-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Environmental Engineering
p-ISSN: 2166-4633 e-ISSN: 2166-465X
2020; 10(3): 51-58
doi:10.5923/j.ajee.20201003.01
Received: Nov. 8, 2020; Accepted: Nov. 30, 2020; Published: Dec. 15, 2020

Effectiveness of Bamboo (Phyllostachys pubescens) and Papyrus (Papyrus cyperus) in uptake of Heavy Metals from Soil Contaminated with Petroleum Sludge
James Kiprotich Mulama1, Sanewu Isaac Fundi2, Oswald Gwaya2, Kenneth Chelule3
1Kenya Pipeline Cooperation, Nairobi, Kenya
2Construction Management, Jomo Kenyatta University of Agriculture and Technology, Nairobi, Kenya
3Kenya Industrial and Development Institute, Nairobi, Kenya
Correspondence to: Sanewu Isaac Fundi, Construction Management, Jomo Kenyatta University of Agriculture and Technology, Nairobi, Kenya.
| Email: |  |
Copyright © 2020 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Petroleum sludge is a residue extracted mostly from petroleum storage. Contaminants from petroleum sludge may exist in the soil for long periods and be transmitted to plants and tissues of living organisms within the soil ecosystem. The aim of this study was to investigate sustainable technology for enhancing bioremediation of soil contaminated with petroleum sludge. The study involved planting of Bamboo (Phyllostachys pubescens) and Papyrus (Papyrus cyperus) plants in contaminated soil. Parts of the plant (roots, stem and leaves) were examined for their effectiveness of absorption and bioaccumulation of three heavy metals (lead, copper and chromium). The plants were harvested after every three months of growing for a period of one year. Presence of heavy metal in the soil and the harvested plant parts were analyzed by Atomic Absorption Spectrophotometry method. After growing the plants in the contaminated soil for 12 months, it was found that highest amounts of chromium (86.86%) were reduced by Bamboo plant while copper (83.33%) was the highest reduced metal by Papyrus plant. Heavy metal analysis on the plant parts depicted that Bamboo is an effective plant in accumulation of heavy metals in petroleum contaminated soil over a long period of time. Conversely, Papyrus accumulates heavy metals over a short period of time even in presences of extra amounts of water in the soil. It was further noted that both Bamboo and Papyrus accumulate copper and lead more in the roots than in leaves, however, chromium is most accumulated in leaves.
Keywords: Absorption, Adsorption, Bioremediation, Phytoremediation, Petroleum Sludge
Cite this paper: James Kiprotich Mulama, Sanewu Isaac Fundi, Oswald Gwaya, Kenneth Chelule, Effectiveness of Bamboo (Phyllostachys pubescens) and Papyrus (Papyrus cyperus) in uptake of Heavy Metals from Soil Contaminated with Petroleum Sludge, American Journal of Environmental Engineering, Vol. 10 No. 3, 2020, pp. 51-58. doi: 10.5923/j.ajee.20201003.01.
Article Outline
1. Introduction
- Petroleum industries produce a great deal of effluent that contains various pollutants, such as heavy metals. Heavy metals pollutants are a great health concern in the environment. Depending on the components of petroleum sludge, they can be classified as soil, air or water contaminant. The contaminants are toxic, mutagenic and carcinogenic [1]. Particularly, contaminants generated from anthropogenic activities can harmfully affect both human and animals who depend on food chains affected by the contaminants. Even low levels of heavy metal contaminants in the food chains pose a health risk due to impending accumulation at higher trophic levels in the food chains through bio-magnification. The inorganic contaminants such as non-essential heavy metals are some of the common soil contaminants. They include lead (Pb), chromium (Cr), and Copper (Cu) in high concentrations.Metal uptake by plants is generally aided by certain channel proteins or H+ (transporters) coupled carrier proteins found at the cell membrane of the roots. Accidental uptake of heavy metals also takes place via other cell membrane transporters like phosphorus transporters which transports Arsenic (As) in form of Arsenic oxidearsenous trioxide (As2O3). It is therefore essential to find a sustainable solution to this potential threat with the ever-increasing demands of petroleum products. Among the most effective and additionally cost-effective suitable solution is phytoremediation. Phytoremediation bioremediation is much dependent on the physiological behavior of plants’ roots to the soil contaminants and particularly heavy metals.
1.1. Phytoremediator Potentials of Bamboo and Papyrus
- The use of plants as a technique for bioremediation technology has been embraced and found effective. Bamboo, which is a woody member of the grass family has been used as one of the bio remedies for soil pollution and erosion. About 18 species of Bamboo are reliably hyper accumulative of zinc (Zn) and other heavy metals [2]. In hydrophonic experiments, Bamboo has shown high Zn tolerance up to application of 400µM. Bamboo can grow on metalliferous soils and can accumulate extremely high concentration of heavy metal in both the shoot and root without suffering phytotoxic effects in hyperaccumulators [3,4,5].Experimental results by Liu et al. [4] have indicated that at the highest lead (Pb) concentration (400 μmol/L), the growth of moso bamboo seedlings is repressed and Pb concentrations in roots, stems and leaves, can reach a maximum of 4282.8, 482.2, 148.8 mg/kg, respectively. In the same research, scanning electron microscopy showed that the too much Pb lessened rooting inclusions, stomatal opening, and just few stem inclusions. Even though ultrastructural analysis showed some internal physiological injuries, the plants subjected to 400 μmol/L Pb endured no physically observable Pb toxicity signs like chlorosis and necrosis. Liu et at. [4] noted that even at very high Pb treatment of the growth medium, no considerable variance was recorded for the biomass of stem as compared to controls. Bamboo is significantly tolerant to heavy metals and thus can tolerate high concentration of the heavy metals without any significant effect on its physiological behavior [6,7,8]. Bamboo has therefore been found to be a suitable phytoremediatory plant that is both sustainable and cost-effective. Papyrus on the other hand has also been used in attempts to rehabilitate wetlands. Cyperus papyrus, a species of Papyrus, has been found by Lema et al [9] to have a greater tendency for removal of copper and iron with k values of 0.333 d-1 and 0.168 d-1 respectively. In their study, Cyperus papyrus was able to reduce an initial concertation of iron from 3.515 ppm to 0.077(±0.021) ppm in the soil. A study by Home and Muthigo [10] found that with a blend of Cyperus papyrus with Polygonum spp and Typha latifolia at the ratio of 2:1:1 while the plants grown in a wetland, had optimum removal of 89.8% of lead from raw sewage.
1.2. Bioaccumulation
- Bioaccumulation is a type of biosorption in which living organisms gradually accumulate metals in their biomass through incorporation or assimilation [11]. The accumulation depends, in most cases, on active uptake, and partly on passive uptake [12]. Living organism incorporate metals within their biomass to protect themselves against metal toxicity. While in the body, the metals can be immobilized by complexation with intracellular proteins like sulfur-containing peptides [13]. Consequently, the success of bioaccumulation depends much on plant roots’ affinity toward a given metal, and on the physicochemical conditions which affect metal toxicity like Eh, and pH.
1.3. Bioavailability
- In phytoremediation, bioavailability is the presence of heavy metals in the form and quantities that can be absorbed and immobilized by the root system of a plant. It is thus a factor which define whether a heavy metal is available for uptake, and whether the form in which the metal occurs in the soil allows for its natural uptake. Bioavailability is defined by the transfer factor (TF) and bioaccumulation factor (BCF). In other words, to quantify bioavailability, one must determine the TF and the BCF. A TF of less than 1 and BCF of more than 10 implies that the bioavailability of a metal is very high and thus a lot of bioaccumulation occurs [6].
1.4. Phytoextraction
- Phytoextraction is the removal of heavy metals from a contaminated medium such as soil or water through their uptake by plant roots. The process is advantageous over traditional techniques, because it is cost effective, can be used to treat pollution by more than one metal, can be conducted in situ and it is environmentally friendly [14]. However, Fawzy et al. [15] have found the following factors to influence the success of phytoextraction: dissolution of the metal in a medium that is absorbable by plant roots, bioavailability of the metal for the plant roots, chelation of the metal to protect itself and make the metal more mobile.It is in this backdrop that the study sought to determine the effectiveness of Bamboo (Phyllostachys pubescens) and Papyrus (Cyperus papyrus) species in accumulating heavy metals in soil contaminated with petroleum sludge.
2. Methodology
- The study followed an experimental design which included soil collection, cultivation of plants and analysis for heavy metals. Soil contaminated with petroleum sludge from a dump site in Makueni County, Kenya was analyzed through Atomic Absorption Spectrophotomentry (AAS) for lead, chromium and copper. Bamboo and papyrus plants were grown in the soil contaminated with petroleum sludge for a period of 1 year. The roots, stems and leaves of the plants were harvested and analyzed for the heavy metals. The plants were grown in a greenhouse to enhance monitoring under similar conditions. An investigation into the bioaccumulation of the heavy metals in the various parts of the plants was studied.
2.1. Sample Collection
- Soil contaminated with petroleum sludge was sampled from a Kenya Pipeline Cooperation (KPC) sludge dumping site located at Sultan-Hamud in Makueni County. Soil auger was used to scoop contaminated soil from about 30-60 cm of depth from the site. The samples were then put in cooler boxes for transport and temporary storage at 4°C.Red clay soil was sampled from Jomo Kenyatta University of Agriculture and Technology (JKUAT) soil stock used for horticultural studies. The soil was sampled following the same procedure as for the contaminated soil. Both soil samples were safely stored in a tightly sealed container to avoid any contamination. Since Bamboo seedlings were sourced from Kitil farm in Kajiado County and Papyrus rhizomes from JKUAT, the soil samples were divided into two and transported to the respective areas where the seedlings were to be sourced.Bamboo seedlings (Phyllostachys pubescens) were sampled from Kitil Farm in Isinya – Kajiado County, and isolated into a greenhouse within the farm. Papyrus rhizomes (Papyrus cyperus) on the other hand were obtained from parts of the paddy farms within JKUAT. These rhizomes were used to grow and develop seedlings which were then used for the study.
2.2. Experimental set-up
- The clean red clay soil was thoroughly mixed with the soil contaminated with petroleum sludge, compost manure and NPK fertilizer at a ratio of 3:3:1:0.1 by volume respectively. The blend was mixed until homogenous was achieved. Equal amounts of the blend were put in 32 plastic containers and water added just to make the blend moist. Four bamboo plant seedlings (300 mm tall), were planted in each of the first 16 containers having the soil blend and papyrus seedlings of the same size were also planted in each of the remaining 16 containers. This ensured that for every specific treatment to be done for the plants, there were four sets (quadruplet). These containers were placed on benches within one of the green houses at Kitil farm for bamboo, and another green house in JKUAT for papyrus (Figure 1).
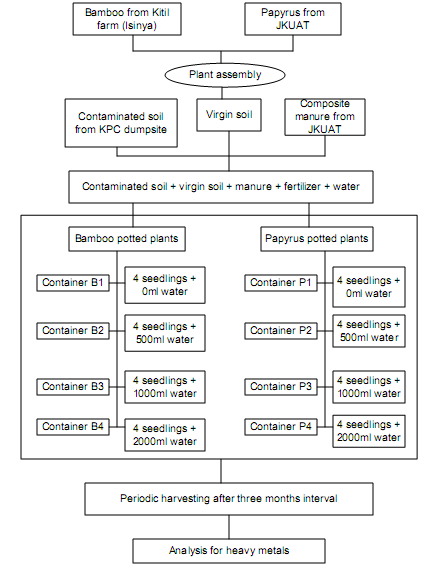 | Figure 1. Schematic flow of the experimental set-up |
2.3. Preparation for Analysis
- The plants were harvested after every three months of growing for a period of one year. The plants, on harvesting, were divided into leaves, stems and roots. They were then dried under sun. The dry samples were chopped into small pieces that could easily fit into crucibles. Samples of between 1.5 gm and 2.0 gm were weighed and placed into labeled crucibles. The crucibles were placed on wire gauze on hot coils under a fume hood and the temperature was increased gradually until smoking ceased and sampled completely charred. The charred sample were transferred to a muffle furnace and temperature adjusted to 250°C where they were allowed to heat for 1 hour. The temperature was then re-adjusted to 5500°C and left to run for 6 hours. At the end of the six hours, the temperature was lowered to 3000°C as suggested by Skoog [18]. The processes were conducted in duplicates.The ashes were quantitatively transferred to a 25ml beakers using 10ml of 1N HNO3. The samples were heated at 90ºC on a hot plate for 5 minutes. The sample was then transferred to a 50ml volumetric flask and filled up to the mark with 1N HNO3. It was shaken thoroughly to mix well. The mixture was filtered into clean dry polyethylene sample bottle and labelled according to the name of sample.Three standard solutions of copper, lead and chromium were prepared from analytical stocks of 1000 ppm. The solutions were made into 2 ppm, 1.5 ppm, 1 ppm, 0.5 ppm, 0.2 ppm and 0.1 ppm as recommended by FAO and WHO [19]. These series were used to calibrate Atomic Absorption Spectrophotomentry (AAS) machine so that standard curves for all the three elements could be established.
2.4. Heavy Metal Analysis
- All the samples were analyzed for three heavy metals elements: copper, lead and chromium. An Atomic Absorption Spectrophotomentry (AAS) machine was used in carrying out the analysis. The machine was turned on and the lamp allowed to warm up for 15 minutes. The machine was aligned and the monochromator positioned at the correct wavelength through selection of proper monochromator slit width. The hollow cathode current was placed according to the manufacturer’s recommendation. The flame was lite and the flow of fuel and oxidant regulated. The burner and nebulizer flow rate were adjusted for maximum percent absorption and stability and balanced the photometer. A calibration curve was constructed by plotting the concentrations of the standards against the absorbance after running a series of standards of the standard solution. The samples were then aspirated and their concentrations determined from the calibration curve.
3. Results and Discussion
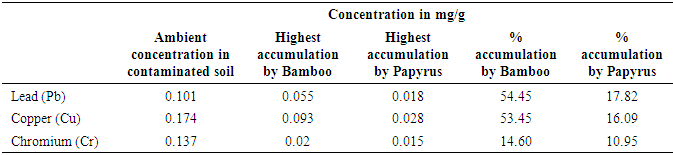
- The raw unblended contaminated soil contained copper (0.204mg/g) as the highest concentration of the heavy metals (Table 1). However, the heavy metal concentration levels for pure sludge were within the range observed by Akpoveta et al [20]. On testing the blended soil where the plants had grown for 12 months it was found that highest amounts of chromium (86.86%) were reduced in presence of Bamboo seedlings while Papyrus highly reduced copper amounts by 83.33%. After 12 months, lead was reduced by 42.57% in presence of Bamboo compared to 39.60% in Papyrus seedlings. It can be inferred from Table 1 that Bamboo seedlings are effective in reducing the amounts of chromium and lead in contaminated soils while Papyrus is effective in reducing copper.
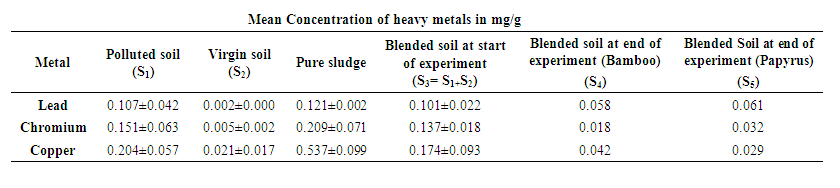 | Table 1. Concentration of heavy metals |
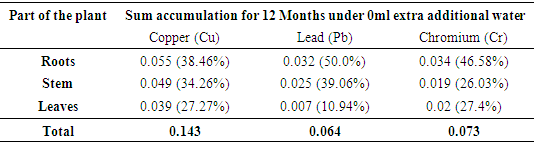
3.1. Accumulation of Heavy Metals by Bamboo
- The trend of uptake of the heavy metals by Bamboo after the 3rd, 6th, 9th and 12th month of planting are indicated in Table 2. The table present the amounts of the heavy metals accumulated by the various parts of the plant on adding varied extra amount of water in the four seasons of testing.The Bamboo roots are the most effective in the accumulation of the three heavy metals (Table 3). According to Plants Rescue [21], the roots accumulation is accounted by both adsorption and absorption of the heavy metals, while the stem only accumulates in terms of assimilation in the biomass and cellular localization. Additionally, the roots also accumulate more of the contaminates because they are point of entry of the contaminates into the plants. The leaves accumulate the least of the three metals and this pattern can be attributed to phytovolatilization.
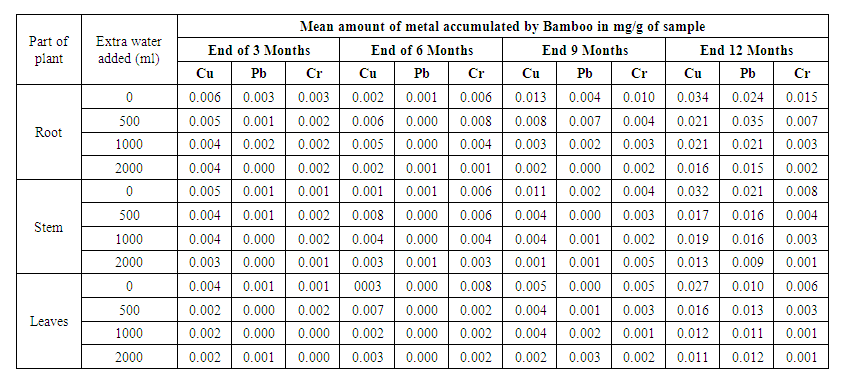 | Table 2. Mean accumulation of Lead, Copper and Chromium in 12 months by Bamboo |
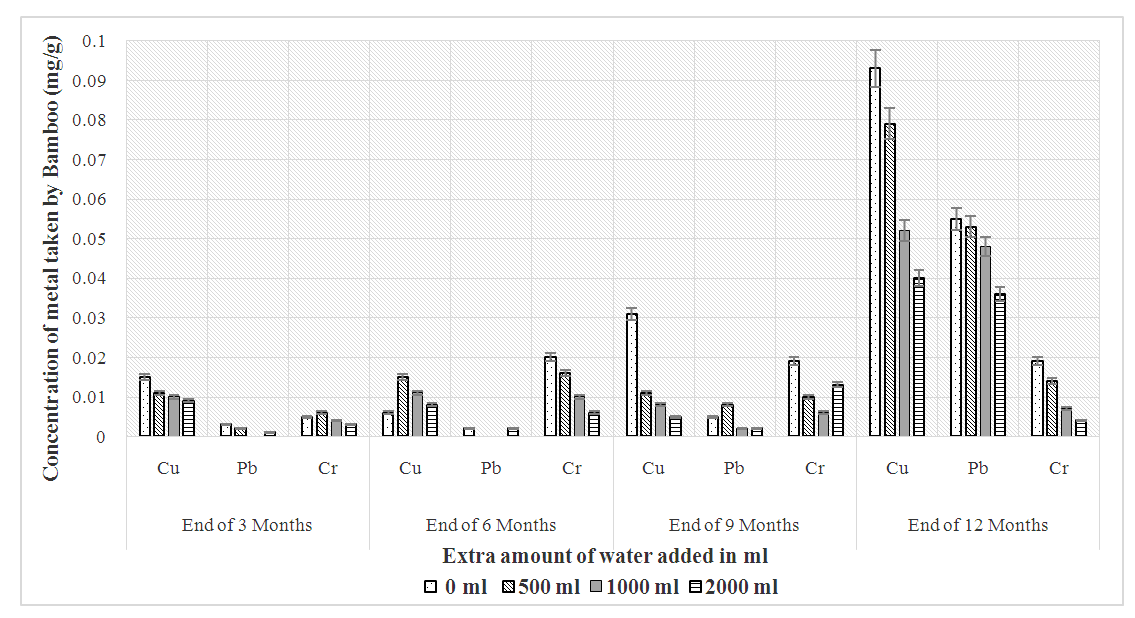 | Figure 2. Heavy metals absorption by Bamboo as a plant |
|
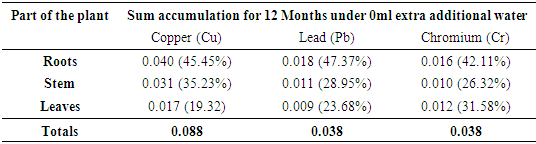
3.2. Accumulation of Metals by Papyrus
- The trend of uptake of the heavy metals by Papyrus (Papyrus cyperus) after 3rd, 6th, 9th and 12th month of planting is as shown in Table 4. Compared to the metal accumulated by Bamboo over the first 3 months, it was observed that Papyrus accumulated more quantities of the heavy metals. There was 81.25% fall of total bio-accumulated copper on adding extra water from 0ml to 2000ml after 3 months. On the contrary, the mean accumulation of chromium was steady on the addition of water with a 40% accumulation noted.
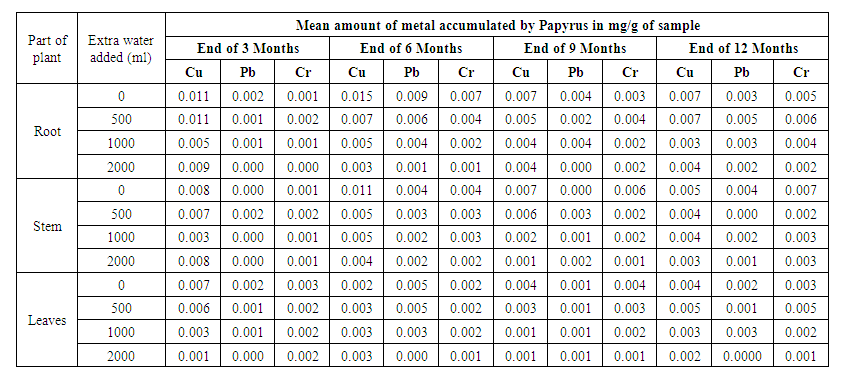 | Table 4. Trend of heavy metal uptake by Papyrus for the 12 months |
|
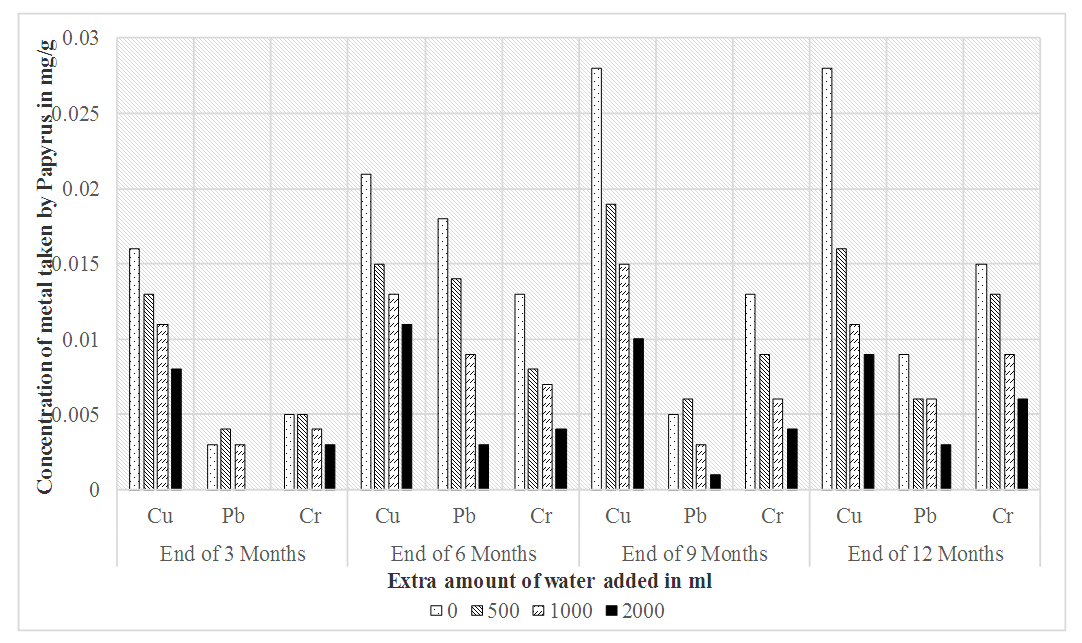 | Figure 3. Accumulation of heavy metals by Papyrus plant |
|
4. Conclusions
- This study sought to determine the effectiveness of Bamboo and Papyrus plant species in bioremediation of soils contaminated with petroleum sludge. The following conclusions were drawn;i. Both Bamboo (Phyllostachys pubescens) and Papyrus (Papyrus cyperus) exhibited a definite trend of absorbing lead, copper and chromium from soil contaminated with petroleum sludge.ii. The most accumulation of heavy metals taken up by Bamboo and Papyrus occurs in the roots with the least in the leaves. This accumulation is due to both absorption and adsorption of the plants.iii. Longer time of growth of Bamboo and Papyrus in soil contaminated with petroleum sludge improves bioaccumulation. Bioaccumulation is directly dependent on the ambient concentration of the heavy metals in soil and rate of leaching.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML