemotherapy no longer kill cancer cells, they can significantly change their metabolism. This already leads to a decrease in the immunosuppressiveness of tumor cells, a slowdown in their growth and, often, simply to aging. Moreover, low doses of cytostatics can very actively suppress angiogenesis in the tumor by blocking vascular endothelial growth factors (VEGF). All this further inhibits tumor growth.c) Before the end of the course of chemotherapy (7-10 days in advance), the patient begins to take drugs that suppress the explosive growth of tumor cells. The group of such drugs may include Ablast (Uzbekistan), Celecoxib, Aspirin, Metformin, selenium preparations and others. This stage lasts for 15-25 days.d) During the next, recovery stage (1-2 months), the patient takes adaptogens (Ginseng, Eleutherococcus, vitamin C and D3 and other drugs) and continues the course of therapy with TOR according to a certain scheme. At this time, the effectiveness of the completed course is determined. With incomplete destruction of the tumor, after the recovery period, the above course, taking into account the necessary adjustments, especially in chemotherapy agents, can be repeated.It should be noted that the goal of this optimized approach is at least partial regression of the tumor mass and the absence of its further growth. One should not strive to quickly and by any means destroy the entire tumor to the last cell, as this can enhance the already pronounced effects of intoxication of the body and immunosuppression.
2. Materials and Research Methods
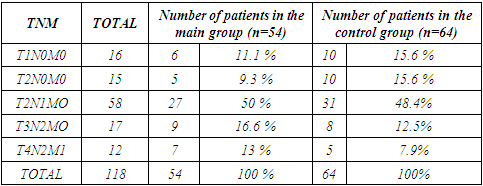
- An analysis was made of the immediate results of treatment of 118 patients with breast cancer in the initial stages who were treated in a hospital. The patients were divided into two groups: the control group (64 patients) who received traditional therapy and the main group (54 patients) who received traditional therapy with modulated accompanying immunocorrective drug therapy with preliminary exposure to medium frequency radio waves (350+15 kHz). The age of the studied women ranged from 31 to 73 years: 30-40 years-16 (13.5%); 41-50 years old - 32 (27.2%), 51-60 years old - 41 (34.7%), 61-70 years old -25 (21.2%), 71 and older - 4 (3.4%). 83 (70.3%) patients were young and of working age. According to the histological picture: infiltrative form - 59 (50%), invasive - 33 (28%), ductal - 18 (15.3%), adenocarcinoma - 7 (5.9%) and Paget's cancer 1 (0.8%). Depending on the molecular genetic features of breast cancer, Luminal subtype A was found in 8- (6.9%), luminal subtype B in 20- (16.5%), HER2-positive subtype in 33- (28%) and Triple negative » subtype in 27 (23%) and in 30 patients (25.6%) molecular genetic study was not performed.For the diagnosis of breast cancer, a complex of laboratory and instrumental studies was used. General clinical blood tests, detailed immunograms were performed in almost all patients at admission and during treatment according to the standard method. Studies were carried out: biochemical parameters of blood, coagulograms, blood electrolytes. To assess the functional state of the liver, pancreas, kidneys, patients underwent biochemical studies and paid attention to the levels of bilirubin, total protein, sugar, transaminase, amylase, etc. diagnostics, monitoring, control of treatment effectiveness). MSCT, chest MRI and mammogram data were taken into account in preoperative preparation. With pathological changes, corrective therapy was carried out, the effectiveness of which was evaluated by repeating the study. Non-invasive intervention in patients with breast cancer was performed in different combinations. Table 1 shows combinations of the optimized approach.
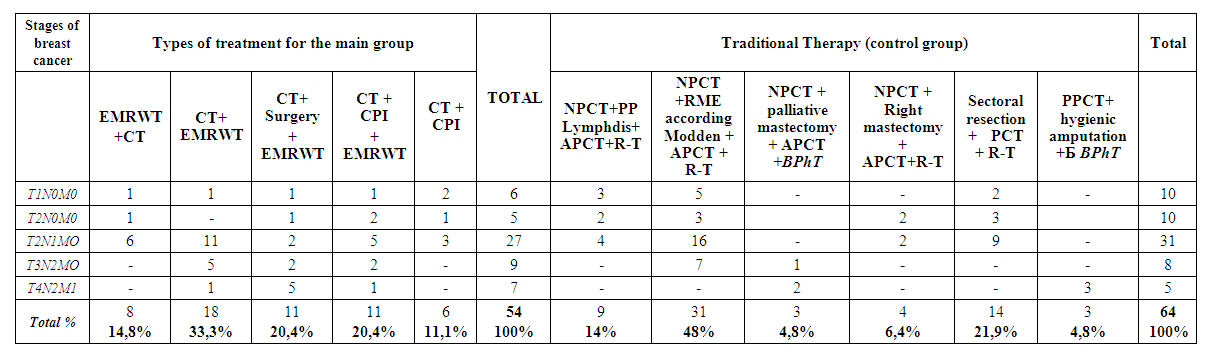 | Table 1. Combinations of the Optimized Approach |
|
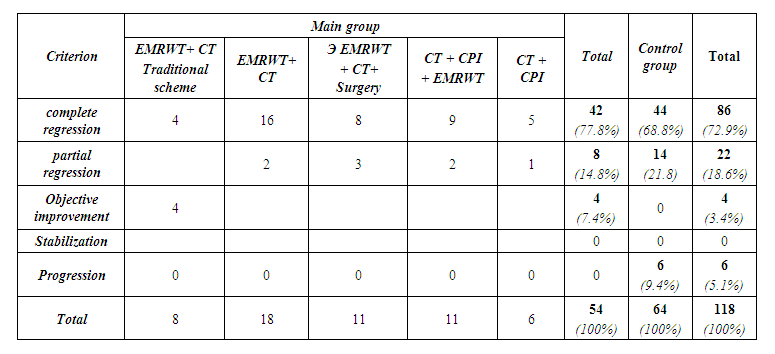 | Table 3. Evaluation of the effectiveness of breast cancer treatment |
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML