-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Condensed Matter Physics
p-ISSN: 2163-1115 e-ISSN: 2163-1123
2013; 3(3): 80-88
doi:10.5923/j.ajcmp.20130303.03
Spectroscopic Properties of Pr3+ in Lithium Bismuth Borate Glasses
Beena Bhatia1, Vishal parihar1, Sanjay Singh2, A. S. Verma3
1Ceramics Laboratories, Department of Physics, Jai Narain Vyas University Jodhpur, 342001, India
2Department of Physics, Shyam Lal College, University of Delhi, Delhi, 110032, India
3Department of Physics, Banasthali Vidyapith, Rajasthan, 304022, India
Correspondence to: A. S. Verma, Department of Physics, Banasthali Vidyapith, Rajasthan, 304022, India.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
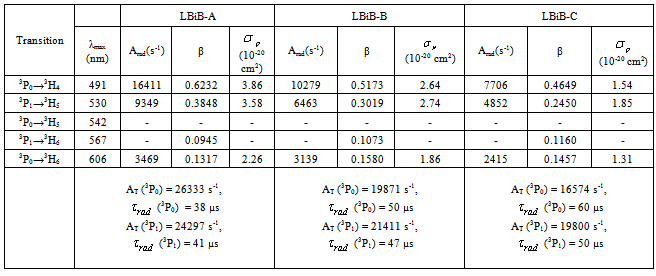
Glasses of the system: xB2O3-10Bi2O3-30Li2O-xPr6O11 where x =1, 1.5 and 2 were prepared by melt quenching technique. Optical absorption and emission spectra have been recorded. The intensities of f-f transitions are calculated in term of Judd-Ofelt (JO) intensity parameters Ωλ (λ = 2, 4 and 6). Using the fluorescence data and these Ωλ parameters, various radiative properties like transition probability (A), branching ratio (βR), radiative lifetime (τR), and stimulated emission cross section (σp), of various emission lines have been evaluated. The values of these parameters indicate that for blue (3P0 → 3H4, 491 nm) transition can be considered to be good laser transition in the visible region.
Keywords: Doped Lithium Bismuth Borate Glasses, Judd-Ofelt Analysis, Optical Properties, Rare Earth Elements
Cite this paper: Beena Bhatia, Vishal parihar, Sanjay Singh, A. S. Verma, Spectroscopic Properties of Pr3+ in Lithium Bismuth Borate Glasses, American Journal of Condensed Matter Physics, Vol. 3 No. 3, 2013, pp. 80-88. doi: 10.5923/j.ajcmp.20130303.03.
Article Outline
1. Introduction
- Rare earth ion doped glasses are often used as lasing materials for glass lasers, light emitting diode, memory device and fiber amplifiers for optical telecommunication. Among the lanthanides ions trivalent Praseodymium (Pr3+) has wide applications such as up-converters [1], fiber amplifiers in 1.3 µm region[2], UV, visible and near infrared lasers[3], electro optic device[4-6] and optical fiber laser[7] due to a large number of available absorption bands in UV-VIS-NIR region. There has been increasing interest in synthesis, structure and physical properties of heavy metal oxide (HMO) glasses such as Bi2O3, PbO, GeO2, GaO2 etc.[8-11]. It has been reported that lithium bismuth borate glasses containing Bi2O3 as the network intermediate(NWI) to the glass network former B2O3 (NWF) possess high refractive index, high infrared transparency , high density, moisture resistant and have extended transmission in mid IR region. The addition of network modifier (NWM) Li2O is to improve both electrical and mechanical properties of such glasses. In the present work, we have studied on the absorption and emission properties of Pr3+ doped lithium bismuth borate glasses. The Judd-Ofelt[12, 13] theory has been applied to compute the intensity parameters Ωλ (λ = 2, 4 and 6). These intensity parameter have been used to evaluate important optical properties such as radiative transition probability for spontaneous emission, branching ratio, radiative life time of the excited state and stimulated emission cross section in order to optimize the best configuration of the rare earth ions- host to improve the laser efficiency of a given electronic transition. To understand the laser efficiency of these materials, the value of spectroscopy quality factor (Ω4/ Ω6) has been evaluated.
2. Experimental
2.1. Glass Preparation
- The following molar compositions with rare earth doped and reference glass samples were prepared by melt quenching technique.
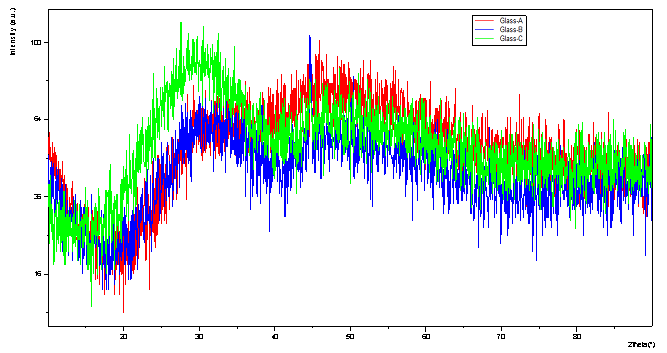 | Figure 1. XRD Pattern of Pr3+ : LBiB glasses |
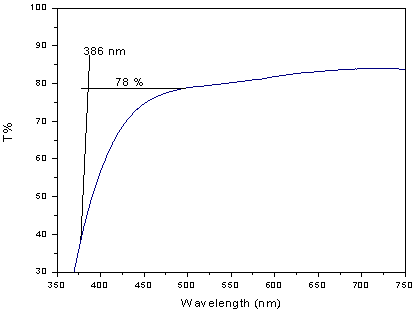 | Figure 2. UV-Vis transmission spectra of LBiB glass |
- The starting materials used in the present work were of AR grade chemicals (H3BO3, Bi2O3, Li2CO3 and Pr6O11) having purity 99.99%. All weighed chemicals were powdered by using an Agate pestle mortar and mixed thoroughly. The raw mixed materials (15 g) were melted in alumina crucibles in silicon carbide based electrical furnace for 2h at 1050℃. The glass formed by quenching the melt on a pre-heated stainless steel mould was immediately transferred to muffle furnace and were annealed at temperature of 300℃ for 60 minutes to remove thermal strains and stresses. Sample of the size 20mm×15mm×1.5mm were cut and polished on all sides to make their faces flat and parallel. Every time fine powder of cerium oxide was used for polishing the samples.The characterization of the specimens was done to ensure the glass formation by X-ray diffraction. It was recorded on PANalytical X′pert Pro MPD diffractometer of CuKα radiation (1.5406 Ǻ) operated at 45 kV and 40 mA with a scanned step size of 0.05o and time for each step 1 sec. in the region of 2θ= 10º to 90º. The absorption spectra of these glasses were recorded between wavelength ranges 400-650 and 900-2250 nm with a Perkin-Elmer Lambda 750 UV/VIS / NIR Spectrophotometer at room temperature. The emission spectra of the glass samples were recorded using Varian Make Cary Eclipse fluorescence spectrophotometer in the spectral range 460-650 nm using the exciting wavelength 445 nm.
2.2. Physical Properties
- By Appling Archimede’s principle, the glass densities were measured with Xylene as an immersion liquid on a single-pan electrical balance to the nearest 0.0001 g. The error in density measurement is estimated to be ±0.004 g cm−3. The refractive indices of these glasses have been measured at λ = 589.3 nm on an Abbe refractometer with an accuracy of ±0.001. The sample being glassy, it requires an adhesive coating on its surface, preferably 1-monobromonaphthalene as the contact layer between the sample and prism of the refractometer by using a sodium vapor lamp.The rare earth ions concentration was calculated [14]
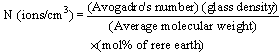 | (1.1) |
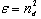 | (1.2) |
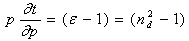 | (1.3) |
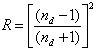 | (1.4) |
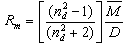 | (1.5) |
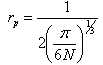 | (1.6) |
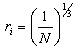 | (1.7) |
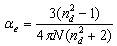 | (1.8) |
|
3. Theoretical
3.1. Nephelauxetic Ratio (β) and Bonding Parameter (b1/2)
- The nature of the R-O bond is known by the Nephelauxetic Ratio (β) and Bonding Parameters (
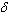 ), which are computed by using following formulae[21]. The Nephelauxetic Ratio is given by
), which are computed by using following formulae[21]. The Nephelauxetic Ratio is given by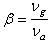 | (1.9) |
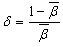 | (2.0) |
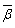 is the average value of
is the average value of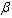 .
.3.2. The Oscillator Strengths
- The intensity of spectral lines are expressed in terms of oscillator strengths using the relation [22].
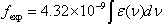 | (2.1) |
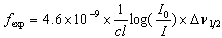 | (2.2) |
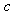 represents the concentration of rare earth ion in the glass and ‘
represents the concentration of rare earth ion in the glass and ‘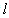 ’ is its optical path length.
’ is its optical path length. 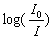 is called optical density,
is called optical density, 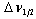 is the half band width. In the present work, the intensities of all the bands are measured by the area method. The experimental and calculated oscillator strengths for Pr3+ ions in lithium bismuth borate glasses are given in Table 2.
is the half band width. In the present work, the intensities of all the bands are measured by the area method. The experimental and calculated oscillator strengths for Pr3+ ions in lithium bismuth borate glasses are given in Table 2.3.3. Judd-Ofelt Parameters
- According to Judd[12] and Ofelt[13] theory, independently derived expression for the oscillator strength of the induced forced electric dipole transitions between an initial J manifold │4f N ( S, L) J> level and the terminal J’ manifold │4fN (S’L’) J’> is given by:
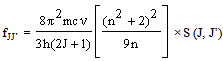 | (2.3) |
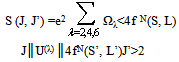 | (2.4) |
|
|
3.4. Radiative Properties
- The Ωλ parameters obtained using the absorption spectral results have been used to predict radiative properties such as spontaneous emission probability (A) and radiative life time (τR), and laser parameters like fluorescence branching ratio (βR) and stimulated emission cross section (σp).The spontaneous emission probability from initial manifold │4f N (S’, L’) J’> to a final manifold │4fN (
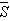 ,
,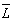 )
)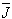 >| is given by:
>| is given by: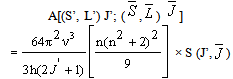 | (2.5) |
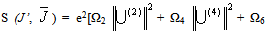
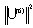 ]For Pr3+ ion, J’ = 0 or 1 and matrix elements of the doubly reduced unit tensor operator reported by P. Babu et al.[25] have been used.The fluorescence branching ratio for the transitions originating from a specific initial manifold │4fN (S', L')J'> to a final manifold
]For Pr3+ ion, J’ = 0 or 1 and matrix elements of the doubly reduced unit tensor operator reported by P. Babu et al.[25] have been used.The fluorescence branching ratio for the transitions originating from a specific initial manifold │4fN (S', L')J'> to a final manifold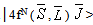 is given by
is given by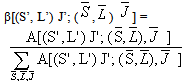 | (2.6) |
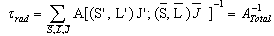 | (2.7) |
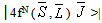 is expressed as
is expressed as 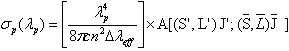 | (2.8) |
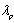 the peak fluorescence wavelength of the emission band and
the peak fluorescence wavelength of the emission band and 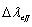 is the effective fluorescence line width.
is the effective fluorescence line width.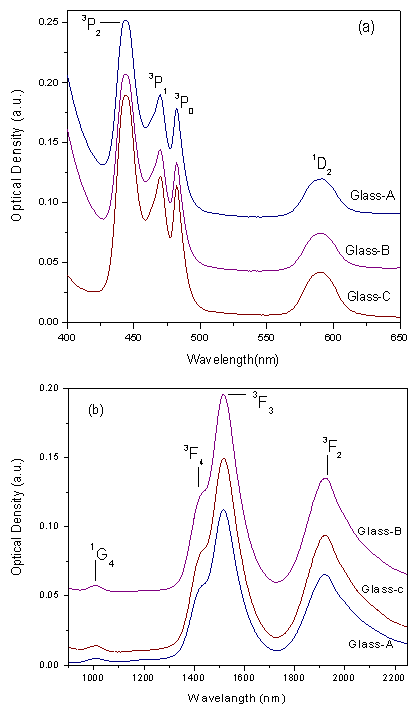 | Figure 3. Optical absorption spectra of Pr3+: LBiB glasses (a) UV-Vis region (b) NIR region |
4. Results and Discussion
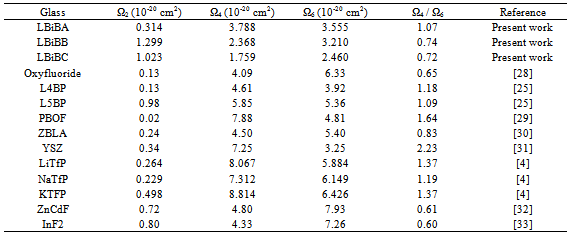
- The amorphous nature of all samples was confirmed by the absence of Bragg’s peak in X-ray diffraction pattern (Fig. 1). The various physical properties of all the glass samples are presented in Table 1. The electronic polarizability (αe), polaron radius (rp), inter ionic distance (ri) slightly decrease with the increase of rare earth ion concentration. The value of nephelauxetic ratio and bonding parameters may be positive or negative indicating covalent or ionic bonding. It is found that all Pr3+ glasses under study have covalent bonding nature. The optical absorption spectra of Pr3+ doped xB2O3-10Bi2O3-30Li2O-xPr6O11 ( where x =1,1.5 and 2 mol%) glass samples are measured at room temperature in the wavelength ranges 400-650 and 900-2250 nm and are shown in Fig. 3. The optical absorption bands around the 3P2 (443nm), 3P1 (469 nm), 3P0 (482 nm), 1D2 (591 nm), 1G4 (1008 nm), 3F4 (1441 nm), 3F3 (1519 nm) and 3F2 (1922 nm), are assigned from the ground state, 3H4. Assignments have been made by published article [26]. The observed absorption bands can be divided into three groups, transition from 3H4 → 1G4 and 3H4→3F4,3,2 in the infrared region, the 3H4→1D2 transition at 591 nm and 3H4→3P2,1,0 complex group of transitions in violet to blue region. From the absorption spectra, experimental oscillator strengths have been calculated for all the absorption bands. Further, Judd–Ofelt intensity parameters Ωλ (λ = 2, 4, and 6) were calculated by using the fitting approximation of the experimental oscillator strengths to the calculated oscillator strengths with respect to their electric dipole contributions. The fairness of the fitting approximation is examined by the root mean square deviations
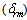 . Low
. Low 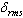 values clearly indicate the accuracy of fitting. The experimental, calculated oscillator strengths and Judd–Ofelt intensity parameters of all Pr3+ lithium bismuth borate glasses are presented and compared with the other glass systems which are listed out in Table 2. From Table 2, it is observed that, for the transition 3H4→3P2 at 443 nm, the oscillator strength is very high compared with the other absorption transitions in all glasses. Thus, the transition 3H4→3P2 is known as the hypersensitive transition and follows the selection rules
values clearly indicate the accuracy of fitting. The experimental, calculated oscillator strengths and Judd–Ofelt intensity parameters of all Pr3+ lithium bismuth borate glasses are presented and compared with the other glass systems which are listed out in Table 2. From Table 2, it is observed that, for the transition 3H4→3P2 at 443 nm, the oscillator strength is very high compared with the other absorption transitions in all glasses. Thus, the transition 3H4→3P2 is known as the hypersensitive transition and follows the selection rules ,
, , and
, and 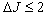 [27]. Table 3 shows that Ω2 increases whereas Ω4 and Ω6 decrease with increase in the Pr6O11 content in host glasses. The order of magnitude of Judd-Ofelt intensity parameter is Ω4< Ω6 < Ω2 for glass-A and Ω6< Ω4 < Ω2 for glass-B and glass-C. These intensity parameters reflect the local structure and bonding in the vicinity of rare earth ions to some extent. As a general conclusion, Ω2 parameter increases with asymmetry of the local structure and with the degree of covalency of the lanthanide-ligand bonds, wherease Ω6 parameter decreases with the tendency of covalency. Ω2 is higer for the present glasses in comparison to oxy-fluoride[28], lithium borate[25], PBOF[29], ZBLA[30], yttria-stabilized-zirconia (YSZ: ZrO2 -Y2O3)[31], tellufluoro-phosphate[4], ZnCdF (ZnF2 –CdF2)[32] and InF2[33]. Weber and Jacob[34] have reported that the ratio (Ω4/Ω6 ) known as the spectroscopic quality factor characterizes the glass concerned. The values of (Ω4/Ω6) for glasses under study are given in Table 3. Glass-A is having larger value of (Ω4/Ω6) than other two glasses and it shows that glass-A is a kind of better optical glass.Fig. 2 shows the transmission spectra of reference glass (LBiB glass) in the UV-Vis range. It is clear from this figure that, the LBiB glass has above 78% transmission in the optical window up to 0.387 µm. Fig. 4 present the excitation spectrum of Pr3+ ions doped glasses, which was measured by monitoring an intense emission at 606 nm. The excitation peaks from the excitation spectrum are assigned electronic transition with ground level 3H4 to higher energy level of Pr3+ , i.e., 3H4→3P0 (491.5 nm). Fig 5 shows the emission spectra of Pr3+: LBiB glasses, the most intense band in the absorption spectra around 3P2 level with 445 nm (diode laser) has been used for the excitation of Pr3+ ions. As a consequence of which, three strong fluorescence bands around 491, 530 and 606 nm, have been observed in the wavelength region 450-650 nm. The very weak fluorescence bands around 542 and 567 nm are not included in the analysis because of the uncertainties in the
[27]. Table 3 shows that Ω2 increases whereas Ω4 and Ω6 decrease with increase in the Pr6O11 content in host glasses. The order of magnitude of Judd-Ofelt intensity parameter is Ω4< Ω6 < Ω2 for glass-A and Ω6< Ω4 < Ω2 for glass-B and glass-C. These intensity parameters reflect the local structure and bonding in the vicinity of rare earth ions to some extent. As a general conclusion, Ω2 parameter increases with asymmetry of the local structure and with the degree of covalency of the lanthanide-ligand bonds, wherease Ω6 parameter decreases with the tendency of covalency. Ω2 is higer for the present glasses in comparison to oxy-fluoride[28], lithium borate[25], PBOF[29], ZBLA[30], yttria-stabilized-zirconia (YSZ: ZrO2 -Y2O3)[31], tellufluoro-phosphate[4], ZnCdF (ZnF2 –CdF2)[32] and InF2[33]. Weber and Jacob[34] have reported that the ratio (Ω4/Ω6 ) known as the spectroscopic quality factor characterizes the glass concerned. The values of (Ω4/Ω6) for glasses under study are given in Table 3. Glass-A is having larger value of (Ω4/Ω6) than other two glasses and it shows that glass-A is a kind of better optical glass.Fig. 2 shows the transmission spectra of reference glass (LBiB glass) in the UV-Vis range. It is clear from this figure that, the LBiB glass has above 78% transmission in the optical window up to 0.387 µm. Fig. 4 present the excitation spectrum of Pr3+ ions doped glasses, which was measured by monitoring an intense emission at 606 nm. The excitation peaks from the excitation spectrum are assigned electronic transition with ground level 3H4 to higher energy level of Pr3+ , i.e., 3H4→3P0 (491.5 nm). Fig 5 shows the emission spectra of Pr3+: LBiB glasses, the most intense band in the absorption spectra around 3P2 level with 445 nm (diode laser) has been used for the excitation of Pr3+ ions. As a consequence of which, three strong fluorescence bands around 491, 530 and 606 nm, have been observed in the wavelength region 450-650 nm. The very weak fluorescence bands around 542 and 567 nm are not included in the analysis because of the uncertainties in the  determination.
determination.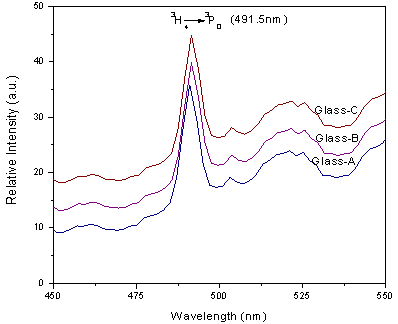 | Figure 4. Excitation spectrum of Pr3+: LBiB glasses |
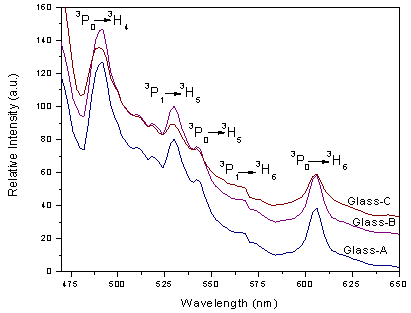 | Figure 5. Emission spectrum of Pr3+: LBiB glasses |
5. Conclusions
- Optical absorption and fluorescence analyses of Pr3+ doped lithium bismuth borate glasses have been performed. The Judd–Ofelt model has been applied to calculate the JO intensity parameters Ωλ (λ = 2, 4, and 6) from the measured oscillator strength of the absorption spectra of Pr3+: LBiB glasses. From the magnitudes of the bonding parameter δ, for Pr3+ ions in various glasses, it is found that Pr3+ ions have a covalent character in all the glasses. The spectroscopic quality factor (Ω4/Ω6), branching ratio (
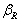 ) and the stimulated emission cross section (σp) values are calculated for present glasses. It could be observed that glass-A possess better values compared to the other two glass (B and C) system. The large stimulated emission cross section in lithium bismuth borate glasses suggests the possibility of utilizing these systems as laser materials. Thus, we could suggest that Pr3+: LBiB glasses are the suitable potential candidates for laser materials for blue (3P0 → 3H4, 491 nm) wavelength.
) and the stimulated emission cross section (σp) values are calculated for present glasses. It could be observed that glass-A possess better values compared to the other two glass (B and C) system. The large stimulated emission cross section in lithium bismuth borate glasses suggests the possibility of utilizing these systems as laser materials. Thus, we could suggest that Pr3+: LBiB glasses are the suitable potential candidates for laser materials for blue (3P0 → 3H4, 491 nm) wavelength.ACKNOWLEDGEMENTS
- The authors are thankful to Prof. S.S.L. Surana, Jai Narain Vyas University, Jodhpur (Raj.) for his help in optical absorption measurements.
References
| [1] | D.B. Gatch, S.A. Holmstrom, and W.M. Yen, J. Lumin. 83-84 (1999) 55. |
| [2] | Y. Zhao, S. Fleming, IEEE J. Quant. Electron. 33 (1997) 905. |
| [3] | A.A. Kaminskii, Laser Crystals, 2nd Edition, Springer, Berlin 1990. |
| [4] | R. Moorty, M. Jagasin, Mater. Chem. Phys. 93 (2005) 455. |
| [5] | Y.K. Sharma, S.P. Tandon, S.S. Surana, Mater. Sci. Eng. B 77 (2000) 167. |
| [6] | Y.C. Ratnakaram, A.V. Kumar, D.T. Naidu, N.O. Gopal, Mater.Lett. 58 (2004) 3908. |
| [7] | D.M. Baney, L. Yang, J.Ratcliff, K.W. Chang, Electron. Lett. 31 (1995) 1842. |
| [8] | C.H. Kam and S. Buddudu, J. Quant. Spectrosc. Radiant. Trans 325-337 (2004) 87. |
| [9] | G.A. Kumar, A. Martinez, E. Mejia and C.G. Eden, J. Alloys. Comp. 117-120 (2004) 365. |
| [10] | E. Culea, L. Pop and S. Simoon, Mater. Sci. Engg. 59-63 (2004) B 112. |
| [11] | J. Yang, S. Dai, Y. Zhou, Leiwen. Lili Hu and Z.H. Jiang, Appl. Phys. 977-983 (2003) 93 (2). |
| [12] | B.R. Judd, Phys. Rev. 127 (1962) 750. |
| [13] | G.S. Ofelt, J. Chem. Phys. 37 (1962) 511. |
| [14] | A. Shankar, A. Dasgupta, B. Babsu, A. Paul, J. Mater. Sci. Lett. 4 (1983) 697. |
| [15] | B. Bendow, P. K. Banerjee, M.G. Drexhage and J. Lucas, J. Am. Ceram. Soc. 65 (1985) C 92. |
| [16] | J. Schroeder, J. Non-Cryst. Solids 40 (1980) 549. |
| [17] | Y. Ohisti, S.Mitachi and T. Tanabe, Phys. Chem. Glasses 24 (1983) 135. |
| [18] | J.E. Shelby, and J. Ruller, Phys. Chem. Glasses 28 (1987) 262. |
| [19] | M.M. Ahmed, C.A. Hogarth and M.N. Khan, J. Mater. Sci. Lett. 19 (1984) 4040. |
| [20] | A. Klonkowski, J. Non-Cryst. Solids 72 (1985) 117. |
| [21] | S.P. Sinha, in: Complexes of the rare earths. Oxford: Pergamon Press; 1996. |
| [22] | C. Gorller-Walrand, K. Binnemans, Spectral intensities of f-f transition, in: K.A. Gschneidner Jr., L. Eyring (Eds.), Handbook on the physics and Chemistry of rare earths, Vol. 25, North-Holland. Amsterdom, 1998 (Chapter 167). |
| [23] | Y.K. Sharma, S.S.L. Surana and R. K. Singh, J. of Rare Earth. 773 (2009) 27 (5). |
| [24] | C.H. Goublen, Mathods of Statistical Analysis, Asian Publishing House, Bombay, Ch. 8 (1964) 138. |
| [25] | P. Babu and C.K. Jayasankar, Physica B 326-340 (2001) 301. |
| [26] | I. Pal, A. Agarwal, S. Sanghi, M.P. Aggarwal, J. Alloys comp. 7625-7631 (2011) 509. |
| [27] | S. Tanabe, T. Ohyagi, N. Soga, T. Hanada, Phys. Rev. B 46 (1992) 3305.Za |
| [28] | R.T. Genova, I.R. Martine, U.R.R. Mendoza, F. Lahoz, A.D.L. Garrin, P. Nunez, J.G. Platas, V. Lavine, J. Alloys Compd. 380 (2004) 167. |
| [29] | P. Nachimuthu, R. Jagannathan, Phys. Chem. Glasses 36 (1995) 77. |
| [30] | M. Eyal, E. Greenberg, R. Reisfeld, Chem. Phys. Lett. 117 (1985) 108. |
| [31] | B. Savoini, J.E. Munoz Santiuste, R. Gonzalez, Phys. Rev. B 56 (1997) 5856. |
| [32] | M.A. Bunuel, R. Cases, M.A. Chamarro, R. Alcala, Phys. Chem. Glasses 33 (1992) 16. |
| [33] | A.B. Arauzo, R. Cases, R. Alcala, Phys. Chem. Glasses 35 (1994) 202. |
| [34] | R.R. Jacobs, M.J. Weber, IEEE J. Quant. Electron. 12 (1976) 102. |
| [35] | G.H. Dieke, Spectral and Energy Level of Rare Earth Ions in Crystal, Interscience, New York, 1968. |
| [36] | Q. Nie, X. Li, S. Dai, T. Xu, Z. Jin,X. Zhang, Spectrochim Acta A 70 (2008) 537. |
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML
 )and calculated (
)and calculated ( ) oscillator strength of Pr3+ ions in lithium bismuth borate glasses
) oscillator strength of Pr3+ ions in lithium bismuth borate glasses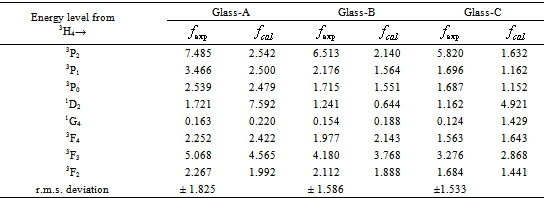
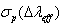 ), total radiative transition probability (AT), radiative life time (ɽr) and total emission cross-section(
), total radiative transition probability (AT), radiative life time (ɽr) and total emission cross-section(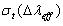 ) for various transitions in Pr3+ doped LBiB glasses
) for various transitions in Pr3+ doped LBiB glasses