-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2013; 3(4): 61-67
doi:10.5923/j.ajmms.20130304.02
Red Blood Cell Alloimmunization among Sudanese Homozygous Sickle Cell Disease Patients
Mohammed Abbas1, Ahmed Bolad2, Nasreldin Jiefri3, Adil Mergani4
1College of Applied Medical Sciences, Medical Laboratory Department, Salman bin Abdulaziz University, Riyadh-Al Kharj, 11942, Saudi Arabia (KSA)
2Department of Microbiology, Faculty of Medical Laboratory Sciences, Al Neelain University, Khartoum, Sudan
3Hematology department, Faculty of Medicine , Al Neelain University , Khartoum, Sudan
4Department of Molecular Biology and Biotechnology, College of Applied Medical Sciences, Medical Laboratory Department, Taif University, –Tariba, Taif, Saudi Arabia (KSA)
Correspondence to: Mohammed Abbas, College of Applied Medical Sciences, Medical Laboratory Department, Salman bin Abdulaziz University, Riyadh-Al Kharj, 11942, Saudi Arabia (KSA).
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
Background: Transfusion of RBCs is a major therapeutic option for anemia in homozygous SCD. Numerous studies conducted in the world to determine the frequency of alloimmunization in SCD patients, but there are no studies conducted in Sudan in open literature, to our knowledge. The objective of this study was to assess the incidence of RBC alloimmunization in SCD HbSS patients in Sudan. Methods: This study was conducted in Omdurman between August 2011 - Aril 2012, in 100 SCD HbSS patients of age between 6 month –17 years, who had received at least 2 transfusions with units of ABO and D matched RBCs, the alloimmunization was determined. Results: The mean No of blood units transfused per patient was 6.73 (SD, 4.461). 55% of the patients had been transfused from non relative donors, while 45% from relative donors. Only four patients was found to have antibodies 4% were detected among the 100 SCD (HbSS) patients who received transfusion.Conclusions: Although limited precompatibility testing is done in blood banking services in Sudanese hospital, low incidence of red cell alloimmunization (4%) was observed in this study. We recommend that to improve the blood bank services and polices to overcome the risk of alloimmunization.
Keywords: SCD, Transfusion, Alloimmunization
Cite this paper: Mohammed Abbas, Ahmed Bolad, Nasreldin Jiefri, Adil Mergani, Red Blood Cell Alloimmunization among Sudanese Homozygous Sickle Cell Disease Patients, American Journal of Medicine and Medical Sciences, Vol. 3 No. 4, 2013, pp. 61-67. doi: 10.5923/j.ajmms.20130304.02.
Article Outline
1. Introduction
- Sickle cell disease (SCD) is an inherited disorder of hemoglobin (Hb), it is the most prevalent genetic disease in African region[1] with high mortality rate at age one to five years.[2] Since description of the first case of SCD in Sudan at 1926.[3] There was and still increasing research activities in this field. The highest prevalence of SCD in Sudanese is among the population from the western Sudan.[4-6]SCD is caused by an autosomal recessive inherited of an abnormal beta globin gene (sickle cell gene) which leads to substitution of thymine by adenine in glutamic acid which in turn results in the substitution of valine to glutamic acid at the sixth position on the beta globin chain. Hemoglobin S (HbS), is the defective Hb that produced as a result of this defect, is a tetramer (alpha2/beta2) that is poorly soluble and polymerizes when deoxygenated.[7] The two characteristic features in the pathophysiology of SCD is the chronic hemolytic anemia and the vaso-occlusion.[8]Transfusion of red blood cells is a major therapeutic option in SCD. It improve the oxygen-carrying capacity by increasing the total Hb level, decrease blood viscosity and increasing oxygen saturation by diluting the concentration of HbS; and suppressing endogenous production of sickle RBCs by increasing tissue oxygenation[9-11].The repeated or chronic RBC transfusions and the exposure to different red cells antigen are often complicated by RBC alloimmunization, which results in difficulties in the selection of compatible blood, acute or delayed transfusion reactions.[12] A considerable number of studies have dealt with Alloimmunization in SCD, the range of RBCs alloimmunization has been reported to be in the range of 2.6% to 76%.[13-29,50].The precompatibility testing procedures in the majority of the Sudanese hospital is limited to perform only ABO Rh grouping, and cross-matching by using saline and anti human globulin techniques by tube method. Although some major hospitals start to use the column technique in across-matching procedures, but they still use limited tests. There are no antibody screening , limited phenotyping, or even further antibody identification were done as a routine work far as we know. Despite of the numerous studies conducted in the world to determine the frequency of red cells alloimmunization in SCD patients[13-29,50], unfortunately there are no enough studies conducted in Sudan in open literature, to our knowledge. The objective of this study was to assess the incidence of RBC alloimmunization in SCD patients in Sudan.
2. Material and Method
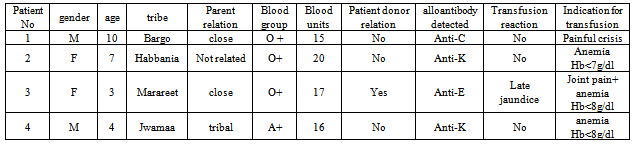
- After appropriate ethical approval prior the commencement of the study, a cross-sectional study was carried out, random samples were analyzed. 5 ml of venous blood in K2 ethylenediaminetetraacetic acid (EDTA) for each sample. Questionnaires were used to collect demographic and clinical data include sex, age, tribe, number and indication for transfusion, transfusion reaction, and malarial attack . A total of hundred (100) homozygous sickle cell disease (HbSS) patients of age between 6 month –17 years, both males (57%) and females (43%) belonging to different tribes who had received at least 2 transfusions with units of ABO and D matched RBCs were selected from Albuluk pediatric teaching hospital and Omdurman pediatric hospital between August 2011 and Aril 2012. The number of blood transfusion ranged from 2 to 20 units per patient, the total number of transfusion to all patients was 673 units.
2.1. Laboratory Investigation
- After ABO and Rh blood grouping by the standard tube method, the following tests were done for every sample: Antibody screening. The collected plasma were tested for the presence of alloantibodies using a 3-cell panel with homozygous expression of the antigens (Panoscreen I,II and III, Immucor, Inc, USA) by using saline phase and indirect human antiglobulin test (indirect Coombs) in low-ionic-strength solution (LISS) by the tube method.Antibody identification. Antibody specificity was determined with a standard panel of red cells reacting to known antigens using the same technique in the positive antibody screening (Panocell -10, Immucor, Inc, USA).Patients were considered to be alloimmunized if antibodies to one or more RBC antigens were identified.
2.2. Data Analysis
- Statistical software packages (Excel 5.0, Microsoft, Redmond, WA; and Statistical Package for the Social Sciences 20.0, SPSS, Inc., Chicago, IL) were used for data management and analysis, respectively.
3. Results
- A total of 100 patients with SCD (HbSS) formed the study population. Fifty seven patients were males (57%), and forty three were female (43%) of age between 6 month – 17 years. (67%) of patients ages fall between 2 and 8 years, (25%) between 9 and 17 years and the lower proportion of patients aged less than two years (8%). Over (97%) of the patients involved in this study belong to tribes from the West of Sudan. Miserria, Marareet, Rizegat and Bargo constituted the largest proportions of the tribes, (28%, 22%, 12% and 8% respectively). Other tribes represents low percentage include: Tama, Habbania, Jawamaa, Zagawa, Masalma, Slihab, Bnehalba and Kwahla. (59%) of the patients parents are close relatives, (26%) are tribal, (5%) are far relation, while (10%) are not relatives. Only (2%) of the patients has had a history of malarial attack.Figure 1 shows the rank order of blood group among subjects: O Rh + (n = 45 patients), A Rh + (n = 22), B Rh + (n = 20), AB Rh + (n = 8) , B Rh – (n = 2) ,A Rh – (n = 2) ,O Rh – (n = 1).
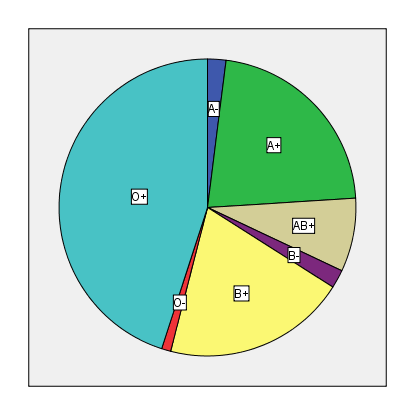 | Figure 1. The frequency of ABO RH group among patients |
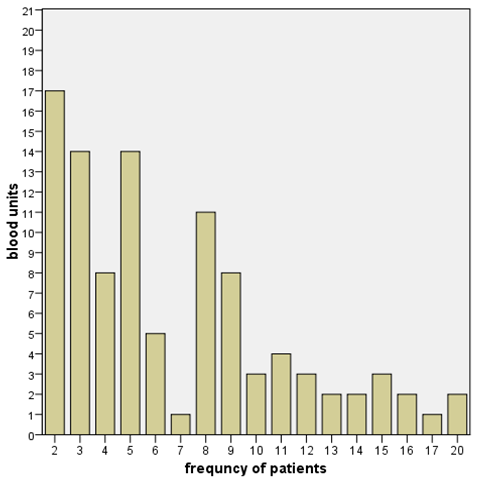 | Figure 2. The frequency of blood units taken by the patients |
4. Discussion
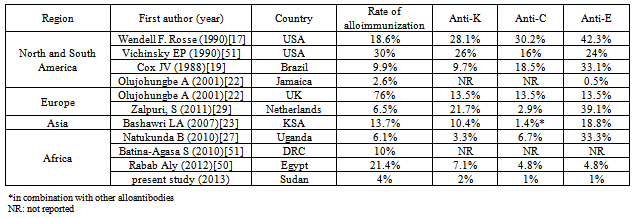
- The alloimmunization is considered one of the major blood transfusion complications in SCD patients who received regular transfusion therapy, unfortunately, there are no published studies to determine the frequency of alloimmunization in SCD Sudanese patients as far as we know. In this study we try to establish the incidence of the alloimmunization in this category in Sudan.The sickle cell anaemia (SCA) is the one of the major types of anaemia especially in Kordofan and Darfur states[4], western Sudan. Over (97%) of the patients involved in this study belong to tribes from that area, Elderdery, A[30] also noticed the high frequency of homozygous HbSS in his study among western Sudan tribes. Misseria which is a sub group of Baggara tribe, showed the highest percentage of the entire study group of patients in this study (28%). This tribe showed the prevalence of SCD to be (30%), (16%) among immigrants from the province of Blue Nile[31].Blood group O (46%) was predominant among patients blood groups, this finding in agreement with three previous studies performed among Sudanese (52.7%) ,(51.5%) and (43.7% )[32-34], respectively. The result also consistent with other studies performed in African countries, shown an increase in group O percentage : Nairobi- Kenya (69%)[35], Nigeria (48.9%)[36]. The predominance of group O also observed in Western countries, in UK blood group O constitute (46.7%)[37], (47%) and (46%) of Caucasians and blacks in USA were group O respectively[38]. The increase percentage of group O in this study may be related to the low incidence of malarial attack (2%), which in agreement with some findings suggested the diverse relations between group O and different types of malaria[39-42]. Rowe JA et al; observed that group O was associated with a (66%) reduction in the odds of developing severe malaria compared with the non-O blood groups[43]. Furthermore, there is some convincing evidence to suggest that patients with SCD are protected from malaria[44-48]. The low prevalence of malaria observed in this study is matched with that reported by Makani J, et al.[49] the prevalence of malaria parasitemia was (0.72%) of 1808 SCA patients at the outpatient clinics and (3.04%) of 497 SCA patients during hospitalization.
|
|
5. Conclusions
- The present study is the first study to determine the incidence of alloimmunization among homozygous SCD Sudanese patients. Although the limited tests were performed as a routine precompatibility testing in blood banks in Sudan hospitals, but fortunately low incidence of alloimmunization was detected (4%) which can be compared with other studies with low incidence of alloimmunization [22,27, and 29]. We recommend that to improve the blood bank services and polices in the way of RBCs compatibility tests, limited phenotype matching in Kell and Rhesus blood group systems specially in K, C, and E antigens, and improve the laboratory techniques to involve the alloantibody detection and identification. After making these improvement the alloantibodies formation in chronically transfused patients can easily be prevented.
ACKNOWLEDGEMENTS
- The authors thank the staff of the haematology department at Albuluk pediatric teaching hospital and Omdurman pediatric hospital , Sudan, specially: dr. Areej Aljizoli and Ms. Israa Abdul Mahmoud for the help in recruiting patients and taking of blood samples and the Research Center in the medicine faculty in Alneelain University , Sudan, for the storage of samples during the period of the study. They also extend their appreciation to the dr. Abdelmoneim A. Sulieman and dr. Omar H. Aldirdeery for they help and advices.
Conflict of Interest
- The authors certify that they have no affiliation with or financial involvement in any organization or entity with a direct financial interest in the subject matter or materials discussed in this manuscript.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML