| [1] | A. G. Kaningini et al., “Effect of Optimized Precursor Concentration, Temperature, and Doping on Optical Properties of ZnO Nanoparticles Synthesized via a Green Route Using Bush Tea (Athrixia phylicoides DC.) Leaf Extracts,” ACS Omega, vol. 7, no. 36, pp. 31658–31666, Sep. 2022, doi: 10.1021/acsomega.2c00530. |
| [2] | H. M. Saleh and A. I. Hassan, “Synthesis and Characterization of Nanomaterials for Application in Cost-Effective Electrochemical Devices,” Sustainability, vol. 15, no. 14, p. 10891, Jul. 2023, doi: 10.3390/su151410891. |
| [3] | S. Ying et al., “Green synthesis of nanoparticles: Current developments and limitations,” Environ. Technol. Innov., vol. 26, p. 102336, May 2022, doi: 10.1016/j.eti.2022.102336. |
| [4] | J. Jalab, W. Abdelwahed, A. Kitaz, and R. Al-Kayali, “Green synthesis of silver nanoparticles using aqueous extract of Acacia cyanophylla and its antibacterial activity,” Heliyon, vol. 7, no. 9, p. e08033, Sep. 2021, doi: 10.1016/j.heliyon.2021.e08033. |
| [5] | N. C. Nkosi, A. K. Basson, Z. G. Ntombela, N. G. Dlamini, and R. V. S. R. Pullabhotla, “Green synthesis and characterization of iron nanoparticles synthesized from bioflocculant for wastewater treatment: A review,” Biotechnol. Notes, vol. 6, pp. 10–31, 2025, doi: 10.1016/j.biotno.2024.12.001. |
| [6] | P. Kumari, A. Srivastava, R. K. Sharma, D. Sharma, and S. K. Srivastava, “Zinc Oxide: A Fascinating Material for Photovoltaic Applications,” in Nanomaterials for Innovative Energy Systems and Devices, Z. H. Khan, Ed., Materials Horizons: From Nature to Nanomaterials, Singapore: Springer Nature Singapore, 2022, pp. 173–241. doi: 10.1007/978-981-19-0553-7_6. |
| [7] | R. Saxena et al., “A review on green synthesis of nanoparticles toward sustainable environment,” Sustain. Chem. Clim. Action, vol. 6, p. 100071, Jun. 2025, doi: 10.1016/j.scca.2025.100071. |
| [8] | J. Pan, H. Qian, Y. Sun, Y. Miao, J. Zhang, and Y. Li, “Microbially synthesized nanomaterials: Advances and applications in biomedicine,” Precis. Med. Eng., vol. 2, no. 1, p. 100019, Mar. 2025, doi: 10.1016/j.preme.2025.100019. |
| [9] | M. M. Abady, D. M. Mohammed, T. N. Soliman, R. A. Shalaby, and F. A. Sakr, “Sustainable synthesis of nanomaterials using different renewable sources,” Bull. Natl. Res. Cent., vol. 49, no. 1, p. 24, Apr. 2025, doi: 10.1186/s42269-025-01316-4. |
| [10] | D. C. Bouttier-Figueroa, J. M. Cortez-Valadez, M. Flores-Acosta, and R. E. Robles-Zepeda, “Synthesis of Metallic Nanoparticles Using Plant’s Natural Extracts: Synthesis Mechanisms and Applications,” Biotecnia, vol. 25, no. 3, pp. 125–139, Oct. 2023, doi: 10.18633/biotecnia.v25i3.1916. |
| [11] | M. Y. Al-darwesh, S. S. Ibrahim, and M. A. Mohammed, “A review on plant extract mediated green synthesis of zinc oxide nanoparticles and their biomedical applications,” Results Chem., vol. 7, p. 101368, Jan. 2024, doi: 10.1016/j.rechem.2024.101368. |
| [12] | E. K. Tirop, N. N. Maina, J. K. Maina, P. K. Njenga, E. Magiri, and V. W. Ngumi, “Evaluation of Toxicity of Strychnos henningsii (Gilg) (Loganiaceae) Leaves and Root Aqueous Extracts in Mice,” Eur. J. Med. Plants, vol. 25, no. 1, pp. 1–11, Sep. 2018, doi: 10.9734/EJMP/2018/43613. |
| [13] | C. Zhu and X. Wang, “Nanomaterial ZnO Synthesis and Its Photocatalytic Applications: A Review,” Nanomaterials, vol. 15, no. 9, p. 682, Apr. 2025, doi: 10.3390/nano15090682. |
| [14] | G. Philippe, L. Angenot, M. Tits, and M. Frédérich, “About the toxicity of some Strychnos species and their alkaloids,” Toxicon, vol. 44, no. 4, pp. 405–416, Sep. 2004, doi: 10.1016/j.toxicon.2004.05.006. |
| [15] | P. B. Mallikharjuna, L. N. Rajanna, Y. N. Seetharam, and G. K. Sharanabasappa, “Phytochemical Studies of Strychnos potatorum L.f. – A Medicinal Plant,” J. Chem., vol. 4, no. 4, pp. 510–518, Jan. 2007, doi: 10.1155/2007/687859. |
| [16] | A. N. Abdulqodus et al., “Green synthesis of ZnO nanoparticles: effect of pH on morphology and photocatalytic degradation efficiency,” Appl. Phys. A, vol. 131, no. 9, p. 720, Sep. 2025, doi: 10.1007/s00339-025-08874-4. |
| [17] | S. Kumar, V. Singh, and A. Tanwar, “Structural, morphological, optical and photocatalytic properties of Ag-doped ZnO nanoparticles,” J. Mater. Sci. Mater. Electron., vol. 27, no. 2, pp. 2166–2173, Feb. 2016, doi: 10.1007/s10854-015-4227-1. |
| [18] | S. Raha and Md. Ahmaruzzaman, “ZnO nanostructured materials and their potential applications: progress, challenges and perspectives,” Nanoscale Adv., vol. 4, no. 8, pp. 1868–1925, 2022, doi: 10.1039/D1NA00880C. |
| [19] | A. S. Abdelbaky et al., “Green approach for the synthesis of ZnO nanoparticles using Cymbopogon citratus aqueous leaf extract: characterization and evaluation of their biological activities,” Chem. Biol. Technol. Agric., vol. 10, no. 1, p. 63, Jul. 2023, doi: 10.1186/s40538-023-00432-5. |
| [20] | E. Tilahun, Y. Adimasu, and Y. Dessie, “Biosynthesis and Optimization of ZnO Nanoparticles Using Ocimum lamifolium Leaf Extract for Electrochemical Sensor and Antibacterial Activity,” ACS Omega, vol. 8, no. 30, pp. 27344–27354, Aug. 2023, doi: 10.1021/acsomega.3c02709. |
| [21] | M. Thiam et al., “Green Synthesis of ZnO Nanoparticles Using Licania tomentosa Benth (Oiti) Leaf Extract: Characterization and Applications for the Photocatalytic Degradation of Crystal Violet Dye,” Processes, vol. 13, no. 3, p. 880, Mar. 2025, doi: 10.3390/pr13030880. |
| [22] | T. Şimşek, A. Ceylan, G. Ş. Aşkin, and Ş. Özcan, “Band Gap Engineering of ZnO Nanocrystallites Prepared via Ball-Milling,” Politek. Derg., vol. 25, no. 1, pp. 89–94, Mar. 2022, doi: 10.2339/politeknik.647702. |
| [23] | D. Zagorac et al., “Band Gap Engineering of Newly Discovered ZnO/ZnS Polytypic Nanomaterials,” Nanomaterials, vol. 12, no. 9, p. 1595, May 2022, doi: 10.3390/nano12091595. |
| [24] | F. B. Dejene, “Characterization of low-temperature-grown ZnO nanoparticles: The effect of temperature on growth,” J. Phys. Commun., vol. 6, no. 7, p. 075011, Jul. 2022, doi: 10.1088/2399-6528/ac8049. |
| [25] | D. Mutukwa, R. Taziwa, and L. E. Khotseng, “A Review of the Green Synthesis of ZnO Nanoparticles Utilising Southern African Indigenous Medicinal Plants,” Nanomaterials, vol. 12, no. 19, p. 3456, Oct. 2022, doi: 10.3390/nano12193456. |
| [26] | H. Heinz et al., “Nanoparticle decoration with surfactants: Molecular interactions, assembly, and applications,” Surf. Sci. Rep., vol. 72, no. 1, pp. 1–58, Feb. 2017, doi: 10.1016/j.surfrep.2017.02.001. |
| [27] | B. Ding et al., “Tuning oxygen vacancy photoluminescence in monoclinic Y₂WO₆ by selectively occupying yttrium sites using lanthanum,” Sci. Rep., vol. 5, no. 1, p. 9443, Mar. 2015, doi: 10.1038/srep09443. |
| [28] | C. M. Pelicano, E. Magdaluyo, and A. Ishizumi, “Temperature Dependence of Structural and Optical Properties of ZnO Nanoparticles Formed by Simple Precipitation Method,” MATEC Web Conf., vol. 43, p. 02001, 2016, doi: 10.1051/matecconf/20164302001. |
| [29] | A. Jayachandran, A. T. R., and A. S. Nair, “Green synthesis and characterization of zinc oxide nanoparticles using Cayratia pedata leaf extract,” Biochem. Biophys. Rep., vol. 26, p. 100995, Jul. 2021, doi: 10.1016/j.bbrep.2021.100995. |
| [30] | S. P. Prakoso and R. Saleh, “Synthesis and Spectroscopic Characterization of Undoped Nanocrystalline ZnO Particles Prepared by Co-Precipitation,” Mater. Sci. Appl., vol. 3, no. 8, pp. 530–537, 2012, doi: 10.4236/msa.2012.38075. |
| [31] | G. Baranović and S. Šegota, “Infrared spectroscopy of flavones and flavonols. Reexamination of the hydroxyl and carbonyl vibrations in relation to the interactions of flavonoids with membrane lipids,” Spectrochim. Acta A Mol. Biomol. Spectrosc., vol. 192, pp. 473–486, Mar. 2018, doi: 10.1016/j.saa.2017.11.057. |
| [32] | M. Sowmiya, K. Selvam, and M. S. Shivakumar, “Sustainable fabrication of zinc oxide nanoparticles using Secamone emetica leaf extract: Biological and environmental applications,” J. Nat. Pestic. Res., vol. 12, p. 100122, Jun. 2025, doi: 10.1016/j.napere.2025.100122. |
| [33] | D. R. Jaishi et al., “Plant-mediated synthesis of zinc oxide (ZnO) nanoparticles using Alnus nepalensis D. Don for biological applications,” Heliyon, vol. 10, no. 20, p. e39255, Oct. 2024, doi: 10.1016/j.heliyon.2024.e39255. |
| [34] | L. H. Qian, S. C. Wang, Y. H. Zhao, and K. Lu, “Microstrain effect on thermal properties of nanocrystalline Cu,” Acta Mater., vol. 50, no. 13, pp. 3425–3434, Aug. 2002, doi: 10.1016/S1359-6454(02)00155-6. |
| [35] | M. Mahajan et al., “Green synthesis of ZnO nanoparticles using Justicia adhatoda for photocatalytic degradation of malachite green and reduction of 4-nitrophenol,” RSC Adv., vol. 15, no. 4, pp. 2958–2980, 2025, doi: 10.1039/D4RA08632E. |
| [36] | M. Abdel-Rahman, H. Ibrahim, M. Y. Mostafa, M. Abdel-Rahman, M. Ebied, and E. A. Badawi, “The characterization of ZnO nanoparticles by applying X-ray diffraction and different methods of peak profile analysis,” Physica Scripta, vol. 96, no. 9, p. 095704, 2021. |
| [37] | W. Ahmad and D. Kalra, “Green synthesis, characterization and antimicrobial activities of ZnO nanoparticles using Euphorbia hirta leaf extract,” J. King Saud Univ. Sci., vol. 32, no. 4, pp. 2358–2364, 2020. |
| [38] | G. H. Al-Hazmi, M. G. El-Desouky, and A. A. El-Bindary, “Synthesis, characterization and microstructural evaluation of ZnO nanoparticles by Williamson–Hall and size–strain plot methods,” Bull. Chem. Soc. Ethiop., vol. 36, no. 4, 2022. |
| [39] | N. S. M. Alnagar, M. Hasanin, W. B. Suleiman, S. A. Zaki, and A. H. Hashem, “Preparation of nanocomposite based on zinc oxide nanoparticles and biopolymers: Characterization, antimicrobial and anticancer activities,” Microbial Biosystems, vol. 10, no. 1, pp. 68–81, 2025. |
| [40] | A. M. Awwad, M. W. Amer, N. M. Salem, and A. O. Abdeen, “Green synthesis of zinc oxide nanoparticles (ZnO-NPs) using Ailanthus altissima fruit extracts and antibacterial activity,” Chem. Int., vol. 6, no. 3, pp. 151–159, 2020. |
| [41] | E. K. Droepenu, B. S. Wee, S. F. Chin, K. Y. Kok, and M. F. Maligan, “Zinc oxide nanoparticles synthesis methods and its effect on morphology: A review,” 2022. |
| [42] | R. R. Gandhi and D. K. Koche, “An insight of zinc oxide nanoparticles (ZnO NPs): Green synthesis, characteristics and agricultural applications,” Biosci. Biotechnol. Res. Asia, vol. 21, no. 3, pp. 863–876, 2024. |
| [43] | M. A. L. Grace, K. V. Rao, K. Anuradha, A. J. Jayarani, and A. Rathika, “X-ray analysis and size–strain plot of zinc oxide nanoparticles by Williamson–Hall,” Mater. Today Proc., vol. 92, pp. 1334–1339, 2023. |
| [44] | F. Güell et al., “ZnO-based nanomaterials approach for photocatalytic and sensing applications: Recent progress and trends,” Mater. Adv., vol. 4, no. 17, pp. 3685–3707, 2023. |
| [45] | H. Hameed et al., “Green synthesis of zinc oxide (ZnO) nanoparticles from green algae and their assessment in various biological applications,” Micromachines, vol. 14, no. 5, p. 928, 2023. |
| [46] | N. A. S. M. Idris et al., “Optical and optoelectronic metal oxide-based sensors (optical sensors, principle, computational modeling, and application-based development),” in Metal Oxides for Optoelectronics and Optics-Based Medical Applications, Elsevier, 2022, pp. 151–164. |
| [47] | A. Jayachandran, T. Aswathy, and A. S. Nair, “Green synthesis and characterization of zinc oxide nanoparticles using Cayratia pedata leaf extract,” Biochem. Biophys. Rep., vol. 26, p. 100995, 2021. |
| [48] | S. T. Karam and A. F. Abdulrahman, “Green synthesis and characterization of ZnO nanoparticles by using thyme plant leaf extract,” Photonics, 2022. |
| [49] | A. U. Khan et al., “Biosynthesis and characterization of zinc oxide nanoparticles (ZnONPs) obtained from the extract of waste strawberry,” J. Umm Al-Qura Univ. Appl. Sci., vol. 9, no. 3, pp. 268–275, 2023. |
| [50] | M. Kotresh, M. Patil, and S. R. Inamdar, “Reaction temperature based synthesis of ZnO nanoparticles using co-precipitation method: Detailed structural and optical characterization,” Optik, vol. 243, p. 167506, 2021. |
| [51] | A. Kumar, M. Kumar, N. Srivastava, and A. K. Atul, “Nanoscale characterization,” in Fundamentals of Low Dimensional Magnets, CRC Press, 2022, pp. 245–268. |
| [52] | K. L. Naidu et al., “Methods for characterization and quantitation of nanomaterials,” in Nanomaterials in the Battle Against Pathogens and Disease Vectors, CRC Press, 2022, pp. 83–118. |
| [53] | R. Naik et al., “Characterization of metal oxide nanomaterials,” in Advances in Space Radiation Detection: Novel Nanomaterials and Techniques, Springer, 2024, pp. 37–57. |
| [54] | M. U. Rashid et al., “Green synthesis and characterization of zinc oxide nanoparticles using Citrus limetta peels extract and their antibacterial activity against brown and soft rot pathogens and antioxidant potential,” Waste Biomass Valorization, vol. 15, no. 6, pp. 3351–3366, 2024. |
| [55] | N. Sedefoglu, “Characterization and photocatalytic activity of ZnO nanoparticles by green synthesis method,” Optik, vol. 288, p. 171217, 2023. |
| [56] | Y. Sun, W. Zhang, Q. Li, H. Liu, and X. Wang, “Preparations and applications of zinc oxide based photocatalytic materials,” Adv. Sensor Energy Mater., vol. 2, no. 3, p. 100069, 2023. |
| [57] | S. Vyas, “A short review on properties and applications of zinc oxide based thin films and devices: ZnO as a promising material for applications in electronics, optoelectronics, biomedical and sensors,” Johnson Matthey Technol. Rev., vol. 64, no. 2, pp. 202–218, 2020. |



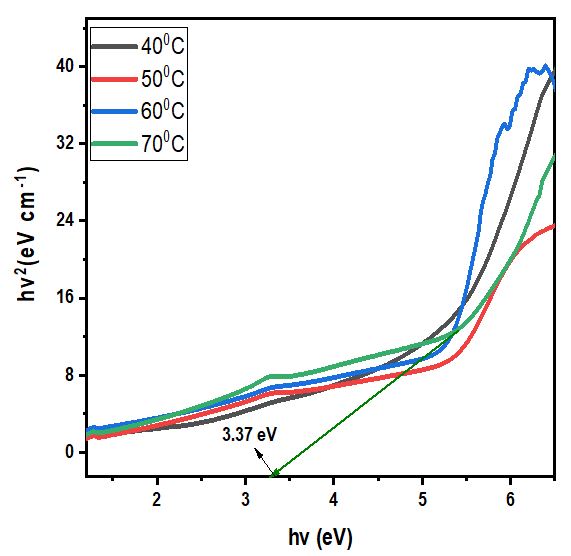
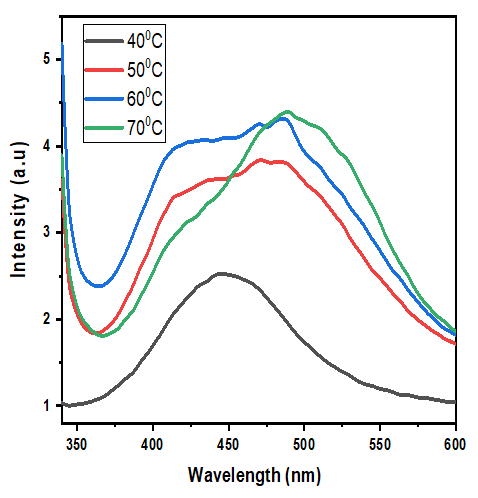
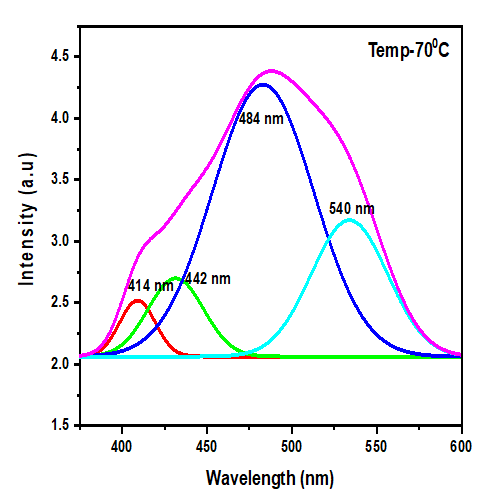
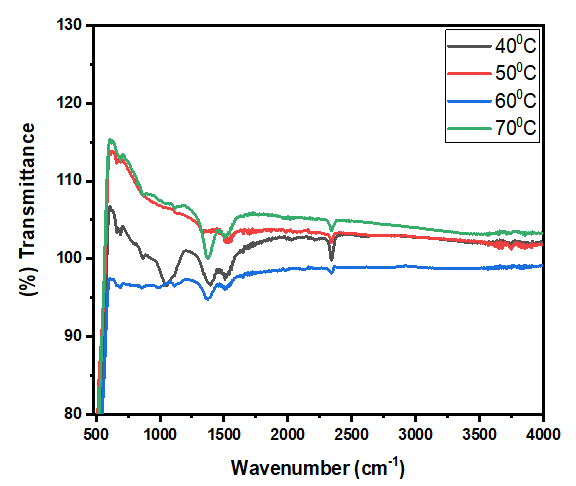
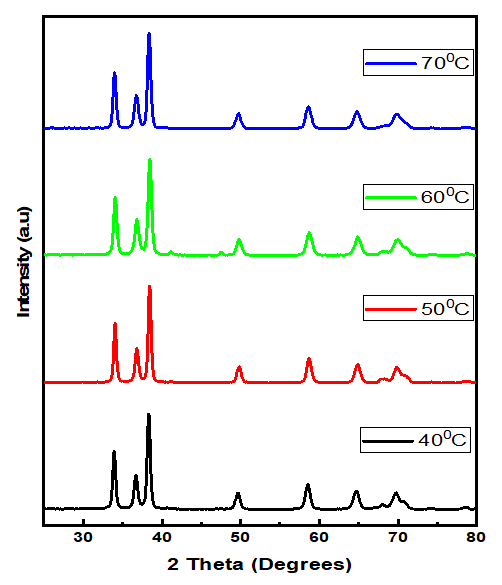
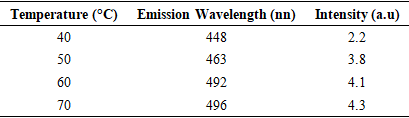
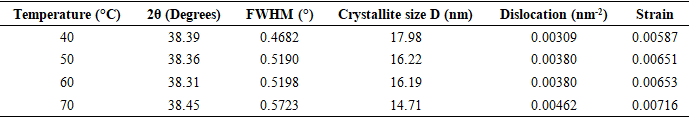
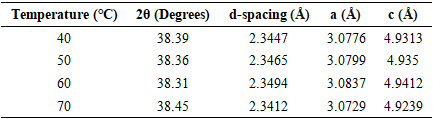
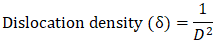 Thus, according to this relation, smaller grains tend to have more defects per unit volume, or rather more dislocations [50]. This observation also aligns with findings of various research groups [42,43], who reported increased dislocation and for smaller crystallite size in crystals of metal oxide nanoparticles with increasing calcination temperatures.Lattice strain contributes to XRD peak broadening in the same way small crystallite does [44] Strain emanates from the distortion of the crystal lattice in the form of defects, mismatch between planes and sometimes from dislocations [45]. Data in Table 3 reports a direct variation between microstrain and synthesis temperature. Notable, strain increased with increase in processing temperature. An indication of increased lattice distortion i.e. existence of imperfections such as defects of stacking faults at higher temperatures. These findings are consistent with those reported by Al-Hazmi et al, (2022) [38], a study that also revealed a close link between strain (or small size) and XRD peak broadening.Table 4 shows calculated d-spacing, and lattice parameters of synthesized samples at temperature values.
Thus, according to this relation, smaller grains tend to have more defects per unit volume, or rather more dislocations [50]. This observation also aligns with findings of various research groups [42,43], who reported increased dislocation and for smaller crystallite size in crystals of metal oxide nanoparticles with increasing calcination temperatures.Lattice strain contributes to XRD peak broadening in the same way small crystallite does [44] Strain emanates from the distortion of the crystal lattice in the form of defects, mismatch between planes and sometimes from dislocations [45]. Data in Table 3 reports a direct variation between microstrain and synthesis temperature. Notable, strain increased with increase in processing temperature. An indication of increased lattice distortion i.e. existence of imperfections such as defects of stacking faults at higher temperatures. These findings are consistent with those reported by Al-Hazmi et al, (2022) [38], a study that also revealed a close link between strain (or small size) and XRD peak broadening.Table 4 shows calculated d-spacing, and lattice parameters of synthesized samples at temperature values.

 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML