-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Condensed Matter Physics
p-ISSN: 2163-1115 e-ISSN: 2163-1123
2011; 1(1): 1-7
doi: 10.5923/j.ajcmp.20110101.01
Structural, Thermal and Magnetic Characterization of Ni-Mn-Ga Ferromagnetic Shape Memory Alloys
R. Jesintha Rani 1, 2, R. Senthur Pandi 1, S. Seenithurai 1, S. Vinodh Kumar 1, M. Muthuraman 1, M. Mahendran 1
1Smart Materials Lab, Department of Physics, Thiagarajar College of Engineering, Madurai, 625015, India
2Department of Physics, Namakkal Kavignar Ramalingam Government Arts College for Women, Namakkal, 637001, India
Correspondence to: M. Mahendran , Smart Materials Lab, Department of Physics, Thiagarajar College of Engineering, Madurai, 625015, India.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
Ni-Mn-Ga Ferromagnetic Shape Memory Alloys (FSMA) are highly interested in industrial actuators and energy harvesters because of its uniqueness in microstructure and multiple phases of these materials at different temperatures. The type of crystal structure, lattice parameters and magnetic properties depend on the alloy composition. The transformation and crystallographic properties are usually mapped to average number of valence electrons per atom. The present work deals with the fundamental characterization of single crystal and polycrystalline nature of these alloys. Though, the polycrystal shows low magnetic field induced stain (MFIS), the easy preparation techniques and cost effective aspects compared with the single crystals makes an impressive interest in dealing with that. The structural, thermal and magnetic parameters are observed and compared for these single and polycrystals.
Keywords: Ni-Mn-Ga Polycrystals, Smart Materials, Magnetic Properties, Phase Transformations, Magnetic Field Induced Stain, Sensors And Actuators
Cite this paper: R. Jesintha Rani , R. Senthur Pandi , S. Seenithurai , S. Vinodh Kumar , M. Muthuraman , M. Mahendran , "Structural, Thermal and Magnetic Characterization of Ni-Mn-Ga Ferromagnetic Shape Memory Alloys", American Journal of Condensed Matter Physics, Vol. 1 No. 1, 2011, pp. 1-7. doi: 10.5923/j.ajcmp.20110101.01.
Article Outline
1. Introduction
- Ferromagnetic Shape Memory Alloys (FSMAs) are a new class of active materials that have generated considerable interest among the scientific community. They are next generation to Shape Memory Alloys (SMAs), and they share many common properties with SMAs. Conventional SMAs are slow, especially in cooling, thus limiting their efficiency in applications. On the other hand, FSMAs show larger Magnetic Field-Induced Strain (MFIS) with quick response at low frequencies than the other active materials such as piezoelectric and magnetostrictive materials[1]. Two alloy systems have predominantly demonstrated field-induced strain, 0.5%[2] in Fe-Pd and 6-10%[3-7] in Ni-Mn-Ga. Ni-Mn-Ga FSMA is focused among the different types of active materials because of their large field induced strain[8,9]. They show upto 10% strain response to an applied magnetic field by the twin boundary motion present in the martensite phase[7]. This is the main advantage of the material; it would overcome the ‘frequency bound’ of the conventional SMAs. Both the magnetic shape memory effect and the mechanical properties of single crystal Ni-Mn-Ga alloys vary significantly due to strong dependence on crystal structure,material composition, initial microstructure, and variant volume fraction[10-30]. Material composition plays a critical role in the determination of the martensite to austenite transformation temperatures. It has been observed in particular that the martensite start temperature of Ni-Mn-Ga is sensitive to composition[9,13,15]. For practical applications of Ni-Mn-Ga, it is necessary to use material compositions that place the austenite to martensite transformation temperature well above the expected operating temperature. This is important because the effects of martensite crystal structure, temperature and maximum strain capability are all interrelated. Material composition must be considered as an important parameter for practical applications. The present work is tried to explain about the composition significance in structural, thermal and magnetic properties of these alloys in single crystal as well as polycrystal.
2. Experimental Procedure
2.1. Single Crystal Preparation
- The single crystal used for this work is received from Prof. Robert C. O’Handley, MIT, USA. Usually the Ni-Mn-Ga single crystal is prepared by the Bridgman technique. High-purity nickel, manganese and gallium are arc melted into buttons and then drop cast into a chilled copper mould. As-cast ingots are placed in an alumina crucible and heated at 1350℃ for one hour for homogenization. The crucible is then withdrawn from the hot zone at a minimum rate per hour. In order to reduce the evaporation of manganese during the crystal growth, the furnace is back-filled to a positive pressure of 6.8 x 105Pa with purified argon gas after the chamber has been out gassed at 1350℃ under vacuum. A piece is cut along the length from the crystal which is heat treated at 900℃ for 24 hrs in a purified argon atmosphere. The samples are wire-cutted for various characterizations.
2.2. Polycrystal Preparation
- The polycrystalline ingots are produced using an arc-melting furnace. The arc melting furnace consists of a copper plate as hearth, which is maintained at room temperature by water circulation. The grooves in the plate are used as containers for melting the raw materials. A vacuum of up to 10-3m.bar is created inside the chamber and flushed with high pure argon gas before melting process. During melting, the chamber is completely filled with argon gas. The intensity of the arc for melting the samples can be reached by increasing the power to the Tungsten electrode. The raw materials of 4N purity are weighed in the stoichiometric ratio. The materials in the chunk and granules form are used for melting. As the powder sometimes flies out of the copper plate due to the force of arc, shifting the stoichiometry, the powder is made into granules for melting. After the first melt, all the ingots are reverted to their same place and melted again in the argon atmosphere. This melting procedure is repeated up to 6 times to ensure a good homogeneity in the sample.After melting, the samples are sealed in quartz ampoules filled with argon atmosphere. Thermal treatment is carried out in a box furnace with 1100℃ for 10 days for the phase transformation. The samples are kept in a furnace for a long time to form the required phase structure. Quenching process is done to stabilize the phase. Table 1 shows the different compositions of Ni-Mn-Ga single and polycrystals.
3. Characterization of Ni-Mn-Ga Alloys
3.1. Structural Studies
- X-Ray Diffraction (XRD) pattern is used to study the structure of the prepared samples. The samples are crushed into powders. Then the crushed Ni-Mn-Ga powders are annealed in an inert atmosphere at 500-700℃ for 3 hours to remove the residual stresses imparted during the crushing of the samples. The powder X-ray diffraction data are collected at room temperature using an 18 kW copper rotating anode-based powder diffractometer fitted with a graphite monochromator in the diffracted beam. The data collection in the 2θ range of 10-80° is carried out using scintillation counter with the scan rate of 2°/min. Figure 1 & 2 shows the XRD pattern for Ni-Mn-Ga single crystals.
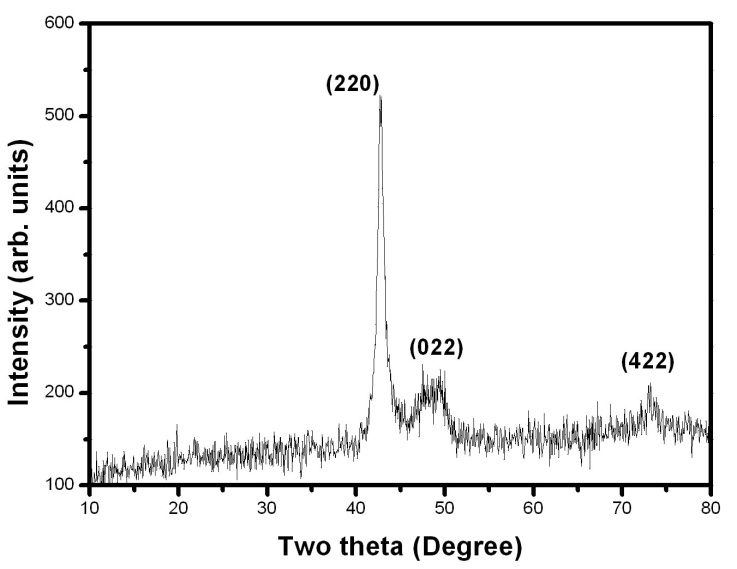 | Figure 1. X-ray diffraction pattern of the single crystal 1. The reflectiopeaks are indexed in the figure |
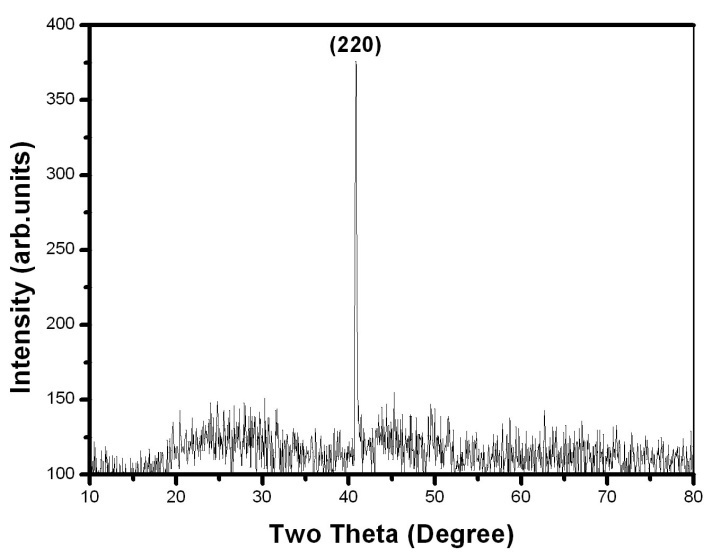 | Figure 2. X-ray diffraction pattern of the single crystal 2. The observed dominant peak is indexed as (220) |
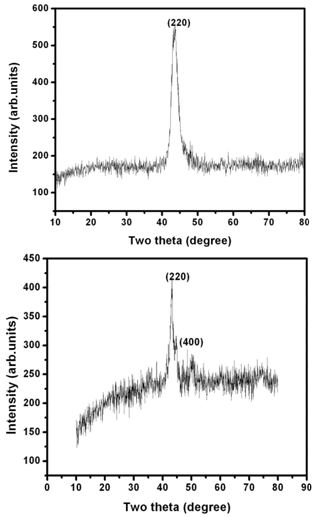 | Figure 3&4. X-ray diffraction pattern of the polycrystal 1 and 2 respectively. The The reflection peaks are indexed in the figure |
| ||||||||||||||||||||||||||||||||||||
3.2. Thermal Studies
- Whenever the phase transformation occurs from martensite to austenite, energy is released or absorbed. This can be measured by Differential Scanning Calorimetry (DSC). The martensite transformation strongly depends on the composition. A small change in the composition may lead to a large shift in the transformation temperature. But the Curie temperature is less sensitive to the composition. Figure 5 is taken for the Ni-Mn-Ga single crystals. The graph is plotted between the heat flow and temperature. The graph shows first order (martensite) and second order (magnetic) transformation temperatures of the sample. In single crystal 1, the martensite finish temperature (Mf) starts in the temperature region of 308 K. The peak has some fluctuations. These fluctuations may be due to the existence of allotropic transformation in the solid-state.
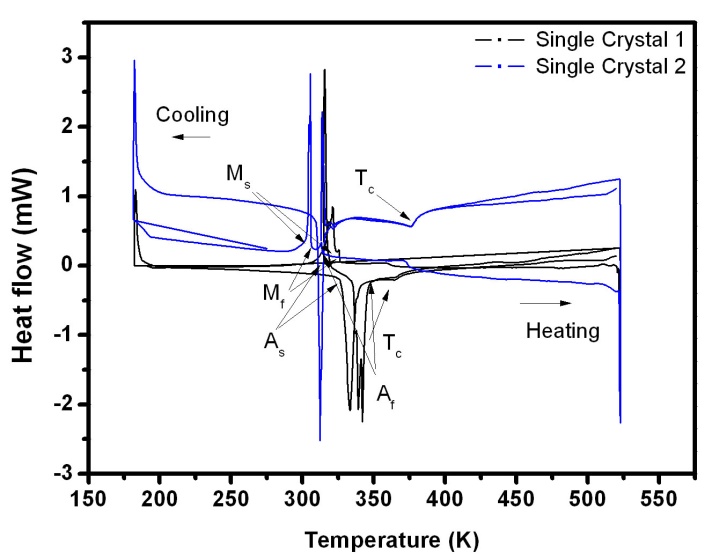 | Figure 5. Thermal transformation studies of the Ni-Mn-Ga single crystals. The results are obtained from the DSC |
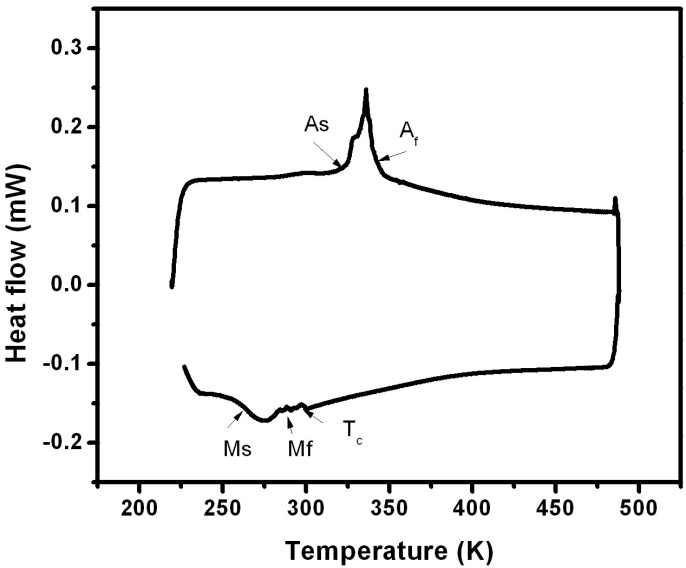 | Figure 6. Thermal transformation studies of Ni-Mn-Ga polycrystal 1 |
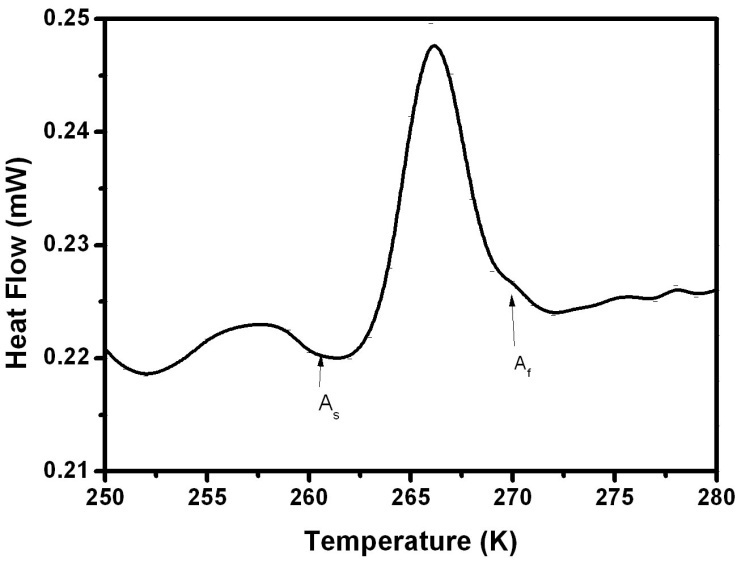 | Figure 7. Thermal transformation of Polycrystal2 during the heating process |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
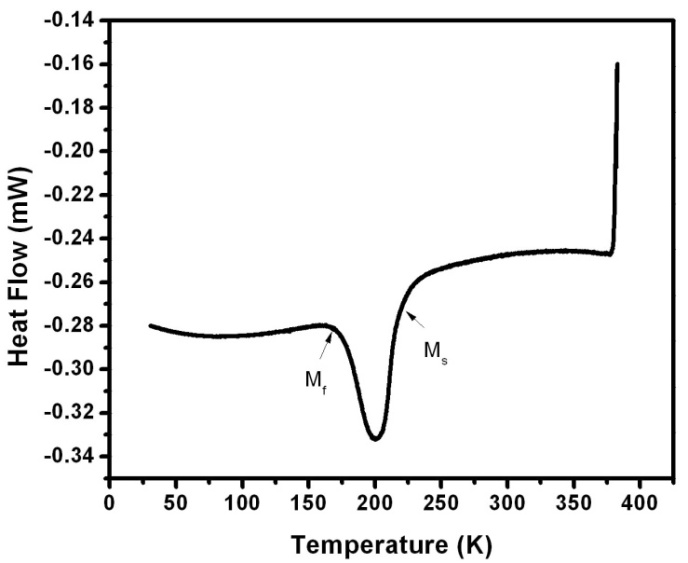 | Figure 8. Thermal transformation studies of the Ni-Mn-Ga polycrystal 2. The result shows the cooling curve obtained from the DSC |
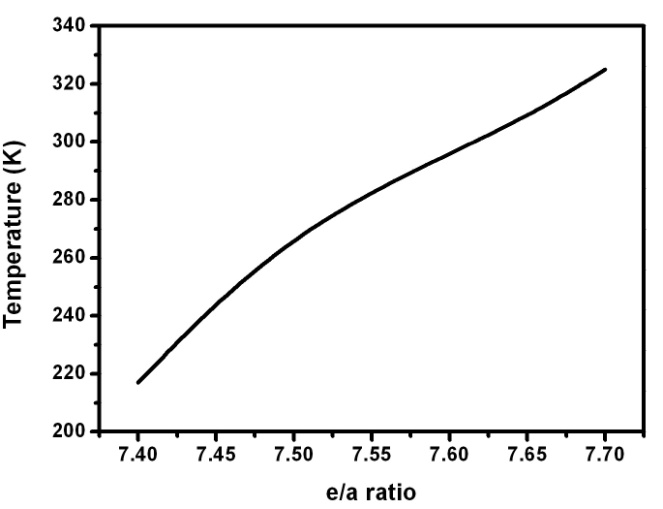 | Figure 9. The thermal transformation temperature is correlated with the e/a ratio of the prepared compositions |
3.3. Magnetic Studies
- The magnetic characteristics of the single crystal and polycrystal alloys are studied through the Vibrating Sample Magnetometer (VSM). The isothermal magnetization as a function of field at room temperature is done for the alloys. The magnetic moment Vs Magnetization curve is plotted for the prepared alloys. The magnetic studies of the single crystals are shown in figure 10. The saturation magnetization of the single crystal alloys is comparatively lower than the polycrystal. It is because of the tendency to align the domains in the field is comparatively good in the single crystals. The very low coercivity of the crystal should be useful for their actuator applications. The crystal structure of single crystals plays a vital role in determining the magnetic moment of the material. In the L21 ordered structure, Mn atom possess a large magnetic moment of 3-4µB. In this structure, ferromagnetic coupling between two Mn atom is taking place when the Mn occupies the ‘b’ site with high atomic order. However, anti-ferromagnetic coupling will occur if the Mn atom occupy a wrong sites (usually Mn atom occpies d site) in an atomically disordered case. The magnetic parameters are tabulated in the Table 4.
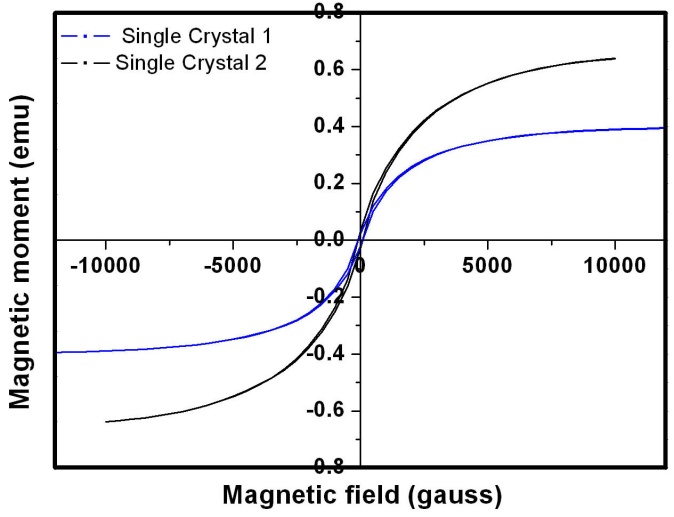 | Figure 10. Magnetization curve for SC1 and SC2 recorded by VSM |
|
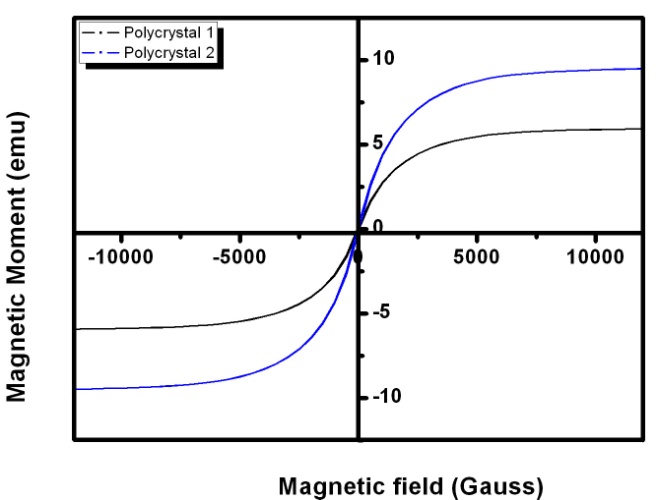 | Figure 11. Magnetization curve of PC1 and PC2 recorded by VSM |
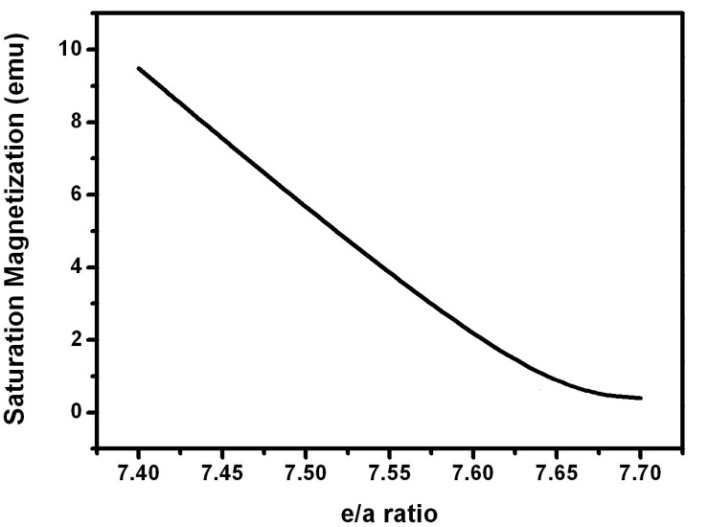 | Figure 12. Magnetic saturation against the e/a ratio of the prepared alloys |
4. Conclusions
- Structural, thermal and magnetic studies are carried out for Ni-Mn-Ga Single and Poly Crystals. The structural study confirms the presence of the martensite structure in the single crystal alloys and the austenite phase in the polycrystal alloys. The thermal measurements evidence the phases present in the alloys with their transformation temperatures. The room temperature martensite structure is confirmed and the transition temperature is calibrated through DSC data. The magnetic nature of the alloys is identified through VSM studies. The magnetic parameters such as saturation magnetization, coercivity and remanance are identified and analyzed with e/a ratio. The mechanical studies and ferromagnetic shape memory effect of these crystals have to be studied in the near future.
ACKNOWLEDGEMENTS
- MM thanks Dr. Robert C.O’Handley for introducing him to the subject. The authors acknowledges the UGC, New Delhi and UGC- DAE CSR, Indore for providing financial support.
References
| [1] | Marioni, M. A., O’ Handley, R.C., and Allen, S.M., 2003, Pulsed magnetic field-induced actuation of Ni-Mn-Ga single crystals, Applied Physics Letters, 83, 3966-3968 |
| [2] | James, R. D., Tickle, R., and Wutig, M., 1999, Large field-induced strains in ferromagnetic shape memory materials, Materials Science and Engineering A, 273-275, 320-325 |
| [3] | Likhachev, A. A., and Ullakko, K., 2000, Magnetic-field-controlled twin boundaries motion and giant magneto-mechanical effects in Ni-Mn-Ga shape memory alloy, Physics Letter A, 275, 142-151 |
| [4] | Murray, S. J., Marioni, M., Allen, S. M., O’Handley, R. C., and Lograsso, A.T., 2000, 6% magnetic-field-induced strain by twin-boundary motion in ferromagnetic Ni–Mn–Ga, Applied Physics Letters, 77, 886-888 |
| [5] | Heczko, O., Sozinov, A., and Ullakko, K., 2000, Giant field-induced reversible linear strain in magnetic shape memory NiMnGa at room temperature, IEEE Transactions on Magnetic, 36, 3266-3268 |
| [6] | Sozinov, A., Likhachev, A. A., Lanska, N., and Ullakko, K., 2002, Giant magnetic-field-induced strain in NiMnGa seven-layered martensitic phase, Applied Physics Letters, 80, 1746-1748 |
| [7] | Müllner, P., Chernenko, V. A., and Kostorz, G., 2004, Large cyclic magnetic-field-induced deformation in orthorhombic (14M) Ni-Mn-Ga martensite, Journal of Applied Physics, 95, 1531-1-6 |
| [8] | Kokorin, V. V., and Chernenko, V. A., 1989, Martensitic transformation in a ferromagnetic Heusler alloy, Physics of Metals and Metalography, 68, 111-115 |
| [9] | Chernenko, V. A., Amengual, A., Cesari, E., Kokorin, V. V., and Zasimchuk, I. K., 1995, Thermal and magnetic properties of stress-induced martensites in Ni–Mn–Ga alloys, Journal de Physique IV, 5 (C2), 95–98 |
| [10] | Chokkalingam, R., Senthur Pandi, R., Vallal Peruman, K., et al., 2010, Shape memory behavior of Ni-Mn-Ga ferromagnetic shape memory alloy, Proc. of ICPEFM, CP1313, 199-201 |
| [11] | Vallal Peruman, K., Mahendran, M., Seenithurai, S., Chokkalingam, R., Singh, R.K., and Chandrasekaran, V., 2010, Internal stress dependent structural transition in ferromagnetic Ni–Mn–Ga nanoparticles prepared by ball milling, Journal of Physic sand Chemistry of Solids, 71, 1540-1544 |
| [12] | Pushpanathan, K., Chokkalingam, R., Senthur Pandi, R., and Mahendran, M., 2011, Effect of annealing on transformation temperature and magnetization in the Ni–Mn–Ga alloy, Materials and Manufacturing Process, 26, 223-229 |
| [13] | Pons, J., Chernenko, V. A., Santamarta, R. and Cesari, E., 2000, Crystal structure of martensitic phases in Ni-Mn-Ga shape memory alloys, Acta Materialia, 48, 3027-3038 |
| [14] | Vallal Peruman, K., Chokkalingam, R., and Mahendran, M., 2010, Annealing effect on phase transformation in nano structured Ni-Mn-Ga ferromagnetic shape memory alloy, Phase Transitions, 83, 509-517 |
| [15] | Malla, A., Dapino, M., Lograsso, T. and Schlagel, D., 2003, Effect of composition on the magnetic and elastic properities of shape-memory NiMnGa, Proc. of SPIE., 5053, 147-158 |
| [16] | Richard, M., Feuchtwanger, J., Schlagel, D., Lograsso, T., Allen, S. M. and O'Handley R. C., 2006, Crystal structure and transformation behaviour of Ni-Mn-Ga martensites. Scripta Materialia, 54, 1797-1801 |
| [17] | Vallal Peruman, K., Mahendran, M., and Seenithurai, S., 2010, Effect of Mn concentration on the phase transformation in Ni–Mn–Ga single crystal, Physica B, 405, 1770-1774 |
| [18] | Yanwei Ma, Awaji, S., Watanabe, K., Matsumoto, M., and Kobayashi, N., 2000, X-ray diffraction study of the structural phase transition of Ni2MnGa alloys in high magnetic fields. Solid State Communications, 113, 671–676 |
| [19] | Rama Rao, N.V., Gopalan, R., Manivel Raja, M., Arout Chelvane, J., Majumdar, B., and Chandrasekaran, V., 2007, Magneto structural transformation studies in melt spun Ni-Mn-Ga, Scripta Materialia, 56, 405-408 |
| [20] | Singh, R.K., and Gopalan, R., 2008, Thermal, structural and magnetic characterization of Ni–Mn–Ga sheets fabricated by powder in tube roll bonding technique, Materials Science and Engineering B, 151, 199-204 |
| [21] | Wirth, S., Leithe-Jasper, A., Vasil’ev, A. N., and Coey, J. M. D., 1997, Structural and magnetic properties of Ni2MnGa, Journal of Magnetism and Magnetic Materials, 167, L7-L11 |
| [22] | Jin, X., Marioni, M., Bono, D., Allen, S.M., O’ Handley, R.C., and Hsu, T.Y., 2002, Empirical mapping of Ni–Mn–Ga properties with composition and valence electron concentration, Journal of Applied Physics, 91, 8222-8224 |
| [23] | Tanja Graf, Claudia Felser, and Parkin Stuart, S.P., 2011, Simple rules for understanding Huesler compounds, Progress in Solid State Chemistry, 39, 1-50 |
| [24] | Xuan, H.C., Ma, S.C., Cao, Q.Q., Wang D.H., and Du, Y.W., 2011, Martensitic transformation and magnetic properties in high-Mn content, Mn50Ni50−xInx ferromagnetic shape memory alloys, Journal of Alloys and Compounds, 509, 5761–5764 |
| [25] | Singh, R.K., Shamsuddin, M., Gopalan, R., Mathur, R.P., and Chandrasekaran, V., 2008, Magnetic and structural transformation in off-stoichiometric NiMnGa alloys, Materials Science and Engineering A, 476, 195–200 |
| [26] | Babita Ingale, Gopalan, R, Rajasekhara, M., and Ramb, S., 2009, Studies on ordering temperature and martensite stabilization in Ni55Mn20−xGa25+x alloys, Journal of Alloys and Compounds, 475, 276–280 |
| [27] | Morawiec, H., Goryczka, T., Seguib, C., Gigla, M., and Szczeszek, P., 2008, Martensitic transformation in an inhomogeneous off-stoichiometric Ni–Mn–Ga alloy, Materials Science and Engineering A, 481–482, 288–292 |
| [28] | Kanomata, T., Yasuda, T., Sasaki, S., Nishihara, H., Kainuma, R., Ito, W., Oikawa. K., Ishida, K., Neumann. K.U., and Ziebeck, K.R.A., 2009, Magnetic properties on shapememory alloys Ni2Mn1+xIn1-x, Journal of Magnetism and Magnetic Materials, 321, 773–776 |
| [29] | Satish Kumar, A., Ramudu, M., and Seshubai, V., 2011, Effect of selective substitution of Co for Ni or Mn on the superstructure and microstructural properties of Ni50Mn29Ga21, Journal of Alloys and Compounds, 509, 8215– 8222 |
| [30] | Soderberg, O., Aaltio, I., Ge, Y., Heczko, O., and Hannula, S.P., 2008, Ni–Mn–Ga multifunctional compounds, Materials Science and Engineering A, 481–482, 80–85 |
| [31] | Annadurai, A., Nandakumar, A. K., Jayakumar, S., Kannan, M. D., Manivel Raja, M., Bysak, S., Gopalan, R., and Chandrasekaran, V., 2009, Composition, structure and magnetic properties of sputter deposited Ni–Mn–Ga ferromagnetic shape memory thinfilms, Journal of Magnetism and Magnetic Materials, 321, 630–634 |
| [32] | Scheerbaum, N., Kraus, R., Liu, J., Skrotzki, W., Schultz, L., and Gutfleisch, O., 2012, Reproducibility of martensitic transformation and phase constitution in Ni–Co–Al, Intermetallics, 20, 55-62 |
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-Text HTML
Full-Text HTML