Ulmasova Shakhnoza Rakhmatjanovna 1, Rakhimov Shukhrat Malikovich 1, Sobirova Guzal Naimovna 2
1Central Military Hospital, Tashkent, Uzbekistan
2Republican Specialized Scientific and Practical Medical Center for Therapy and Medical Rehabilitation, Tashkent, Uzbekistan
Correspondence to: Ulmasova Shakhnoza Rakhmatjanovna , Central Military Hospital, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2020 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
Evaluation of all treatment methods and pharmacotherapy of non-alcoholic fatty liver disease (NAFLD). The development of obesity and the rapid spread of diabetes have led to the fact that NAFLD is now the most famous liver disease. The main non-pharmacological treatments for NAFLD are a low-calorie diet with a sufficient content of polyunsaturated fats and dosed physical activity in order to reduce BMI. Among the drugs, the leading positions are occupied by insulin sensitizers, antioxidants (vitamin E) and polyunsaturated fatty acid preparations (Essential Forte N). Several new drugs for the treatment of NAFLD are in the process of clinical trials. Due to the high increase in the frequency of NAFLD, new methods for diagnosing and treating this disease are being actively developed.
Keywords:
Non-alcoholic fatty liver disease, Non-alcoholic steatohepatitis, Treatment, Metabolic syndrome
Cite this paper: Ulmasova Shakhnoza Rakhmatjanovna , Rakhimov Shukhrat Malikovich , Sobirova Guzal Naimovna , Modern Principles of Management of Patients with Non-Alcoholic Fatty Liver Disease, American Journal of Medicine and Medical Sciences, Vol. 10 No. 9, 2020, pp. 639-642. doi: 10.5923/j.ajmms.20201009.02.
Article Outline
The concept of non-alcoholic fatty liver disease (NAFLD) is today one of the most insidious liver diseases. The etiology of this is the epidemic of obesity. According to the WHO, the prevalence rate of obesity in the world has doubled since 1980. The total number of obese patients in the world is more than 600 million people. The global prevalence of diabetes over the same period increased from 4.7% to 8.5% in 2014 [1]. This led to the fact that NAFLD is found in 25% of the world's inhabitants [2].In recent years, it has been proven that NAFLD can be characterized by an adverse course. In the United States, non-alcoholic steatohepatitis (NASH) came in third among the causes of liver cirrhosis [3] and in second place among the causes of liver transplantation [4]. In this regard, great attention is paid to the management of patients with NAFLD. The purpose of the review is to analyze not only existing treatment methods, but also the immediate prospects for pharmacotherapy of this pathology.NAFLD is defined today as a disease in which there is an accumulation of fat in the liver (more than 5% of the mass of hepatocytes) in the absence of secondary etiological causes of this pathology. There are 3 main phenotypes of the disease: steatosis, NASH, cirrhosis [5]. An important point in the diagnosis of NAFLD is the limitation of alcohol consumption to 20–30 g of ethanol per day [6]. It is important to note that there is no “gold standard” for non-invasive diagnosis of NAFLD [1], that is, it remains the diagnosis of exclusion. For the diagnosis of NAFLD, it is necessary to conduct differential diagnostics with alcoholic liver disease, hepatitis C, Wilson – Konovalov’s disease, hemochromatosis, autoimmune liver diseases, and drug hepatitis [7].Screening is important among patients with risk factors, which include the presence of metabolic syndrome, obesity, type 2 diabetes and / or abnormal liver tests in a blood test [8]. In three modern international NAFLD guidelines, it is postulated that for the diagnosis of this disease, a comprehensive assessment of the results of a biochemical analysis of blood and data obtained using radiation methods of liver imaging (the leading ones are transient elastography and magnetic resonance spectroscopy), as well as necessary, liver biopsies [1,6,9,10]. The diagnostic algorithm for NAFLD is adequately presented in the European guideline for this pathology (Fig. 1) [5].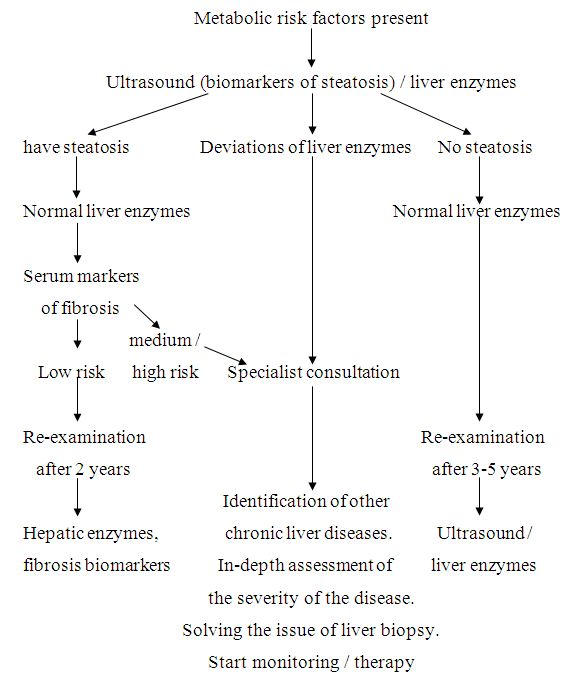 | Figure 1. Algorithm for the diagnosis of non-alcoholic fatty liver disease [4] |
Today, it is customary to include NASH, a combination with liver fibrosis and / or the presence of several components of the metabolic syndrome, as indications for drug treatment of NAFLD [5,6,7,8]. Modern European [6], American [9], Asian [10] and Russian [11] guidelines contain recommendations for lifestyle modification, increased physical activity and weight loss in patients with NAFLD as first-line therapy. Recommendations include a reduction in the calorie content of the diet (a decrease of 500–1000 kcal per day compared to the usual regimen), an increase in foods containing ω3 polyunsaturated fatty acids (PUFAs) (seafood), a decrease in refined carbohydrates, and systematic physical exercises of medium intensity and in general, an increase in physical activity, a decrease in BMI in case of its redundancy by 7–10% of the initial values.M. Romero-Gomez et al. note that lifestyle changes and a decrease in BMI are a difficult task [12]. It has been repeatedly established that weight loss as a result of dietary changes is maximum for 6 months, after which its recovery often occurs [13]. In this regard, it is important not only to solve particular problems of changing the diet, increasing physical activity and lowering BMI, but generally changing the patient’s behavioral stereotypes.Great importance is now given to the latest treatment methods for NAFLD. A number of drugs are currently in phase 3 clinical trials. Obeticholic acid (OA) is a semisynthetic derivative of chenodeoxycholic acid and is an agonist of farnesoid receptors. OA is involved in the regulation of glucose in the liver and lipid metabolism, changes the endogenous production of bile acids, limiting the enzymes that convert cholesterol to bile acids. In the placebo-controlled trail FLINT, 283 patients with a morphologically confirmed diagnosis of NASH without cirrhosis were randomized into 2 equal groups, one of which received OA at a dose of 25 mg per day, the second received placebo for 70 weeks. At the end of the study, positive morphological dynamics were observed in 45% of those receiving ОA and only in 21% of patients in the placebo group (p = 0.0002). In 23% of those who took OA, itching and adverse changes in the lipid profile in the blood were noted [14].In another study, which is currently not completed, 2000 patients with morphologically confirmed NASH and stage 1-3 fibrosis according to METAVIR were randomized into 3 equal groups: treated with OA at a dose of 25 mg per day, OA at a dose of 10 mg per day, and placebo. In the next 1-2 years, the results of work are expected to be obtained [15].Elafibranor, an agonist of α / δ receptors activated by the peroxisome proliferator (PPARs), regulates metabolic homeostasis, inflammation, growth and differentiation of cells by affecting the transport and β-oxidation of fatty acids. In a placebo-controlled study, 276 patients with morphologically confirmed NASH without cirrhosis were randomized into 3 groups: receiving 80 mg of elafibranor per day, 120 mg of the drug per day and placebo for 52 weeks. After the introduction of strict definitions, a morphological response was achieved in 13% of participants in group 1, in 19% in group 2, and in 12% who received placebo (p = 0.045 to compare the second group and placebo) [11]. Currently, a study is being conducted of 2,000 patients with NASH randomized to groups receiving elafibranor at a dose of 120 mg per day and placebo [3].Cenicriviroc, an antagonist of the chemokine receptor CCR2 / CCR5, plays a key role in the development of inflammation and liver fibrosis, as well as selonsertib, an inhibitor of kinase 1 (ASK-1), which indicates the severity of apoptosis, oxidative stress and fibrosis, and other drugs [5,6,11].Current international recommendations show the feasibility of using vitamin E, insulin sensitizers, statins (in the presence of hyperlipidemia) for the treatment of NAFLD [6,8,9].The authors of the recommendations of the Russian Society for the Study of the Liver and the Russian Gastroenterological Association consider it possible from drugs with cytoprotective activity to use essential phospholipids, urodeoxycholic acid, ademethionine, glycerrhizic acid and preparations containing silibinin for the treatment of NAFLD [12]. It is necessary to conduct continuous therapy of pathology for a long time, at least 1 year. Z.M. Younossi et al. explain this approach by the close association of NAFLD with metabolic syndrome, which in most cases requires long-term therapy [13].The prevention of gastroenterological diseases is very important [14]. The primary prevention of NAFLD is to prevent the action and prevent the development of risk factors - obesity, diabetes and metabolic syndrome.PUFA preparations. Particular importance in the treatment of NAFLD is given to PUFA preparations [15]. The official guideline guidelines of the European Association for the Study of the Liver, published in the Journal of Hepatology in 2016, explain that PUFA preparations lower lipids in the blood and liver and can be used to treat NAFLD [4,6]. Position 33 of the guideline of the American Association for the Study of the Liver indicates that PUFA preparations can be used to treat hypertriglyceridemia in patients with NAFLD [7,9]. Regulation 7.11 of the 2018 Asia-Pacific Recommendations states that PUFA preparations lower liver fat and blood lipids in patients with NAFLD [6,10].The influence of current PUFA medications is that long-chain ω3-PUFAs have a very strong effect on the biologically active metabolites involved in inflammation and on changes in the activity of nuclear transcription agents involved in inflammatory processes and the metabolism of lipids in the liver, such as PPARs, sterol regulatory element-binding protein 1c (SREBP-1c) and carbohydrate-reacting element-binding protein (ChREBP) [1,4,15].One of the especially studied PUFA preparations is Essentiale forte N. It can be prescribed both parenterally and orally. Parenterally Essential Forte N is prescribed in a dose of 1–4 ampoules (5–20 ml) per day for 10–30 days. At the same time or after a course of intravenous administration, it is recommended to start taking the drug inside at a dose of 6 capsules per day in 3 divided doses. The duration of therapy and the multiplicity of courses is individually, their acceptable duration is 3 months.ConclusionsDue to the high increase in the frequency of non-alcoholic fatty liver disease (NAFLD), new methods for diagnosing and treating this pathology are now being dynamically developed. Non-drug therapy of NAFLD includes a low-calorie diet with enough polyunsaturated fats and dosed physical activity to reduce weight. Among drug therapy, insulin sensitizers, antioxidants (vitamin E) and polyunsaturated fatty acid preparations (Essentiale forte N) occupy leading positions.
References
| [1] | V.V. Tsukanov, A.V. Vasyutin, Yu.L. Thin. Modern principles of management of patients with non-alcoholic fatty liver disease. Gastroenterology 2019, No. 3 (158), 11-14. |
| [2] | Younossi Z., Tacke F., Arrese M., Sharma B.C., Mostafa I., Bugianesi E. et al. Global perspectives on non-alcoholic fatty liver disease and non-alcoholic steatohepatitis. Hepatology. 2018. DOI: 10.1002/ hep.30251. URL: https://aasldpubs.onlinelibrary.wiley.com/ doi/10.1002/hep.30251 (date of the application — 15.12.2018). |
| [3] | Konerman M.A., Jones J.C., Harrison S.A. Pharmacotherapy for NASH: current and emerging. J. Hepatol. 2018; 68(2): 362–75. DOI: 10.1016/j.jhep.2017.10.015. |
| [4] | Sobirova G.N., Khamroeva D.M., Karimov M.M., Alyavi A.L., Adilov B.Sh. GG Genotype of Pnpla3 Rs738409 Polymorphism Associated with Nash in Patients of Uzbek Nationality. Journal of Gastroenterology & Digestive Systems. 2018; V2: I 3:1-6. |
| [5] | European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO); Marchesini G., Day C.P., Dufour J.F. et al. EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J. Hepatol. 2016; 64(6): 1388–402. DOI: 10.1016/j.jhep.2015.11.004. |
| [6] | Rotman Y., Sanyal A.J. Current and upcoming pharmacotherapy for non-alcoholic fatty liver disease. Gut. 2017; 66(1): 180–90. DOI: 10.1136/gutjnl-2016-312431. |
| [7] | Leoni S., Tovoli F., Napoli L., Serio I., Ferri S., Bolondi L. Current guidelines for the management of non-alcoholic fatty liver disease: a systematic review with comparative analysis. World J. Gastroenterol. 2018; 24(30): 3361–73. DOI: 10.3748/wjg.v24.i30.3361. |
| [8] | Chalasani N., Younossi Z., Lavine J.E., Charlton M., Cusi K., Rinella M. et al. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018; 67(1): 328–57. DOI: 10.1002/hep.29367. |
| [9] | Chitturi S., Wong V.W., Chan W.K., Wong G.L., Wong S.K., Sollano J. et al. The Asia-Pacific Working Party on non-alcoholic fatty liver disease guidelines 2017-Part 2: management and special groups. J. Gastroenterol. Hepatol. 2018; 33(1): 86–98. DOI: 10.1111/jgh.13856. |
| [10] | Romero-Gómez M., Zelber-Sagi S., Trenell M. Treatment of NAFLD with diet, physical activity and exercise. J. Hepatol. 2017; 67(4): 829–46. DOI: 10.1016/j.jhep.2017.05.016. |
| [11] | Ratziu V., Sanyal A.J., MacConell L., Shringarpure R., Marmon T., Shapiro D. et al. Regenerate: a Phase 3, double-blind, randomized, placebo-controlled multicenter study of obeticholic acid therapy for nonalcoholic steatohepatitis. J. Hepatol. 2016; 64(2 suppl.): S294–5. DOI: 10.1016/S0168-8278(16)00372-X. |
| [12] | Ratziu V., Francque S., Harrison S., Anstee Q.M., Bedossa P., Brozek J. et al. Improvement in NASH histological activity highly correlates with fibrosis regression. Hepatology. 2016; 64(6): 1118A–40A. DOI: 10.1002/hep.28909. |
| [13] | Townsend S.A., Newsome P.N. Review article: new treatments in non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2017; 46(5): 494–507. DOI: 10.1111/apt.14210. |
| [14] | Jump D.B., Lytle K.A., Depner C.M., Tripathy S. Omega-3 polyunsaturated fatty acids as a treatment strategy for nonalcoholic fatty liver disease. Pharmacol. Ther. 2018; 181: 108–25. DOI: 10.1016/j.pharmthera.2017.07.007. |
| [15] | Scorletti E., Byrne C.D. Omega-3 fatty acids and non-alcoholic fatty liver disease: Evidence of efficacy and mechanism of action. Mol. Aspects Med. 2018; 64: 135–46. DOI: 10.1016/j.mam.2018.03.001. |




 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML