-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Condensed Matter Physics
p-ISSN: 2163-1115 e-ISSN: 2163-1123
2013; 3(4): 89-103
doi:10.5923/j.ajcmp.20130304.01
Mechanism and Kinetics of Phase Transitions and Other Reactions in Solids
Yuri Mnyukh
Chemistry Department and Radiation and Solid State Laboratory, New York University, New York, NY 10003, USA
Correspondence to: Yuri Mnyukh, Chemistry Department and Radiation and Solid State Laboratory, New York University, New York, NY 10003, USA.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
The work leading to the universal contact molecular mechanism of phase transitions and other reactions in solids is presented. The two components of the mechanism - nucleation and interface propagation - are investigated in detail and their role in kinetics is elucidated. They were shown to be peculiar: nucleation is pre-coded, rather than resulted from a successful fluctuation, and the interface propagates not by frontal advancement as a whole, but by molecular filling thin layers in the transverse direction. The type of the nucleation sites is determined. An inherent instability and irreproducibility of the kinetics is revealed. A linear kinetics, as opposed to the bulk kinetics, is shown to be in accord with the contact mechanism. Ferromagnetic phase transition and magnetization process are added to the list of solid-state reactions: neither occurs without nucleation-and-growth structural rearrangement.
Keywords: Phase Transitions, Solid-State Reactions, Nucleation, Interface, Kinetics, Crystal Growth, Crystal Defects, Molecular Mechanism
Cite this paper: Yuri Mnyukh, Mechanism and Kinetics of Phase Transitions and Other Reactions in Solids, American Journal of Condensed Matter Physics, Vol. 3 No. 4, 2013, pp. 89-103. doi: 10.5923/j.ajcmp.20130304.01.
Article Outline
1. Introduction
- The basics of solid-state phase transitions[1-16] will be summarized and supplemented here by analysis of their kinetics − an aspect of essential theoretical and applied importance. The term kinetics means relationships between the macroscopic rate of a phase transition and any conditions or parameters it depends on. The notion kinetics implies phase coexistence, for it makes sense only if the mass fraction between the two phases is changing during the transition. The only conceivable way of this change is nucleation and propagation of interfaces. In other words, investigation of kinetics of phase transitions already means recognition of their nucleation-and-growth mechanism. Only two ways to materialize for crystal phase transitions are conceivable: (a) by nucleation-and-growth and (b) by instant change at a critical point[1,2]. If a phase transition is a "critical phenomenon", its kinetics must not exist, for it comprises all the matter at once as soon as its critical temperature Tc is attained. The rate of this instant transition is function neither time t, nor temperature T. The concepts "kinetics" and "critical phenomenon" are incompatible. It should be noted that instant phase transitions have not been found[1,2]. Kinetics of phase transitions is inseparable from their molecular mechanism. As soon as the nucleation-and-growth nature of solid-state phase transitions is recognized, it becomes evident that comprehension of their kinetics requires a certain knowledge of the structure and properties of the nuclei and interfaces. Critical step in that direction has been a discovery of edgewise (or stepwise) molecular mechanism of phase transitions[5,7,10]. The nucleation, in particular, was found quite different from the "classical" interpretation, its features critically affecting the phase transition kinetics, as will be demonstrated. Closely preceding to that discovery was the 1960 International Symposium on Reactivity of Solids[17]. It "focused[its] attention on the mechanism and kinetics of reaction in solids". It dealt with such reactions as polymorphic phase transitions, recrystallization, decomposition, chemical reduction and polymerization, not counting those involving also liquid or gas. The vast literature that treated almost all phase transitions as "continuous" and "critical phenomenon" was noticeably ignored. It was observed that "imperfections are preferred sites for internal nucleus formations". (The term "preferred" means, however, that possibility of a homogeneous nucleation does not eliminated). These imperfections are: vacancies, interstitials, foreign atoms or ions, linear dislocations, screw dislocations. Their interaction and diffusion were discussed. It was noted that the more perfect crystal, the lesser is its reactivity. The term "nucleation and growth" was common. It was not new, however, for in 1930's and 1940's it was used in developing of what can be called "bulk kinetics" theories. Nevertheless, nucleation-and- growth phase transitions were not considered the only way to occur. For example, one contributor[18] classified solid-state transformations as topotactic, epitactic and reconstructive, only the last one to occur by nucleation and growth. In another case[19] the imaginary way of the olivine-spinel restructuring through intermediate states, assisted by dislocations and diffusion, was proposed. It was not mentioned how the notion "kinetics" can be applied to the phase transitions assumed not to occur by nucleation and growth. In the bulk kinetics the mass fraction m of one phase in a two-phase specimen was the value of interest. Its rate depends both on multiple nucleation and growth in unknown proportions, different in every particular case. Nucleation critically depends on the presence, distribution and generation of specific lattice defects. It is quite different in a perfect and imperfect single crystal, in a big and small crystal, in a single crystal and polycrystal, in a fine-grain and coarse-grain polycrystal or powder. Growth is not a stable value either, not repeating itself, for example, in cycling phase transitions. The nucleation and growth, when they act together, are not only irreproducible and uncontrollable, there is no way to theoretically separate their contributions in order to calculate the total bulk rate. In this context, the theoretical approach called "formal kinetics" should be mentioned where an attempt to separate them was undertaken. The Avrami work[20] is most known. One of his main assumptions − isothermal rate of nucleation − was invalid due to a "pre-coded" character of both nucleation sites and the nucleation temperatures[16]. Bulk kinetics can shed no light upon the physics of phase transitions or properly account for their kinetics. As a minimum condition, the nucleation and interface motion contributions must be experimentally separated. The best way to do that is a visual observation of nuclei formation and interface motion in optically transparent single crystals. This is a method of interface kinetics. The purpose is to reveal its physics, rather then phenomenology.As mentioned, there is a reproducibility problem. The absolute velocity V of interface motion is not reproduced even in the same single crystal. Fortunately, valuable information can be obtained from a relative V changes. As will be shown, it helps not only verify and substantiate the suggested contact mechanism, but to penetrate deeper into its details. It will be demonstrated that the universal contact mechanism accounts for all the complexity, versatility, and poor reproducibility of the kinetics of solid-solid phase transitions.
2. Interface and Its Motion
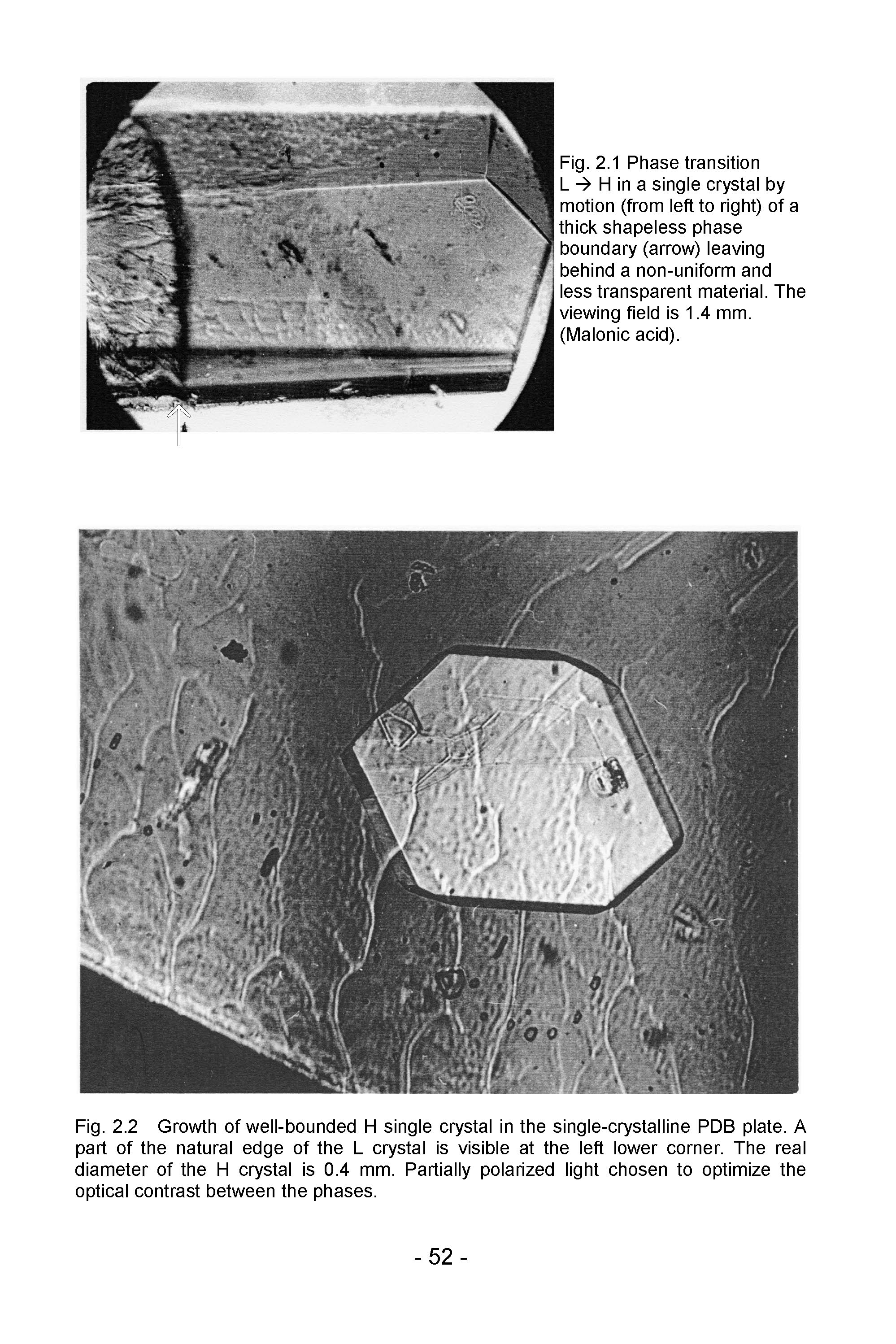
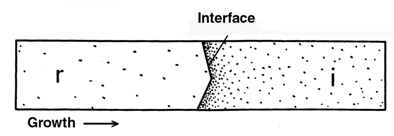
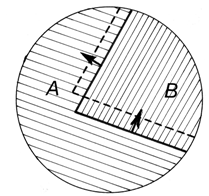
- After the discovery has been made that phase transitions in single crystals of p-dichlorobenzene (PDB) and some other substances were a growth of well-bounded single crystals of the new phase (Fig. 1)[4,6] and that orientation relationship (OR) between the initial and resultant crystals did not exist[6], the attention was concentrated on the mode of interface propagation. Observation of the interface was undertaken under maximum resolution attainable with a regular optical microscope. It was found that its motion has edgewise (or "stepwise") mechanism[5,7]. Its advancement in the normal direction proceeded by transverse shuttle-like strokes of small steps (kinks), every time adding a thin layer to it (Fig. 2). That was the same mechanism of crystal growth from liquids and gases[21,22], going down to the molecular level. A generalization came to light: any process resulting in a crystal state, whichever the initial phase is − gas, liquid, or solid − is a crystal growth. It proceeds by the edgewise molecule-by-molecule formation of layers and layer-by-layer additions to the natural crystal face. While crystal growth from gaseous and liquid phases is called crystal growth, and crystal growth from a solid phase is called phase transformation or phase transition, the difference is semantic. This does not mean, of course, that crystal growth in a crystal medium does not have its specificity.
 | Figure 2. L à H phase transition in PDB. The 2.5 µm high "kink" (step) is moving from left to right. The kink is, in fact, an avalanche of molecular steps |
3. Nucleation in Crystals
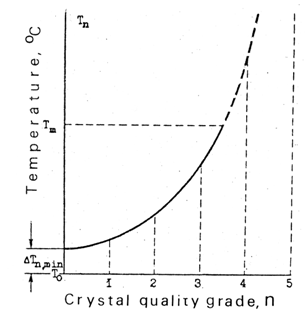
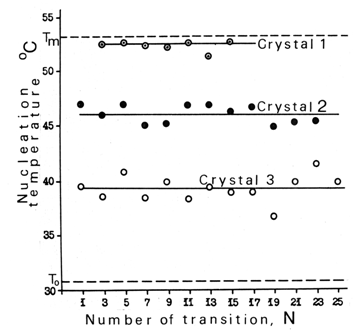
- Nucleation is one of the two elements - nucleation and growth - constituting the phenomenon of solid-state phase transitions. Nevertheless, there is vast theoretical literature that treats them as if the phenomenon of nucleation (and subsequent growth) is nonexistent. For example, nucleation is missing in the three (unrelated) books Structural Phase Transitions[37-39], in many other books on the subject[e. g., 40-47], in most volumes of Phase Transitions and Critical Phenomena[48], and in innumerous journal articles. Yet, some literature on nucleation in a solid state exists, mainly owing to needs of solid-state reactivity, as described above, and physical metallurgy[49,50]. In the latter case the theory of nucleation in a liquid phase was slightly modified to cover solid state[49,51]. But no one previously experimentally verified its theoretical assumptions by using the simplest and most informative objects − good quality small transparent single crystals.The following data were accumulated with thermal-induced phase transitions in tiny (1 - 2 mm) good quality transparent organic single crystals under controlled temperatures, PDB being the main object[16]. Notations: L and H are low- and high-temperature phases; To− temperature when free energies of the phases are equal, Tm− temperature of melting, Tn− temperature of nucleus formation. In PDB, To = 30.8℃ and Tm = 53.2℃. Nucleation requires finite overheating / overcooling. Nucleation never occurs at To or closer than at a certain finite distance from it. The "prohibited" range for PDB is at least 28 to 32℃. Upon slow heating, nucleation in most PDB crystals will not occur until the temperature exceeds 38℃. Often L-phase melts at Tm without transition into H. Nucleation is a rare event. Slow heating (e. g., 1 to 10℃ per hour) does not produce many nuclei. Usually there are only a few units, or only one, or no nucleation sites at all. This observation is at variance with the notion "rate of nucleation" and the statistical approach to nucleation - at least as applied to a single-crystal medium.Exact temperature of nucleation Tn is unknown a priori. Formation of a nucleus upon slow heating of a PDB single crystal will occur somewhere between 34 and (Tm=) 53.2℃, and in some cases it would not occur at all. The Tn vary in different crystals of the same substance. .Only crystal defects serve as the nucleation sites. In other words, the nucleation is always heterogeneous as opposed to the homogeneous nucleation assumed to occur by a successful fluctuation in any point of ideal crystal lattice. The location of a nucleus can be foretold with a good probability when the crystal has a visible defect. Nucleation in sufficiently overheated / overcooled crystals can be initiated by an "artificial defect" (a slight prick with a glass string). In such a case, a nucleus appears at the damaged spot. In cyclic phase transitions L à H à L à H the nucleus frequently appears several times at the same location. The higher crystal perfection, the greater is overheating or overcooling ± Δ Tn = Tn - To. In brief, better crystals exhibit wider hysteresis, for ± ΔTn is a hysteresis[13]. The correlation between the degree of crystal perfection and ΔTn is confirmed in several ways. One, illustrated with the qualitative plot in Fig. 4, is the ΔTn dependence on the estimated quality of the single crystals. Different grades were assigned to sets of crystals depending on their quality, higher grades corresponding to higher crystal perfection. The grades were given on the basis of crystal appearance (perfection of the faces and bulk uniformity) and the way the crystals were grown (from solution, from vapor, rate of growth, temperature stability upon growing, etc.). For the solution-grown PDB crystals the graduation exhibited good correlation with the levels of ΔTn within the grades 1 to 4. Two findings will be noted regarding ΔTn in these crystals: 1. ΔTn <1.9℃ was not found even in the worst grade (grade 1) crystals, indicating existence of a minimum (threshold) overheating required for nucleation to occur.2. The most perfect among the solution-grown crystals were incapable of LàH changing, so it was L that melted. There was no doubt in the availability of dislocation lines, individual vacancies, interstitial and foreign molecules in those crystals, but they were not of the "proper" nucleation type. Yet, an "artificial defect", created by a slight prick at the temperature near Tm, immediately initiated the phase transition from the damaged spot.Tn is pre-coded in the crystal defect acting as the nucleation site. Fig. 5 shows the results of microscopic observation of PDB (L) single crystals upon slow heating. Tn was recorded as soon as nucleation of the H phase was noticed. Growth of the H crystal was quickly stopped and the specimen was returned to the L phase. The cycle was repeated with the same crystal many times. Then the whole procedure was performed with another crystal. The experiments revealed that (a) a nucleus appears every time at exactly the same location, (b) the Tn repeats itself as well, and (c) a particular Tn is associated only with the particular nucleation site. A different Tn is found in another crystal, also associated with the defect acting as the nucleation L site. Thus, every crystal defect acting as a dormant nucleation site contains its own Tn encoded in its structure. If a crystal has more than one dormant nucleation site and is subjected to very slow heating, only one site with the lowest Tn will be activated. Upon faster heating, the second nucleus of the second lowest Tn may have time to be activated before growth from the first nucleus spreads over its location…and so on.
4. Formation of a Nucleus: A Predetermined Act, Rather Than a Successful Random Fluctuation
- The conventional theory of nucleation in solids[49] considered nucleation of new phase to be a consequence of random heterophase fluctuations that give rise to formation of clusters large enough to become stable. The only change made in the formulae over nucleation in liquids and gases is an extra term representing the idea that nucleation barrier in solids is higher due to arising strains. The theory in question originally considered nucleation to be homogeneous (assuming equal probability for nuclei to appear at any point in the crystal), but had later to acknowledge that heterogeneous, i.e. localized at crystal defects, nucleation prevails. Therefore the theory was modified to take this into account. Heterogeneous nucleation is believed to occur at dislocation lines, foreign molecules and vacancies being present in real crystals in great numbers. In other respects the statistical-fluctuation approach has been left intact.The real nucleation in a crystal is quite different. The observation of Δ Tn (solidàsolid) < Δ Tn (liquidàsolid) runs counter to the theoretical premise that activation energy of nucleation in solids is higher due to the internal strains. Formation of a nucleus is a rare and reproducible act bound to a predetermined location. Homogeneous nucleation is impossible. As for heterogeneous nucleation, the formation of nuclei at dislocation lines or vacancies has to be ruled out as inconsistent with evidence. There are plenty of these defects in every real crystal and they change their position under the action of even the slightest internal strains, to say nothing of the strains caused by moving interfaces. Dislocation lines, vacancies, and foreign molecules cannot account for the observed rarity, stability, and reproducibility of a nucleus. They are too primitive to contain encoded individual information about Tn and orientation of the resultant crystal.
5. The Structure of a Nucleation Site
- Finding the particular structure of a crystal defect serving as a nucleation site thus turned out to be the key to nucleation in solids. The solution is presented below. It inevitably involves an element of speculation, but only to link all elements of comprehensive evidence into a self-consistent and clear picture. It is qualitative, but more reliable than a detailed mathematical description of an idea that has not been verified. Let us sum up the properties of these defects: − there are few such defects in a good single crystal, − they reside at permanent locations, − they do not form spontaneously over long-term storage at any level of overheating / overcooling, − they are stable enough to withstand rather strong influences, − they are not quite as stable as macroscopic defects, − they possess a memory large enough to contain individual information both on Tn and nucleus orientation − they are capable of activating the stored information repeatedly, − their structure permits nucleation without development of prohibitive strains.Microcavities (cluster of vacancies) of some optimum size (Fig. 6) are, perhaps, the only type of crystal defect that meets all these requirements. An optimum microcavity (OM) eliminates the problem of the great strains that, possibly, prevent nucleation in a defect-free crystal medium. The OM consists of many individual vacancies and is therefore relatively stable and bound to a permanent location. There can only be a few such large-sized defects in a good single crystal, or even none. Yet, the defect in question is far from a macroscopic size, so it can be affected in some way to lose its nucleation function. Variations of its size and shape account for its capability of storing individual information on nucleation. (One additional vacancy can be attached to OM in many different ways, thus adding many new variants of "encoded" information).
6. Epitaxial Nucleation
- In general, the described nucleation site does not require structural orientation relationship. Not rarely, however, OR is observed in certain phase transitions (setting aside those where OR was incorrectly assumed). That does not mean these transitions occurred by a kind of "deformation" of the original phase or "displacement" of its molecules, as still frequently believed. They occur by nucleation and growth as well. There are two circumstances when strict OR in phase transitions occurs. One is in layered crystal structures [8,52,53.]. A layered structure consists of strongly bounded, energetically advantageous two-dimensional units − molecular layers − usually appearing in both phases. There the interlayer interaction is weak on definition. Since the layer stacking contributes relatively little to the total lattice energy, the difference in the total free energies of the two structural variants is small. This is why layered crystals are prone to polymorphism. Change from one polymorph to the other is reduced mainly to the mode of layer stacking. The layer parameters themselves are only slightly affected by the different layer stacking. In practice, layered structures always have numerous defects of imprecise layer stacking. Most of these defects are minute wedge-like interlayer cracks located at the crystal faces as viewed from the side of layer edges. In such a microcavity there always is a point where the gap has the optimum width for nucleation. There the molecular relocation from one wall to the other occurs with no steric hindrance and, at the same time, with the aid of attraction from the opposite wall. In view of the close structural similarity of the layers in the two polymorphs, this nucleation is epitaxial. Orienting effect of the substrate (the opposite wall) preserves the orientation of molecular layers.Another case of the epitaxial nucleation is when the unit cell parameters of the polymorphs are extremely close even in non-layered crystals, as in the Fe ferromagnetic phase transition. This is also the cause of rigorous OR in case of magnetization of polydomain structures where the "polymorphs" have identical crystal structure[53,54].The kinetics of epitaxial phase transitions differs significantly from the non-epitaxial. Hysteresis ΔTn in epitaxial phase transitions is much smaller. Due to the abundance of the wedge-like microcracks in layered crystals, there is no shortage in the nucleation sites of optimum size. At that, the presence of a substrate of almost identical surface structure stimulates the molecular relocation. Therefore only small overheating or overcooling is required in order to initiate and quickly complete this kind of nucleation-and-growth reaction. Without a scrupulous verification, the phase transitions in question may be taken for being (kinetics-free) "displacive", "instantaneous", “cooperative", "soft-mode", "second-order", etc. ( Note: Epitaxial nucleation is the cause of formation of polydomain structures due to appearance of the nucleus in two or more equivalent positions when allowed by the substrate symmetry[53]).
7. Interface Motion: Additional Considerations
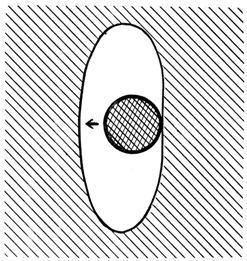
- Section 2 outlined how the interface moves forward by a "molecule-by-molecule" relocation at the molecular steps. The availability of some extra space at the steps to provide sufficient steric freedom for the relocation was noted as an important condition. This extra space eventually comes out on the surface and a new space must appear at the interface for the process to continue. Consequently, a phase transition can take place only in a real crystal with a sufficient concentration of vacancies and/or their clusters. The interface kinetics described in the present section offers strong support to that basic concept. Moreover, it made possible to gain insight into the intimate details of interface motion. We deal with two types of nuclei: 3-D (denoted as OM) to initiate a phase transition and 2-D to initiate a new molecular layer to advance the interface in the direction of its normal. The normal velocity of the interface motion Vn is controlled by the latter. The 2-D nuclei form only heterogeneously, like the OM do. There is a significant difference in their function. While only one OM is needed to start a phase transition, a sufficient concentration of appropriate defects is required to keep the interface moving. These defects are also microcavities, but smaller than OM, although not just individual vacancies. They will be called vacancy aggregates (VAs). One VA acts as a site for the 2-D nucleus only once and then moves to the crystal surface. The VAs may differ by the number and combination of constituent vacancies, as well as how close to the interface they are. In the process of its motion the interface intersects the positions of VAs, which is equivalent to a flow of VAs onto the interface. Intensity of the flow depends on the concentration of VAs and is affected by VAs migration. Not all VAs of the flow can be effective, but only those with the activation energy of nucleation that is lower of a particular level. That level is determined by the overheating / overcooling ΔT2-D = |Ttr - To|, where Ttr is the actual temperature of phase transition. One completed phase transition in a crystal "consumes" only a part of the available VAs, leaving a possibility for the transition to be repeated..Another major phenomenon of interface kinetics results from the fact that the interface motion is a crystallization, and, consequently, a process of purification. The cause of the purification is obvious: attachment of a proper particle to the growing crystal is more probable than of a foreign one. (Due to the "repulsion" of foreign particles by a growing crystal, crystallization from liquid phase is utilized in practice for purification of substances). In this process any crystal defects are "foreign particles" as well. In particular, vacancies and VAs form a "cloud" in front of the moving interface, as sketched in Fig. 7. This phenomenon gives rise to the following effects:− intensification of the VAs flow to the interface, resulting in a faster interface motion;− intensification of coagulation of vacancies into VAs and VAs into larger VAs in the "cloud";− dissipation of the "cloud" with time due to migration of the defects towards their lower concentration, that is, away from the interface.
8. Experimental Facts of Interface Kinetics
8.1. No Phase Transition in Defect-Free Crystal
- This experimental fact, already described in Sec. 3, is fundamental. If crystals are "too perfect" they do not change their phase state at any temperature. Even when OMs to start phase transition are purposely created, the interface motion cannot proceed, lacking a sufficient concentration of VAs. Obviously, the crystals in which the phenomenon was observed were still far from being ideal, still containing defects such as vacancies and linear dislocations. The defects that these crystals were lacking were VAs. A new VA is required to form 2-D nucleus on the interface every time the previous one exits on the crystal surface. If the VAs are present, but their concentration is lower than required for uninterrupted 2-D nucleation, there will be no phase transition. One would be wise to take this fact into account prior to undertaking a theoretical work on phase transitions in ideal crystal medium.
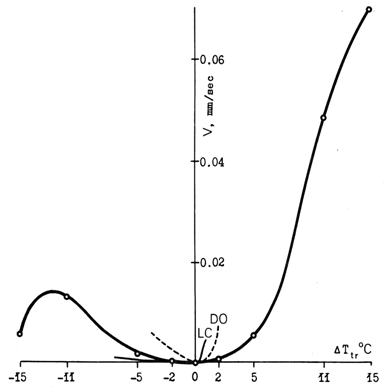
8.2. Temperature Dependence
8.3. Hysteresis of Interface Motion
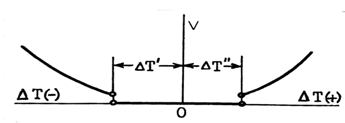
- From the two types of temperature hysteresis, the ΔT2-D to keep the interfaces moving is, as a rule, much smaller. In order to observe it, interface motion should first be stopped by setting Ttr= To, and then set in motion again by deviation from To. However slow and careful the last procedure is performed, one will find that some finite overheating / overcooling ΔTtr ≠ 0 is required in order to resume interface motion. The situation in the vicinity of To is shown schematically in Fig. 9. Interface can move only if ΔTtr is greater than some threshold value. A phase transition is intrinsically a non-equilibrium phenomenon.. This conclusion is far from being trivial, considering that "non-equilibrium phase transitions" (evidently, assuming existence of the equilibrium) is a typical subject in theoretical literature.
8.4. Depletion of the Reserve of Lattice Defects
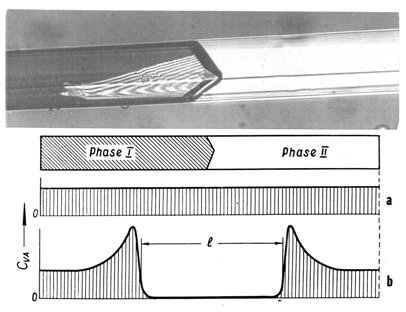
- Interface motion in the small rod-like (0.22 x 5 mm) PDB single crystal shown in Fig. 10 was manipulated on a hot microscopic stage over a prolong time. The crystal, grown from a vapor phase, was of a rather high quality. By temperature control the interface (seen in the photograph) was moved back and forth many times without letting it to reach the crystal ends. After 10 to 15 cycles, the interface was moving slower in every successive cycle under the action of the same ΔTtr. Additional 10 to 15 cycles stabilized the interface at a fixed position: it became completely insensitive to temperature changes. In this state the specimen, consisting of the two phases divided by the interface, could be stored for days at room temperature 20oC, that is, about 11o lower than To= 30.8℃. After several days the resumed experiments revealed that the dependence V = f (ΔT) had been partially restored, but the same ΔTtr produced much lower V. The initial V had not been regained even after several weeks.The schematic in Fig. 10 illustrates the cause of the phenomenon. Cyclical movements of the interface over the length ℓ have a "cleaning" effect. While "consuming" some part of the VAs for the 2-D nucleation, the interface pushed other VAs out of the ℓ area. The interface completely stopped when the concentration of VAs, CVA, fell below the critical level required for the renewable 2-D nucleation. A partial restoration of the motion capability after the long “rest" was due to VAs migration from the end regions to the “working" region ℓ. This experiment makes the intrinsic irreproducibility of V quite evident. The velocity in the same specimen under identical temperature conditions can differ by orders of magnitude.
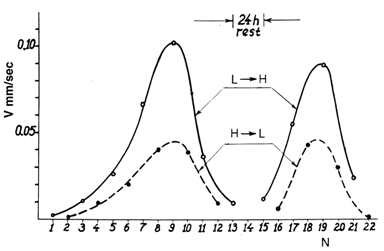
8.5. Velocity V as Function of the Number of Transitions
- This experiment was similar to the previously described, but with two differences: the crystals were not so perfect and V was measured in every interface run. The specimens were oblong PDB single crystals grown from a solution. Due to significant V scatter the measurements were averaged over 20 crystals. A region of 1 mm long was selected in the middle of a crystal and the time required for the interface to travel this distance was measured. The outside regions played a certain auxiliary role. Two microscopes with hot stages set at T1 = To + ΔT and T2 = To - ΔT were used in the measurements. The V dependence on the ordinal number N of transitions in the cyclic process was measured for a fixed |ΔT|. The V = f (N) plots are shown in Fig. 11. Only qualitative significance should be assigned to them.A new finding is the maxima, and more specifically, their ascending side - because their descending side has been explained earlier. In general terms, a moving interface initially creates more VAs than it consumes, but the tendency is reversed after a number of successive transitions. In more detail, it occurs as follows. A moving interface accumulates a "cloud" of vacancies and VAs in front of it. If density of the vacancies in the “cloud" is sufficiently high, their merging into VAs creates more VAs than is expended for the 2-D nucleation. Considering that the number of vacancies in the region is limited, the consumption eventually prevails and V begins dropping. The recurrence of the whole effect after a long "rest" is due to migration of the vacancies from the end parts of the crystal.
8.6. Lingering in Resting Interface Position
- If an interface moving with a speed V1 at a fixed ΔTtr was stopped by setting ΔT = 0, it tends to linger in the resting position once the initial ΔTtr is restored. After some lag the interface leaves the resting position, but under a lower speed V2 < V1. The longer the resting time, the lower the V2. Once resumed, the initially slow movement accelerates to approximately the previous steady V1 level.
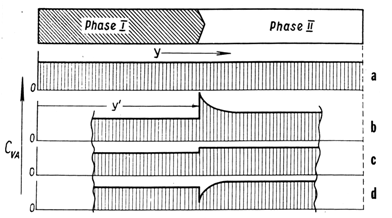
8.7. Memory of the Previous Interface Position
- If after the procedure just described the reverse run immediately follows, the moving interface "stumbles" (is retarded spontaneously) exactly at the position where it was previously resting. The phenomenon is almost a visual proof that the "hole" shown in Fig. 12d really exists. It provides a compelling support to the concept of interface kinetics based on the flow of VAs on the interface.
8.8. Slower Start upon Repetition
- Using temperature control, it is possible to set up a cyclic process in which a single nucleus of H-phase will appear, grow to a certain small size, and then dissipate back to the L-phase. In such a process, growth of the H crystal in every subsequent cycle requires a longer time. Here is the cause: the growing crystal consumes the available surrounding VAs for its 2-D nucleation, while the traveling distance is too short to accumulate a "cloud" of the defects. The concentration of VAs in the area is reduced with every successive cycle, giving rise to a lover V.
8.9. Acceleration from Start
- Just after its nucleation, an H crystal grows very slowly. It takes some traveling distance for the interface to accelerate and attain a steady V level. A "cloud" of VAs, initially absent, is then accumulated. Eventually a kind of equilibrium between their accumulation and consumption is reached, producing (in a uniform crystal medium) a translational interface motion.
8.10. Acceleration by Approaching Interface
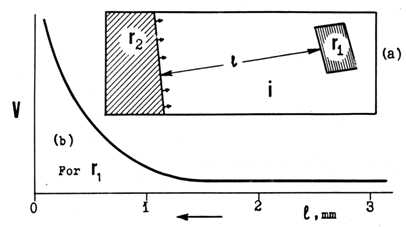
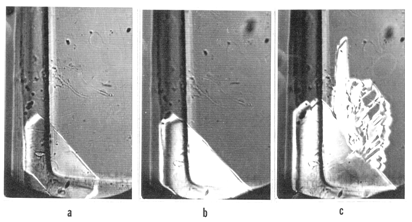
- When there are several H crystals growing from independent nucleation sites in the same L crystal, it can be easily seen that the rates of their growth vary in a wide range. Considering that the initial crystalline matter and ΔTtr are equal for all the growing H crystals, this fact in itself is instructive in regard to kinetics of solid-state phase transitions. Which of these rates does any existing theory account for? There is another phenomenon observed repeatedly: these rates are not quite independent of one another. In one instance, pictured in Fig. 13a, the crystal r1 was almost not growing when a fast-growing interface from r2 began approaching from the opposite end. The latter crystal noticeably activated the growth of the former when the two were still separated by as much as 1.5 mm. As the r2 was coming closer, growth of r1 sharply accelerated (Fig. 13b).
9. Revision of the Activation Energy Concept
- In experimental studies of kinetics of solid-state phase transitions the phase ratio was measured vs. time with the objective to find the "activation energy of phase transition" Ea. The nature of a phase transition is heterogeneous, but Ea was interpreted as the energy barrier to be overcome in the process of a cooperative homogeneous rearrangement of one ideal crystal structure into another. The inconsistence of this approach is conspicuous. Considering that phase transitions between crystal states occur by 3-D nucleation and subsequent growth, there must be at least two activation energies: one for nucleation, the other for rearrangement at the interfaces. The latter process, in turn, involves two major stages: 2-D nucleation of molecular layers and molecular relocation at interfaces. The three basic activation energies that control the above three major stages of a solid-state phase transition are:Ea'. Activation energy of a 3-D nucleus formation. The Ea' depends on the particular structure (size and configuration) of the lattice defect (OM) acting as the nucleation site. The nucleation temperatures encoded in these sites are different, therefore Ea' is not a unique characteristic of a particular phase transition. Rather, it can be of any magnitude greater than E'a,min corresponding to the ΔTtr,min. Absence of even a single OM in the crystal is equivalent to Ea' = ∞. This leads to the conclusion that attempts to find the Ea’ characteristic of a given phase transition would be physically unsound. This activation energy has nothing to do with interface kinetics. Phase transition in a fine-crystalline powder exemplifies the case when the bulk rate of transition under changing temperature is governed exclusively by different Ea’ encoded in the individual particles.Ea". Activation energy of 2-D nuclei formation on a molecular-flat interface. Ea" is not a fixed value either. It varies owing to structural differences (size and shape) of VAs acting as the nucleation sites. The VAs must be present in quantities and located near the interface in order that it could be able to propagate. If this condition is not met, the phase transition (interface motion) will not be possible, which is equivalent to Ea"= ∞. At moderate concentrations of VAs the interface motion is controlled more by the availability of VAs than the E" magnitudes. Different speed of an interface motion at the same temperature is an example of interface kinetics governed by VAs availability. In the case of high VAs concentrations, when only a small part of the available VAs is “consumed" during interface motion, molecular relocation across the interface starts limiting the interface speed.Ea”’. Activation energy of molecular relocation at kinks of a contact interface. As shown in Section 2, the process in question is a “stimulated sublimation". This activation energy is much lower then the previous two and rarely controls the linear kinetics.
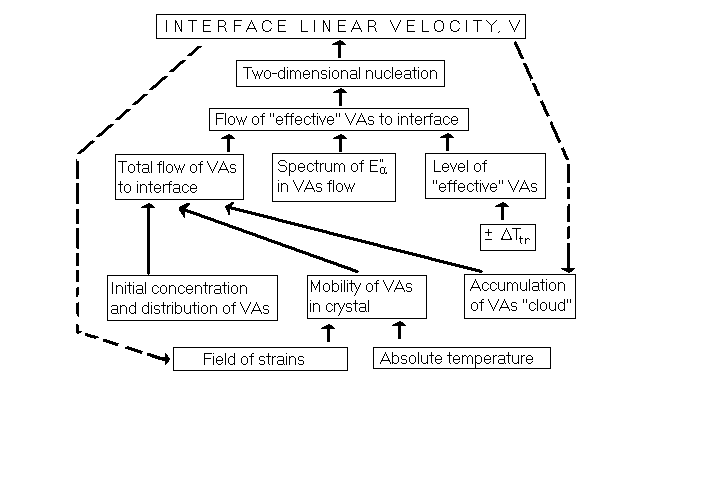
10. Relationships between the Controlling Parameters
- The complications and instabilities of interface kinetics are rooted in feedbacks. An interface needs certain conditions for its motion, but its motion affects these conditions. Flowchart below summarizes relationships between the parameters responsible for the interface kinetics controlled by VAs flow. After the foregoing discussion, the flowchart is self-explanatory even if it may seem cumbersome. Connections between the parameters should be traced from the bottom up following solid-line arrows. The feedbacks that turn the process into autocatalytic are shown by broken lines. The temperature effects are of two kinds. One is ΔTtr, which provides energy gradient for phase transition. The other is absolute temperature - the cause of molecular vibrations and other mobilities. The flowchart illustrates that (1) phase transition in an ideal crystal is not possible and (2) the phenomena of kinetics are complex, multiparameter and irreproducible in spite of the simplicity of the contact mechanism. Yet, the flowchart represents only the simplest case of slow interface motion in a good quality real single crystal and when the accompanying strains are sufficiently small not to create the additional complications described in the next section. There is one more simplification in this flowchart, and it is essential: it does not reflect the phase transition latent heat which can dramatically affect its kinetics - up to explosion in some cases.
11. The “Truly out of Control” Kinetics
- If the previously described interface kinetics may seem "out of control", it still represents the simplest and most orderly case. A smooth advancement of a flat interface takes place only if certain precautions are taken: the specimen is a good small single crystal and ΔTtr is low. It is also helpful if the specific volumes of the polymorphs are close and the crystal has a plate-like shape. Then the strains arising at the slowly moving interface can dissipate before damaging the original crystal medium. If these conditions are not favorable, the crystal growth loses visually orderly character. This disorderly morphology for a century delayed discovery of the underlying phenomenon presented in Section 2: growth of naturally-faced crystals. (Another cause of the delay was not using optical microscopy and transparent single crystals). The phase transition in most instances appears to the observer as a blurred thick "wave" rolling over the crystal and quickly completing the process, leaving behind a less transparent material. What kind of kinetics is that? The X-ray patterns reveal that a single crystal turns into a polycrystal. All facts taken together suggest that the interface generates multiple lattice defects, OMs, acting as the sites for 3-D nucleation immediately in front of itself. This is caused by the strains originating from the fast-moving interface. Because VAs are generated as well, the new growth proceeds quickly in both directions: toward the interface and out of it, creating new strains. Not having time for relaxation they again damage the adjacent lattice. This kinetics is based on the positive feedback:…interface motion à strains à generation of new nuclei in front of interface à growth from those nuclei (interface motion) à strains à …and so on.
12. Solid-State Recrystallization
- The contact mechanism offers an insight into another solid-state reaction − recrystallization (migration of grain boundaries) of polycrystalline solids. It is hard to find any reason to assume one molecular mechanism for interface propagation in phase transitions and another for migration of grain boundaries. The grains in a polycrystal have the same crystal structure, but due to their random orientations the conditions at their boundaries are not different from those at phase transition interfaces. The grain boundaries do migrate. The difference is in their driving forces, namely, to minimize the grain surface energy and / or substitute a more perfect lattice for a less perfect one. They are much weaker, resulting in slower process. In all other respects it is the same crystal growth. Recrystallization in itself is a large topic, a branch of physical metallurgy and some other applied sciences. The contact mechanism tells us in which direction a grain boundary moves, namely, from the grain where it has rational (h,k,ℓ) to the grain where it is irrational. It follows that a major component of the recrystallization driving force is the elimination of irrationally oriented boundaries. But there is more to it. The boundary will migrate only if the two neighboring grains have different orientations. The boundary between two grains of the same orientation will be either equally rational with the same (h,k,ℓ), or equally irrational. In such a case, no driving force to instigate the molecular relocation in one or the other direction exists: Ea is same in either direction. The boundary remains still. In such a straightforward manner the contact mechanism accounts for one of the unexplained "recrystallization laws" which states that the boundary between two grains of the same orientation does not migrate[55].
13. Ferromagnetic Transitions
- Ferromagnetic phase transitions should be added to the list of solid-state reactions (and the same relates to ferroelectrics). Initially everyone believed that they are of the second order − a cooperative phenomenon with strictly fixed ("critical", "Curie") temperature of phase transition. In 1965 Belov wrote in his monograph "Magnetic Transitions"[56] that ferromagnetic and antiferromagnetic transitions are "concrete examples" of secondorder phase transitions. But in 1970's the theorists were puzzled after a number of first-order ferromagnetic phase transitions were reported.. It was not realized that a firstorder phase transition meant nucleation and growth, and not a critical phenomenon. Since then the number of recognized first-order ferromagnetic phase transitions grew dramatically. They turned out to be of the first order even in the basic ferromagnetics − Fe, Ni and Co. This process was accompanied by the increasing realization of structural changes involved. A new term "magnetostructural" transitions appeared and is being used to distinguish them from those not being " structural". There was no explanation why some ferromagnetic phase transitions are "accompanied" by structural change, and others do not. But explanation is simple, although controversial to many (not to this author): all ferromagnetic phase transitions are "structural", meaning they always materialize by nucleation and crystal rearrangements at the interfaces, rather than cooperatively. Moreover, designations of any phase transitions, ferromagnetic or otherwise, as second order were always superficial. Not a single sufficiently documented example exists. Ferroelectric phase transitions also materialize by nucleation and growth.Ferromagnetic phase transition is a structural rearrangement accompanied by (or giving rise to) change of the magnetization. No change in the state of magnetization is possible without the crystal reconstruction. This is a direct consequence of the simple principle that orientation of a spin is imposed by the orientation of its atomic carrier. Therefore, any reorientation of spins requires reorientation of their carriers. The only way to achieve that is replacing the crystal structure. This occurs by nucleation and interface propagation. Everything regarding the nucleation and growth in solid state (in the epitaxial version) is relevant and applicable to ferromagnetic phase transitions. All ferromagnetic phase transitions are "magnetostructural". The term, however, is defective in the sense that it indirectly suggests existence of ferromagnetic phase transitions without structural change. Refer to[1,15] for more details.
14. Magnetization by Interface Propagation
- Magnetization of polydomain crystals is a solid-state reaction as well, whether the driving force is temperature, pressure or applied magnetic field. The conventional theory does not explain why magnetization is realized by propagation of interfaces rather than cooperatively in the bulk. Once again: magnetization is not a spin reorientation in the same crystal structure, it requires turning the atomic / molecular spin carriers. The only way to turn the spin carriers is by crystal rearrangement. The mechanism of crystal rearrangements is nucleation and propagation of interfaces (in this case − polydomain twin boundaries). Possibility of a cooperative magnetization "by rotation" is thus ruled out[1,15].
15. Conclusions
- Phase transitions in solid state were studied under most refined experimental conditions. Rather high-quality small transparent single crystals were used. Perfectly-shaped single crystals of the new phase slowly grown within those crystals were observed. Both parts of the molecular mechanism of their crystal growth - nucleation and interface propagation - were experimentally examined and turned out basically the same as in crystal growth from liquids and gases. The features of that mechanism related to a solid state of the starting material were specified and became the reason for naming it "contact mechanism". The real molecular rearrangement, deduced from the experimental data, was put forward. Its nucleation part, in particular, had nothing in common with the treatment of nucleation in solids by statistical-dynamic theory. The contact mechanism is found to be in good agreement with available data for other reactions in solids. Besides, it is able to coherently account for the irreproducibilities and complications of kinetics of solid-state reactions. A universal molecular mechanism of all reactions in solid state, being it a phase transition, recrystallization, magnetization, etc., is emerging. It is a crystal growth by nucleation encoded in the crystal defects and subsequent molecule-by-molecule relocation at the interfaces. It is universal because it is a case of crystal growth whatever the medium is - this time being a solid state. It is most energy-efficient than statistical-dynamic theories can offer, considering that it needs energy to relocate one molecule at a time rather than myriads molecules at once.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML