-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Dermatology and Venereology
p-ISSN: 2332-8479 e-ISSN: 2332-8487
2017; 6(1): 11-15
doi:10.5923/j.ajdv.20170601.03

Papillon-Lefèvre Syndrome - A Case Report and a Multidisciplinary approach in the Treatment of 18 Years Old Sudanese Female
Abdel Rahman M. A. I. Ramadan1, Adil H. H. Bashir2
1College of Dentistry, Taibah University, El-Madinah El- Munawarah, Kingdom of Saudi Arabia
2Institute of Endemic Diseases, University of Khartoum, Khartoum, Sudan
Correspondence to: Adil H. H. Bashir, Institute of Endemic Diseases, University of Khartoum, Khartoum, Sudan.
| Email: |  |
Copyright © 2017 Scientific & Academic Publishing. All Rights Reserved.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

An eighteen years old Sudanese female patient presented with keratotic plaques over the skin of her palms and soles and swollen gums since the age of 4 years with the subsequent loss of most of her primary and permanent dentition. These findings are consistent with Papillon-Lefèvre syndrome. Papillon-Lefevre syndrome is a rare autosomal recessive genetic disorder with a clinical presentation which includes palmer planter keratoderma associated with the precocious progressive periodontal disease that results in early exfoliation of both the primary and permanent dentitions.
Keywords: Papillon–Lefèvre syndrome (PLS), Palmar-plantar keratoderma, Periodontitis, Premature teeth loss
Cite this paper: Abdel Rahman M. A. I. Ramadan, Adil H. H. Bashir, Papillon-Lefèvre Syndrome - A Case Report and a Multidisciplinary approach in the Treatment of 18 Years Old Sudanese Female, American Journal of Dermatology and Venereology, Vol. 6 No. 1, 2017, pp. 11-15. doi: 10.5923/j.ajdv.20170601.03.
1. Introduction
- Papillon-Lefèvre syndrome (PLS) or palmoplantaris keratoderma with periodontopathia is an inherited autosomal recessive genodermal trait, affecting children characterized by diffuse, transgradient palmoplantar keratoderma (PPK), progressive destructive periodontitis starting in early childhood, between the ages 1-4 years, premature loss of primary teeth and recurrent [1]. It has a prevalence of 1-4 cases per million persons [2]. There are no racial predominance or gender predilection equally affecting both males and females [3].Further symptoms and findings may comprise of recurrent cutaneous and systemic pyogenic illnesses, nail dystrophy and hyperhidrosis [4]. Van Dyke et al. reported that the patients classically have a major disease associated with quantitative neutrophil abnormalities and almost of the afflicted patients are immunocompromised [5]. Pyogenic liver abscess is an unusual presentation of the disease. However, PLS patients appear to be exceptionally susceptible to acquire pyogenic liver abscess [6].
2. Case Presentation
- This case report illustrates a case of PLS classic clinical features and a review the pertinent literature. An eighteen-year-old female born to consanguineous parents was referred to the clinics at the Khartoum College of Medical Sciences (KCMS) with redness and peeling off of the skin of hands (Figure 1) and feet, cough, and pain of one-month duration. Her siblings and cousins did not have similar histories.
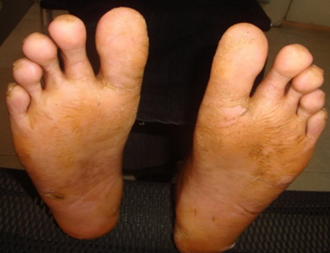 | Figure 1. Diffuse plantar keratoderma both feet |
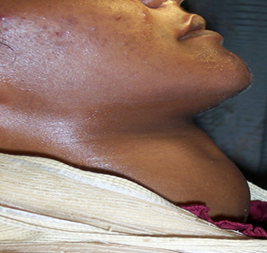 | Figure 2. Lateral view of the neck showing the enlarged thyroid gland |
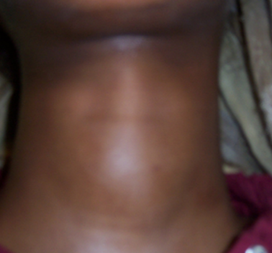 | Figure 3. Frontal view of the neck showing the enlarged thyroid gland |
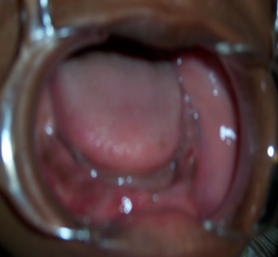 | Figure 4. Intraoral picture of the lower jaw following the extraction of the “hopeless teeth” |
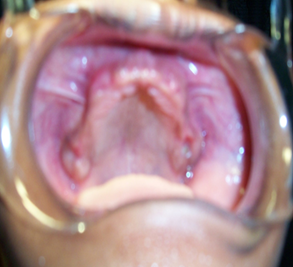 | Figure 5. Intraoral picture of the upper jaw following the extraction of the “hopeless teeth” |
 | Figure 6. The right frontal lateral view of the face following the insertion of the denture |
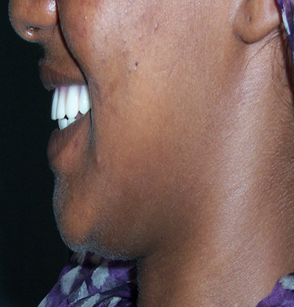 | Figure 7. Left profile of the patient following the insertion of the denture |
3. Discussion
- Papillon-Lefèvre is a rare genodermal condition that was initially described in 1924 by two French Physicians Papillon and Lefèvre, in a brother and sister suffering from palmoplantar hyperkeratosis associated with severe periodontal destruction and premature loss of deciduous as well as permanent dentition [7]. In 1964 Gorlin et al. added a third component to the syndrome; the dural calcification [8]. There are no prevalence studies reported in Sudan. Nevertheless, there are two previous reports in the Sudan of similar conditions [9, 10]. Globally, a prevalence of 1-4 cases per million in the general population has been documented with a carrier frequency of 2-4 per thousand population with no sexual predilection [3, 4].Papillon and Lefèvre syndrome (PLS) is characterized by palmoplantar keratoderma, psoriasiform plaques; that vary from mild psoriasiform scaly skin to overt hyperkeratosis of the elbows and knees. The periodontal disease with consequential premature loss of the primary and permanent teeth, and intracranial calcifications [11], which may occur focally, but more frequently they include the whole surface of the palms and soles [12]. The psoriasiform plaques usually occur within the first 2-3 years of life [13], which are often associated with hyperhidrosis of the soles and the palms producing a foul-smelling odor [2]. Siragusa et al. have reported that dermatological condition may worsen in winter and may be concomitant with painful fissures [14].The deciduous teeth develop and erupt normally, however, these teeth are associated with concomitant gingivitis and ensuing periodontitis. The resulting periodontitis is characteristically refractory to conventional periodontal therapy modalities. Eventually, the deciduous dentition is usually prematurely exfoliated by the age of 4 years. Following the exfoliation, the gingivitis subsides, and the gingiva consequently appears healthy. However, the process of gingivitis and periodontitis is usually repeated with the eruption of the permanent dentition, and there is subsequent premature exfoliation of the permanent teeth as soon as they erupt though the third molars are occasionally spared [15, 16]. As of resorption of underlying alveolar bone as a result of the aggressive form of the periodontitis, the teeth appear as floating in the air in radiographs [17].The pathogenesis of PLS remains controversial. The dermatological lesions are assumed to be due to the disorders in the ectodermal and mesodermal components. However, this does not explain the rapid loss of both the deciduous as well as permanent teeth in the order of their eruption [18].There are three postulations suggested as being responsible for the initiation and progression of the syndrome. Firstly, the associated impairment of qualitative attributed the Polymorphonuclear lymphocytes (PMNL) accompanied by a decrease in cell migration has been noted. Secondly, the presence of numerous virulent gram-negative anaerobic pathogens (as Porphyromonas gingivalis, Capnocytophaga gingivalis, Aggregatibacter actinomycetemcomitans, Peptostreptococcus micros, Fusobacterium nucleatum, and spirochetes) in the periodontal plaques and periodontal pockets have been noted. These periodontopathogens are thought to act as trigger factors. Thirdly, a defect of immune-mediated mechanisms including the reduced lymphocyte response to the periodontopathogens, depression of helper/suppressor T cells ratio, impaired monocytic function, elevation of serum IgG and the degenerative changes in the plasma cells have been identified [17, 19-22].Genetic studies associate Papillon-Lefevre syndrome due to mutations in the cathepsin C (CTSC) gene mapped to chromosome 11q14-2 that codes for cathepsin C (dipeptidyl peptidase I). The CTSC gene is a lysosomal protease that takes part in the epidermal differentiation and desquamation and in the serine proteases activation that is found in cells of the immune system which include polymorphonuclear leukocytes, macrophages, and their precursors. A mutation in the CTSC gene has the total potential loss of cathepsin C activity and reduced activity in the obligate carriers results in the susceptibility to specific virulent pathogens which has been demonstrated in PLS patients [13, 23].The cathepsin-C gene is seen in the epithelial regions of the palms, soles, knees, and the keratinized oral gingiva; regions that are usually affected by PLS. All PLS patients are homozygous for the same cathepsin-C mutations which are inherited from a common ancestor. The parents and the siblings, which are heterozygous for the cathepsin C mutations present neither the palmoplantar hyperkeratosis nor the severe aggressive periodontopathia which is characteristic of PLS [8].The treatment of PLS patients is a multidisciplinary approach. The dermatological manifestations are usually treated with emollients [7]. Oral retinoids such as acitretin, etretinate, and isotretinoin have been reported to be useful for both the oral and dermatological lesions of PLS. The use of retinoid as treatment reduce the palmoplantar keratoderma and slow the alveolar bone lysis and may produce normal dental development if initiated during the eruption of the permanent teeth [24].The periodontitis associated with PLS is usually difficult to control with conventional periodontal therapy modalities. Effective treatment of periodontitis includes extraction of the hopeless teeth combined with an appropriate oral antibiotic, the use of mouthwashes and professional scaling and root planing of the teeth, but usually, this procedure is not enough. Eventually, however, primary or remaining teeth are extracted and are replaced by either removable or fixed prosthetics [25].The vitamin A (synthetic retinoids – etretinate, isotretinoin, and acitretin) have been reported to reduce the development of periodontitis and maintain the teeth. The antibiotic therapy should also be prescribed to reduce the active periodontitis, to maintain the teeth and to avoid the bacteremia, and the treatment of recurrent infections and pyogenic liver abscesses [26].The observation of the thyroid enlargement in this patient is thought to be a potential association with the PLS. However, this from the western region of the Sudan which is a widely assumed endemic areas of iodine deficiency due to environmental and social factors.
Notes
- 1. Alginate, Cavex 37, Netherlands.2. Heraeus Kulzer GmbH, Germany.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML