-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Biomedical Engineering
p-ISSN: 2163-1050 e-ISSN: 2163-1077
2026; 14(1): 9-15
doi:10.5923/j.ajbe.20261401.02
Received: Jan. 31, 2026; Accepted: Feb. 19, 2026; Published: Feb. 24, 2026

Synthesis and Characterization of Hydroxyapatite from Dolomite-based Source for Bone Regeneration
Chinenye Appolonia Ibekwe 1, Grace Modupe Oyatogun 1, Temitope Ayodeji Esan 2, Elizabeth Obhioneh Oziegbe 3
1Department of Materials Science and Engineering, Faculty of Technology Obafemi Awolowo University, Ile-Ife, Osun State, Nigeria
2Department of Restorative Dentistry, Faculty of Dentistry, Obafemi Awolowo University, Ile-Ife, Osun State, Nigeria
3Department of Child Dental Health, Faculty of Dentistry, Obafemi Awolowo University, Ile-Ife, Osun State, Nigeria
Correspondence to: Chinenye Appolonia Ibekwe , Department of Materials Science and Engineering, Faculty of Technology Obafemi Awolowo University, Ile-Ife, Osun State, Nigeria.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This study synthesized and characterized hydroxyapatite (HAp) from Nigerian-based dolomite with the view to investigating its suitability for a scaffold development for bone regeneration. The dolomite was pulverized and subsequently calcined at 900°C to obtain calcium oxide, which was used to synthesize HAp by the wet chemical precipitation method. Furthermore, the chemical characterization of pulverized dolomite and HAp was done using X-ray Fluorescence (XRF) and X-ray Diffraction (XRD) Spectroscopy techniques. In addition, Fourier Transform Infrared (FT-IR) was used to examine the functional group of HAp while the morphological characterization was achieved by Scanning Electron Microscopy (SEM) technique. Moreover, the XRD and XRF results confirmed the presence of calcite and magnesium in dolomite while they also confirmed HAp in synthesized dolomite-based HAp, which was supported by the FT-IR spectrum that showed the presence of phosphate compound with peaks at 1122.6, 1041.6, 991.4, 605.7 and 559.5 cm-1 while the presence of hydroxyl was revealed with peak 3468.1 cm-1. Furthermore, the XRF results showed that the Ca/P molar ratio was more than 1.67, which was close to that of natural bone. The SEM micrograph displayed agglomerated rod structures that resulted in a rough surface, which will facilitate the adhesion and proliferation of bone cells. The study, therefore, affirmed that the dolomite-based hydroxyapatite has similar characteristics to bone hydroxyapatite and may be suitable for the scaffold fabrication for bone regeneration.
Keywords: Bone regeneration, Dolomite, Hydroxyapatite and scaffold
Cite this paper: Chinenye Appolonia Ibekwe , Grace Modupe Oyatogun , Temitope Ayodeji Esan , Elizabeth Obhioneh Oziegbe , Synthesis and Characterization of Hydroxyapatite from Dolomite-based Source for Bone Regeneration, American Journal of Biomedical Engineering, Vol. 14 No. 1, 2026, pp. 9-15. doi: 10.5923/j.ajbe.20261401.02.
Article Outline
1. Introduction
- Orthopedic and dental treatments and reconstruction, including bone repair and bone regeneration, often require the use of implant materials, such as metals, ceramics, polymers and composites, that are biocompatible, biodegradable and osteoconductive [1]. Of these, the most frequently researched are ceramics, with hydroxyapatite (HAp) gaining wide attention due to its close similarity with the inorganic component of bone that results in its excellent biocompatibility [1,2,3]. HAp possesses theoretical formula of
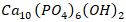 , hexagonal crystal lattice [2-4] and density of between 3.14-3.16 g/cm3 [4]. These features are responsible for its excellent biological properties, which enhance its biomedical application [2,4]. HAp is either extracted from natural sources, such as bone, shells and fish scale [1,5] or synthesized in the laboratory using materials with high calcium components, such as solid minerals [5,6]. Calcite-bearing minerals, such as limestone, marble and dolomite, are precipitated calcium carbonate of both plant and animal remains and have been utilized in the synthesis of HAp [6-8]. Dolomite is an inorganic and rock-forming crystal comprising of calcium magnesium carbonate (CaMg (CO3)2) [9]. It has been used to synthesize magnesium-substituted hydroxyapatite, which has been established to have anti-microbial properties due to the presence of magnesium [8]. Although it is widely available in Nigeria [10], it is widely utilized in the industrial sector, such as construction, fertilizer and cements production [11,12] but limited documentation exists on its biomedical application. Dolomite has been reported to be abundant in Ikpeshi, Edo State [12], which lies within latitudes 7°06′00″N to 7°20′00″N and longitudes 6°08′30″E to 6°20′64″E southern Nigeria [12]. Extensive studies have been carried out on the mineralogical composition, physical and chemical properties Nigerian-based dolomite [11,12]. Johnson et al. [12] studied the mineralogical characterization of the Ikpeshi deposit and observed that the deposit consists of 84.0 wt % dolomite with traces of other minerals, such as calcite (8. 0wt%) and quartz (8.0 wt%). Similarly, Omoseebi and Tanko [10] carried out geochemical analyses on Ikpeshi deposit and reported that the average concentration of the elemental oxides, such as silicon oxide (SiO2), aluminum oxide (Al2O3), iron oxide (Fe2O3), manganese oxide (MnO), magnesium oxide (MgO) and calcium oxide (CaO) present in the dolomite were 1.790, 0.461, 0.299, 0.045, 20.380 and 46.130 respectively. All these results confirmed the abundance of CaO and MgO in the mineral, which makes it a good candidate as a raw material for HAp synthesis. Unfortunately, there is little or no evidence to show that HAp can be synthesized from Nigerian-based dolomite. Hence, this work attempted to synthesize HAp using this abundant solid mineral. The conversion of CaCO3 in calcite-bearing minerals to HAp, firstly involves calcination at high temperature of 900°C to obtain its oxide [6,13], followed by other method, such as sol-gel, hydrothermal and wet chemical precipitation methods [1,3,6,13-15]. Hydrothermal technique involves the HAp precipitation in hydrated solution at elevated temperature and pressure to create an extremely crystalline artifact with a homogeneous chemical composition [16]. However, this method is costly due to its high energy consumption and use of specialized equipment, such as autoclaves [13]. Sol-gel method is another method but it utilizes costly reagents and produces low crystallized product [17]. Meanwhile, wet chemical precipitation method is frequently employed for the synthesis of medical-grade HAp due to ease of technology, low reaction temperature, reduced cost, and most importantly, high purity of the final product [17]. Consequently, this method was adapted for the synthesis of dolomite-based HAp in this study.The purity, crystallinity and morphology of HAp affect its biocompatibility and mechanical properties [18]. Pure HAp has calcium phosphorous ratio (Ca/P) of 1.67 and is thermodynamically stable in its crystalline state [19,20]. On the other hand, HAp products Ca/P ratio may be higher or less than 1.67, which signifies presence of CaO or [19]. This affects the mechanical properties [20] and the biocompatibility of the scaffold [18]. has low mechanical properties and non-stable in physiological fluid while CaO has high mechanical properties. In addition, it is worthy knowing that natural HAp is non-stoichiometric, which is as a result of either deficiency in calcium or phosphorus ion that is attributed to substitution of trace element [20]. Furthermore, crystallinity of HAp is an important parameter because it affects degradation of a scaffold thereby determining the mechanical properties of a scaffold [21]. Studies have shown that crystalline HAp is bio-inert and less degradable; however, very low heat treated HAp may result in amorphous effect and their degradation in the form of macro-particle release is likely to cause cellular damages around an implanted scaffold [22]. This implies that right crystallinity of HAp is needed for bone regeneration. Moreover, since the osteoblast cells of the bone are sensitive to the physical properties of their surroundings, including surface composition, surface energy, roughness and topography, it is essential to have the right morphology of HAp [23]. This demands imitation of the bone hydroxyapatite crystals, which are plate-or needle-shaped with varying size and shapes. This contributes to their structural stability, hardness and functions [23]. Therefore, it is necessary to synthesis HAp with composition, crystallinity and morphology of bone HAp, which may lead to the competing mechanical and the biological requirements need to rapidly stimulate healing of bone defects.In essence to have a pure, right morphology and right crystallinity, factors, such as reaction temperatures, sintering temperatures and pH of the solution, must be optimized [24]. Rodriguez-lugo et al. [25] examined the effect of reaction pH on purity and particle size of HAp, and observed that there is no relationship between the reaction pH and particle size. They, however, reported that pure HAp could only be synthesized at the pH of 10. Similarly, Wang et al. [26] examined the pH effect on HAp morphology, purity and particle size. The TEM image obtained by their study revealed sphere-like nanoparticles structures at the pH of 10 and 11 while aggregated nanowires and rectangular fragments at lower pH of 9 and 8, which was in consistence with the XRD spectra that revealed HAp peaks at pH of 10 while mixture of HAp and other phases peaks at the pH below or above 10. Rafie and Nordin [27] confirmed this finding by obtaining pure HAp at the pH of 10. Thus, the authors confirmed pH of 10 as the optimal value for the synthesis of pure HAp. Furthermore, effect of reaction temperature on purity, morphology and crystallinity of HAp was investigated by Wang et al. [26] and their TEM images revealed change from a mixture of spherical and rod-like to bamboo-leaf-like structure as the temperature changed from 25°C to 80°C while their XRD spectra revealed change from amorphous phase to crystalline phase as the temperature increased from 25 to 80°C. This finding implies that the HAp purity, crystallinity and particle size increase with a rise in the reaction temperature, which was corroborated by Jamarun et al. [28] and Gyorgy et al. [30]. Jamarun et al. [28] also reported 90°C as the optimal reaction temperature for HAp synthesis. This was corroborated by Kazemzadeh et al. [31], who also reported 90°C as the optimal reaction temperature for synthesis of highly purity crystalline HAp. Moreover, scholars have found that HAp purity, crystallinity and particle size, are affected by the sintering temperature [26]. In a study on the effect of sintering temperature on the HAp morphology, Rodriguez-lugo [25] reported that images changed from semi-acicular to a grain-like structure as the sintering temperature increased. Rafie and Nordin [27] confirmed this finding by reporting a change from needle-like particle size of 83 nm to the sphere-like structure of particle size of 228 nm as sintering temperature increased. This trend has been reported by several researchers [2,21,26,27], consequently the evidence that increase in particle size and crystallinity with a rise in sintering temperature was confirmed. In addition, Indrani et al. [21] established 900°C as optimal sintering temperature for HAp, above which resulted in HAp along with secondary phases, such as tri-calcium phosphate. Alias et al. [29] supported this finding by observing a shift in the XRD spectrum peaks for the sample sintered at 1000°C, which signifying the HAp dehydroxylation. Consequently, 900°C was established as the ideal sintering temperature for HAp by these authors. Hence, the established optimal parameters, including pH of 10, reaction temperature of 90°C and sintering temperature of 900°C were utilized for synthesis of dolomite-based hydroxyapatite with intention to obtain pure, medical-grade HAp.
, hexagonal crystal lattice [2-4] and density of between 3.14-3.16 g/cm3 [4]. These features are responsible for its excellent biological properties, which enhance its biomedical application [2,4]. HAp is either extracted from natural sources, such as bone, shells and fish scale [1,5] or synthesized in the laboratory using materials with high calcium components, such as solid minerals [5,6]. Calcite-bearing minerals, such as limestone, marble and dolomite, are precipitated calcium carbonate of both plant and animal remains and have been utilized in the synthesis of HAp [6-8]. Dolomite is an inorganic and rock-forming crystal comprising of calcium magnesium carbonate (CaMg (CO3)2) [9]. It has been used to synthesize magnesium-substituted hydroxyapatite, which has been established to have anti-microbial properties due to the presence of magnesium [8]. Although it is widely available in Nigeria [10], it is widely utilized in the industrial sector, such as construction, fertilizer and cements production [11,12] but limited documentation exists on its biomedical application. Dolomite has been reported to be abundant in Ikpeshi, Edo State [12], which lies within latitudes 7°06′00″N to 7°20′00″N and longitudes 6°08′30″E to 6°20′64″E southern Nigeria [12]. Extensive studies have been carried out on the mineralogical composition, physical and chemical properties Nigerian-based dolomite [11,12]. Johnson et al. [12] studied the mineralogical characterization of the Ikpeshi deposit and observed that the deposit consists of 84.0 wt % dolomite with traces of other minerals, such as calcite (8. 0wt%) and quartz (8.0 wt%). Similarly, Omoseebi and Tanko [10] carried out geochemical analyses on Ikpeshi deposit and reported that the average concentration of the elemental oxides, such as silicon oxide (SiO2), aluminum oxide (Al2O3), iron oxide (Fe2O3), manganese oxide (MnO), magnesium oxide (MgO) and calcium oxide (CaO) present in the dolomite were 1.790, 0.461, 0.299, 0.045, 20.380 and 46.130 respectively. All these results confirmed the abundance of CaO and MgO in the mineral, which makes it a good candidate as a raw material for HAp synthesis. Unfortunately, there is little or no evidence to show that HAp can be synthesized from Nigerian-based dolomite. Hence, this work attempted to synthesize HAp using this abundant solid mineral. The conversion of CaCO3 in calcite-bearing minerals to HAp, firstly involves calcination at high temperature of 900°C to obtain its oxide [6,13], followed by other method, such as sol-gel, hydrothermal and wet chemical precipitation methods [1,3,6,13-15]. Hydrothermal technique involves the HAp precipitation in hydrated solution at elevated temperature and pressure to create an extremely crystalline artifact with a homogeneous chemical composition [16]. However, this method is costly due to its high energy consumption and use of specialized equipment, such as autoclaves [13]. Sol-gel method is another method but it utilizes costly reagents and produces low crystallized product [17]. Meanwhile, wet chemical precipitation method is frequently employed for the synthesis of medical-grade HAp due to ease of technology, low reaction temperature, reduced cost, and most importantly, high purity of the final product [17]. Consequently, this method was adapted for the synthesis of dolomite-based HAp in this study.The purity, crystallinity and morphology of HAp affect its biocompatibility and mechanical properties [18]. Pure HAp has calcium phosphorous ratio (Ca/P) of 1.67 and is thermodynamically stable in its crystalline state [19,20]. On the other hand, HAp products Ca/P ratio may be higher or less than 1.67, which signifies presence of CaO or [19]. This affects the mechanical properties [20] and the biocompatibility of the scaffold [18]. has low mechanical properties and non-stable in physiological fluid while CaO has high mechanical properties. In addition, it is worthy knowing that natural HAp is non-stoichiometric, which is as a result of either deficiency in calcium or phosphorus ion that is attributed to substitution of trace element [20]. Furthermore, crystallinity of HAp is an important parameter because it affects degradation of a scaffold thereby determining the mechanical properties of a scaffold [21]. Studies have shown that crystalline HAp is bio-inert and less degradable; however, very low heat treated HAp may result in amorphous effect and their degradation in the form of macro-particle release is likely to cause cellular damages around an implanted scaffold [22]. This implies that right crystallinity of HAp is needed for bone regeneration. Moreover, since the osteoblast cells of the bone are sensitive to the physical properties of their surroundings, including surface composition, surface energy, roughness and topography, it is essential to have the right morphology of HAp [23]. This demands imitation of the bone hydroxyapatite crystals, which are plate-or needle-shaped with varying size and shapes. This contributes to their structural stability, hardness and functions [23]. Therefore, it is necessary to synthesis HAp with composition, crystallinity and morphology of bone HAp, which may lead to the competing mechanical and the biological requirements need to rapidly stimulate healing of bone defects.In essence to have a pure, right morphology and right crystallinity, factors, such as reaction temperatures, sintering temperatures and pH of the solution, must be optimized [24]. Rodriguez-lugo et al. [25] examined the effect of reaction pH on purity and particle size of HAp, and observed that there is no relationship between the reaction pH and particle size. They, however, reported that pure HAp could only be synthesized at the pH of 10. Similarly, Wang et al. [26] examined the pH effect on HAp morphology, purity and particle size. The TEM image obtained by their study revealed sphere-like nanoparticles structures at the pH of 10 and 11 while aggregated nanowires and rectangular fragments at lower pH of 9 and 8, which was in consistence with the XRD spectra that revealed HAp peaks at pH of 10 while mixture of HAp and other phases peaks at the pH below or above 10. Rafie and Nordin [27] confirmed this finding by obtaining pure HAp at the pH of 10. Thus, the authors confirmed pH of 10 as the optimal value for the synthesis of pure HAp. Furthermore, effect of reaction temperature on purity, morphology and crystallinity of HAp was investigated by Wang et al. [26] and their TEM images revealed change from a mixture of spherical and rod-like to bamboo-leaf-like structure as the temperature changed from 25°C to 80°C while their XRD spectra revealed change from amorphous phase to crystalline phase as the temperature increased from 25 to 80°C. This finding implies that the HAp purity, crystallinity and particle size increase with a rise in the reaction temperature, which was corroborated by Jamarun et al. [28] and Gyorgy et al. [30]. Jamarun et al. [28] also reported 90°C as the optimal reaction temperature for HAp synthesis. This was corroborated by Kazemzadeh et al. [31], who also reported 90°C as the optimal reaction temperature for synthesis of highly purity crystalline HAp. Moreover, scholars have found that HAp purity, crystallinity and particle size, are affected by the sintering temperature [26]. In a study on the effect of sintering temperature on the HAp morphology, Rodriguez-lugo [25] reported that images changed from semi-acicular to a grain-like structure as the sintering temperature increased. Rafie and Nordin [27] confirmed this finding by reporting a change from needle-like particle size of 83 nm to the sphere-like structure of particle size of 228 nm as sintering temperature increased. This trend has been reported by several researchers [2,21,26,27], consequently the evidence that increase in particle size and crystallinity with a rise in sintering temperature was confirmed. In addition, Indrani et al. [21] established 900°C as optimal sintering temperature for HAp, above which resulted in HAp along with secondary phases, such as tri-calcium phosphate. Alias et al. [29] supported this finding by observing a shift in the XRD spectrum peaks for the sample sintered at 1000°C, which signifying the HAp dehydroxylation. Consequently, 900°C was established as the ideal sintering temperature for HAp by these authors. Hence, the established optimal parameters, including pH of 10, reaction temperature of 90°C and sintering temperature of 900°C were utilized for synthesis of dolomite-based hydroxyapatite with intention to obtain pure, medical-grade HAp.2. Materials and Methods
2.1. Materials
- The dolomite utilized as source of calcium precursors for this study was obtained from Ikpeshi in Delta state of Nigeria. The choice of this location was due to massive deposit of this mineral. Ortho-phosphoric acid, nitric acid and ammonium (as a regulator) of analytical grade were utilized in the study while distilled water was the only solvent used throughout the synthesis. The magnetic stirrer, centrifuge, oven and furnace were the equipment used in this work.
2.2. Methods
- This study synthesized HAp with dolomite by calcination and wet chemical precipitation method.
2.2.1. Calcination
- The solid mineral was pulverized, sieved and subsequently 1000 grams of pulverized minerals were calcined at 900°C for 3 hours to convert to its oxide, see equation (i) for the calcination reaction.
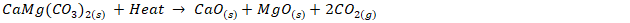 | (1) |
2.2.2. Synthesis of Hydroxyapatite
- Sirait et al. [6] proceedure was used for synthesis of HAp using wet chemical precipitation method. However, due to low solubility, 0.8 M of nitric acid was used to leach out calcium from the dolomite. This method involves dissolving 11.84 g of calcined dolomite with 0.8 M of nitric acid of 200 mls, followed by stirring with a magnetic stirrer for 30 minutes before separation of the filtrate from mixture. The pH of the filtrate was adjusted to 10 with 1M of ammonium solution and 0.3 M of phosphoric acid was added drop wise at rate of 5 mls per minute. Subsequently, the ammonium solution of 1M was used to precipitate HAp from the solution at 90°C and pH of 10. The precipitate was left to age for 24 hours before it was washed, dried and finally sintered at 900°C. See equation ii-iv for synthesis reactions.
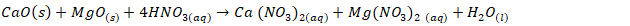 | (2) |
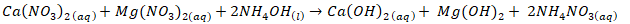 | (3) |
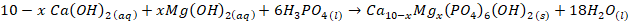 | (4) |
2.3. Material Characterization
2.3.1. X-ray Diffraction (XRD) Characterization
- The phase composition of dolomite and HAp was studied by XRD using Cu Kα radiation (λ = 0.154 nm). The samples were tested following ASTM-E915-10, which involved mounting the powder on the microscope slide face using a suitable amorphous binder, such as 10% solution of nitrocellulose cement diluted with acetone while the specimen surface was made as smooth as possible. The sample was scanned with 2Ѳ ranging from 20° to 80° at a scan step of 2 per minute and the XRD pattern was generated at the voltage of 40 KV and current of 30 mA. The particle size, surface area and crystallinity of synthesized HAp were done with XRD data using Equation v, vi and vi respectively [32].
 | (5) |
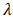 is the wavelength used,
is the wavelength used, 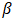 (FWHM) is peak width and
(FWHM) is peak width and 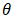 is the Bragg diffraction angle].
is the Bragg diffraction angle].  | (6) |
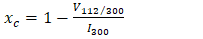 | (7) |
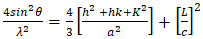 | (8) |
2.3.2. X-ray Fluorescence (XRF) Characterization
- The pulverized dolomite and synthesized HAp elemental composition were determined using XRF technique based on the ASTM C114-11 standard, which involved grinding the XRF samples into fine grains, followed by mixing it with binder in the ratio of 10 to 1 from which four grams were pressed using an aluminum ring in order to obtain tubular pellets suitable for the EDX3600B X-ray fluorescence spectrometer. Each of the pellets from the different samples was mounted on the sample holder’s and was irradiated by an intense X-ray beam from the fixed tube at operating conditions of 25 KVA and 6 MA for 20 minutes. The test outcome was presented on the computer connected to X-ray fluorescence spectrometer, which recorded the elements percentage.
2.3.3. Fourier Transform Infrared (FT-IR) Characterization
- The functional groups present in synthesized HAp were examined using FT-IR spectroscopy based on ASTM E168 standard, which involves mixing five milligrams of analyst with about 100 mg of dried potassium bromide and an appropriate amount of the pellet was prepared by compression. The pellet was placed in the incident IR beam path of varying frequency, subsequently, resonant absorbed by the molecules resulted in a percentage transmission being detected in the FT-IR detector. Finally, the FT-IR spectrum was obtained over the region 400-4000 cm-1.
2.3.4. Scanning Electron Microscopy (SEM) Characterization
- The synthesized HAp morphology, particle size distribution and microstructure were studied using SEM based on ASTM E986-97 standard. This involved coating the sample with platinum and subsequently attaching it on a pure copper sample holder, which was put in the SEM that operated under vacuum at the pressure of 2.5 MPa and an image pattern in the topography form was generated.
3. Results
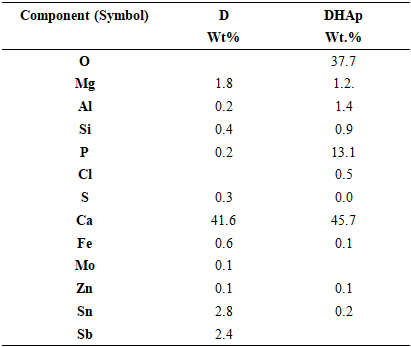
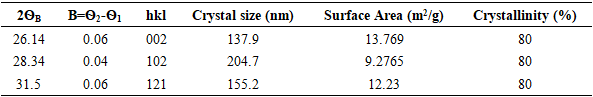
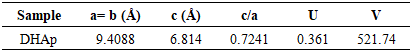
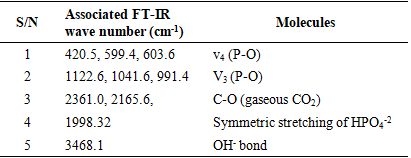
- The XRD spectra obtained from the dolomite (D) and dolomite-based HAp (DHAp) were presented in Figure 1 while XRF results were summarized in Table 1. The crystal size (Dhkl), surface area and crystallinity for DHAp were calculated from X-ray diffractogram data and were presented in Table 2. In addition, their unitary cell parameter (a, c) and unit cell volume (V) were presented in Table 3 while the Ca/P ratio of the DHAp was summarized in Table 4. Furthermore, FT-IR spectrum of DHAp was shown in Figure 2 while the wave number and chemical group of FT-IR absorbtion bands were briefed in the Table 5. Also, representative SEM micrograph of the obtained HAp was presented in Plate 1.
4. Discussion
- The elemental and structural analyses of the dolomite as-received in this study were discussed using XRF and XRD results. Also, chemical properties analysis and morphologyof the synthesized dolomite-based HAp were discussed using results from XRF, XRD and SEM.
4.1. Analysis of the Physicochemical Properties of Dolomite and Dolomite-Based HAp
- • The dolomite and dolomite-based hydroxyapatite XRD SpectraThe XRD and XRF spectroscopy were used to analyze the dolomite as received and dolomite-based HAp as shown in Figure 1 and Table 1 respectively. The XRD patterns were similar to XRD pattern of calcite (CaCO3) with the peaks 29.60° (104), which matched with the primary reference JCPD card no. 00-002-0629. Also, peaks of calcium carbonate silicate was also observed with the following peaks 26.76° (130), 31.23° (-511) and 39.84° (-531), which were similar to JCPD card no. 00-13-0416. Also, the calcium magnesium carbonate was observed with the following peaks 24.220 (012), 33.76° (006), 35.6° (015), 37.6° (110) 41.42° (113), 45.2° (202), 50.8° (018) and 51.4° (116), which matched with standard reference JCPD card no. 00-036-0426 of calcium magnesium carbonate. This result was corroborated with XRF results in which dolomite had calcium contents of 41.61 %, silicon content of 0.4234 % and magnesium (Mg) content of 1.81 wt. %.
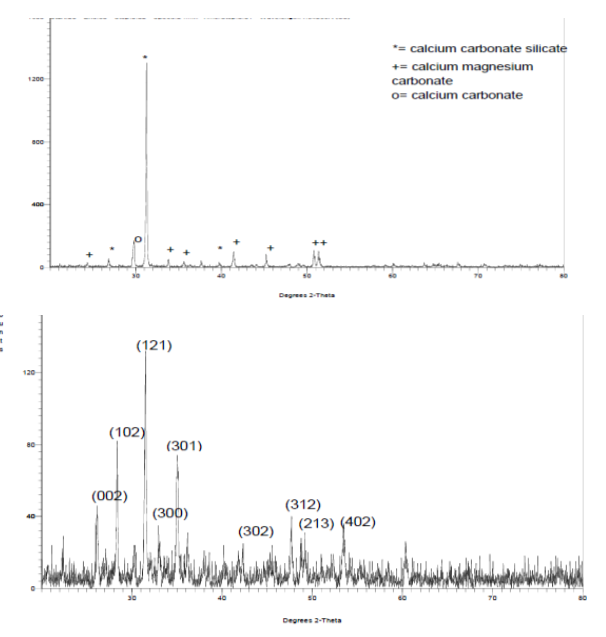 | Figure 1. XRD spectrum of (a) dolomite (b) dolomite-based Hap |
|
 and a= 9.4088
and a= 9.4088 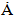 , in Table 3, which showed insignificant diversion from the stoichiometric hydroxyapatite (c = 6.8745
, in Table 3, which showed insignificant diversion from the stoichiometric hydroxyapatite (c = 6.8745 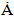 and a= 9.4166
and a= 9.4166 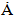 ) [32]. Furthermore, the HAp had narrow peaks, which signifies big particle size and high crystallinity. This observation was also revealed in the calculated crystal size and % crystallinity summarized in Table 2. It can be seen that the particle size was not even, which is one of the feature of wet chemical precipitation product. This result was supported by FT-IR and XRF results.
) [32]. Furthermore, the HAp had narrow peaks, which signifies big particle size and high crystallinity. This observation was also revealed in the calculated crystal size and % crystallinity summarized in Table 2. It can be seen that the particle size was not even, which is one of the feature of wet chemical precipitation product. This result was supported by FT-IR and XRF results.
|
|
|
|
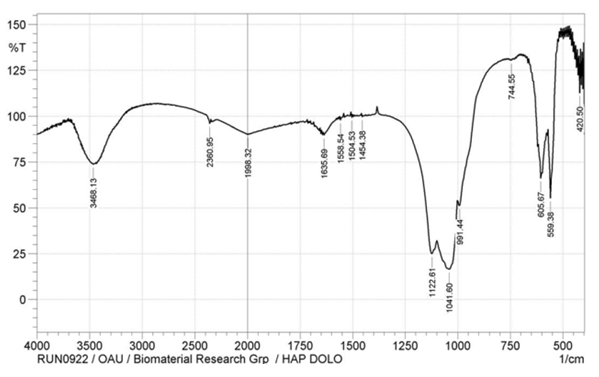 | Figure 2. The FT-IR spectrum of dolomite-based hydroxyapatite |
 | Plate 1. SEM micrograph of dolomite-based hydroxyapatite of Magnification (a) 2000x and (b) 10000x |
5. Conclusions
- The study concluded that hydroxyapatite was successfully synthesized from Nigerian-based dolomite based on the results of elemental, chemical and morphological characterizations. The results, furthermore, support the hypothesis that the HAp synthesized from dolomite may be suitable for bone regeneration.
ACKNOWLEDGEMENTS
- The authors gratefully acknowledge the financial support from Tertiary Education Trust Fund (TETFUND).
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML