-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Biomedical Engineering
p-ISSN: 2163-1050 e-ISSN: 2163-1077
2017; 7(1): 1-8
doi:10.5923/j.ajbe.20170701.01

A Systematic Study on Flowability and Compressibility of Symphonia globulifera Stem Bark Powder for Tablet Dosage Form
Stephen Olaribigbe Majekodunmi, Uwaoma Lucky Aliga
Department of Pharmaceutics and Pharmaceutical Technology, Faculty of Pharmacy, University of Uyo, Uyo, Nigeria
Correspondence to: Stephen Olaribigbe Majekodunmi, Department of Pharmaceutics and Pharmaceutical Technology, Faculty of Pharmacy, University of Uyo, Uyo, Nigeria.
| Email: |  |
Copyright © 2017 Scientific & Academic Publishing. All Rights Reserved.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The stem bark of Symphonia globulifera is widely used in the treatment of several disorders ranging from parasitic disease, body pain, scabies, spiritual remedies, skin diseases to malaria, diabetes, venereal disease and erective problems in Africa and South America. The objective of the present research was to study the original flowability, compressibility and compactibility of Symphonia glubulifera stem bark and develop its tablet formulations by wet granulation and direct compression technology. The consolidation behaviour of drug and tablet formulations was studied by using Heckel and Leuenberger equations. Symphonia globulifera stem bark powder showed very poor flowability and compactibility. Kawakita analysis revealed improved flowability for formulations prepared by direct compression and wet granulation techniques. The Heckel plot showed that Symphonia globulifera stem bark powder is soft in nature and poor in die filling. Granules showed higher degree of plasticity and fragmentation than powder and direct compression formulations. The compression susceptibility parameter for compact formed by direct compression and wet granulation technique indicated that the maximum crushing strength is reached faster at lower pressures of compression as opposed to Symphonia globulifera stem bark powder. From this study, it is concluded that desired flowability, compressibility and compactibility of Symphonia glubulifera stem bark powder can be obtained by direct compression and wet granulation technique, being higher in the wet granulation.
Keywords: Symphonia globulifera, Flowability, Compressibility, Compactibility, Tablets
Cite this paper: Stephen Olaribigbe Majekodunmi, Uwaoma Lucky Aliga, A Systematic Study on Flowability and Compressibility of Symphonia globulifera Stem Bark Powder for Tablet Dosage Form, American Journal of Biomedical Engineering, Vol. 7 No. 1, 2017, pp. 1-8. doi: 10.5923/j.ajbe.20170701.01.
Article Outline
1. Introduction
- Medicinal plants have been playing an important role in providing health care to a large section of the population, especially in developing countries. Higher plants are known to be a rich source of various bioactive compounds [1], some of which have found practical applications in traditional medicine [2]. Symphonia globulifera L (Family Clusiaceae) has been widely used in traditional medicine to fight against various disorders such as parasitic disease [3, 4] or body pain [5]. Extracts of this plant have shown very good biological activities against several pathologies, opening a vast field of research towards the identification of complex metabolites. [6]Since the first publication in 1992 [7] describing some polycyclic polyprenylated acyl- phloroglucinols (PPAPs) from S. globulifera as HIV inhibitors, the interest for this plant and its bioactive compounds has been ever growing. Like the plants of the Garcinia genus, which also contain PPAPs [8], the plants of the species S. globulifera have emerged on both American and African continents, and show some morphological diversity through sites [9]. This morphological differentiation and the existence of some subfamilies and differences in country soil and climate have probably induced a variation in the metabolome and generated a pool of chemodiversity. The family Clusiaceae (Guttiferae) comprises about 40 genera and more than a thousand species. The genus. Symphonia includes 17 species [10]. S. globulifera is broadly distributed across the Neotropics and equatorial Africa. S. globulifera plants are generally tall trees (in general more than 15 m high) with opposite leaves exhibiting characteristic aerial roots and producing bright yellow latex. The flowers are red with a red staminal column and black anthers and organized as a sympodium. Fruits are drupes (4-5 cm), ovoid, or globular. Seeds are intensively red inside [11, 12].In African, S. globulifera preparations are mainly decoctions, with applications ranging from serious disorders, such as scabies, to spiritual remedies. More recently, studies from Nigeria [13] and Uganda [4] describe the use of S. globulifera not only in terms of ethnicity but also depending on the region of occurrence: Akwa Ibom State (Nigeria) and the Sango bay area (Uganda). In Nigeria, leaves of S. globulifera are used as a decoction and are applied on the body to treat skin disease, which is the largest application followed by malaria and diabetes. Other traditional uses in Nigeria are described in the literature to treat erective problems, venereal diseases, or wounds using the fruits and leaves of S. globulifera [14] According to [4], the S. globulifera biological activities are dependent on the vegetal parts. Thus, the bark extract presents broad applications ranging from treating coughs and prehepatic jaundice to fever and intestinal worms. A different application has been observed for the sap extract, which is used for spiritual application to chase away evil spirits. While this traditional use of S. globulifera has been proven to exist, the obvious lack of scientific meaning makes its difficult to understand.The traditional uses of S. globulifera on both the African and American continents are specific but present some similarities. The application of cataplasm directly on the body to treat skin diseases or cutaneous leishmaniasis revealed the presence of polar molecules, which are attractive for cosmetic, dermatologic, and antiparasitic applications. Comparing the practices in both continents, the bark seems to contain the main active metabolites, while the leaves and fruits are poorly used. From all these surveys, a potent and promising antiparasitic activity of S. globulifera metabolites emerges.A need for systematic study on formulation development of a tablet dosage form of Symphonia globulifera stem bark powder has been undertaken. The measurement of porosity change as a function of compression pressure is widely used in describing the powder compressional behavior. The compressibility of a powder bed could be inferred from the relationship between porosity and applied pressure [15]. Due to poor flowability and compaction behavior, Symphonia globulifera stem bark powder frequently requires alteration prior to tabletting. Direct compression of powders requires materials exhibiting good flowability, compactibility and compressibility [16]. These parameters become more critical when the formulation contains large amount of active substances with poor compressional properties. Wet granulation method is selected for production of porous and free-flowing granules, which enables to form tablets with high mechanical strength at low compression pressure. In the present study, the influence of alteration in particle size and direct compression on the development of tablet formulations of Symphonia globulifera stem bark powder was investigated. Flowability, compactibility and compressibility [17] were systematically investigated with an aim to understand the consolidation behavior.
2. Materials and Methods
2.1. Materials
- The stem bark of Symphonia globulifera was collected from a local farm in Afaha Ube Itam in Itu Local Government of Akwa-Ibom State. The stem bark was identified by Mr. Etafia, a botanist and taxonomist in the Department of Pharmacognosy and Natural Medicine, Faculty of Pharmacy, University of Uyo, Uyo, Akwa-Ibom State, Nigeria.
2.2. Methods
2.2.1. Pre-Extraction Treatment
- The collected bark was spread on a brown paper and air-dried for 5 days before being blended in a local mortar to a coarse powder and then by a laboratory mill. They were then weighed.
2.2.2. Extraction of Powder
- Symphonia glubulifera (SG) stem bark powder (1Kg) was macerated in a 90% 2.5L ethanol in an extraction jar and mixed thoroughly with intermittent vigorous shaking. The mixture was allowed to stand for 72 hour under room temperature and covered with a lid to prevent evaporation of the solvent. After 72 hours, the liquid extra was separated from the marc through filtration. The filtrate was concentrated to dryness in vacuum (water bath) (Model: DKZ, Chaking Water Bath, Gallenham, UK) at 40°C. After concentration, the extract was weighed on a balance and stored in a small beaker that is covered with aluminium foil.
2.2.3. Identification of Extract
- The extract was physically examined for standard organoleptic properties of colour, odour, texture and taste. The extract was subjected to phytochemical screening to ascertain the presence of phytochemicals, such as, tannins, alkaloids, glycosides etc [18]. Solubility characteristics of the extract in various solvents were also elucidated.
2.3. Preparation of Granules
- The wet granulation method of massing and screening was used with a batch size of 500 tablets. SG stem powder (80% w/w) and Avicel PH 101 (15% w/w) were dry mixed for 10 minutes in a Kenwood planetary mixer. The dry mix was moistened with an appropriate amount of 5% (w/v) acacia mucilage and subjected to wet mixing for 15 minutes in the same mixer. The wet mass was passed through mesh sieve No. 12 (1,400µm). The granules were dried in a hot air oven (Model: 0384635, Selecta, India) for 4 hrs at 60°C and then re-sieved through sieve No.16. (1000 µm). Talc (1%) and magnesium stearate (1%) were added and mixed for 5 min in a mixer.
2.4. Preparation of Direct Compression Formulation
- SG stem powder (80% w/w), Avicel PH102 (10% w/w), silicified MCC (8%), talc (1%) and magnesium steerage (1%) were thoroughly mixed for 10 min in a Kenwood planetary mixer and then stored in airtight containers.
2.5. Powder Properties
2.5.1. True Density
- The true densities of the SG stem powder, granule and direct compression formulations were determined by the liquid displacement method using immiscible solvent and the true density (ρT) was computed (n = 3) according to the equation:
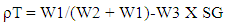 | (1) |
2.5.2. Bulk and Tap Density
- SG granules (30g) were placed in a 100mL clean, dry measuring cylinder and the bulk volume Vb occupied without tapping was determined. After 40 taps on a flat horizontal surface, the tapped volume Vt occupied was also determined. The bulk and tapped were calculated as the ratio of weight to volume (Vb and Vt respectively). Carr’s Index = Tapped Density – Bulk Density / Tapped Density
2.5.3. Flow Properties
- 2.5.3.1. Flow Rate The flow rate [19] of the SG stem powder, granule and direct compression formulations were determined as the ratio of mass (g) to time (seconds) using a steel funnel with an orifice diameter of 10 mm (n =3). 2.5.3.2. Kawakita Analysis Flowability was determined using the Kawakita analysis [20, 21]. The method involved pouring a 10 g of powder into a 50 ml glass measuring cylinder and the heap of the particles in the cylinder was scraped off horizontally with a thin metallic spatula, and the bulk volume Vo was accurately measured. Then tapping was started mechanically and the change in volume of the powder column VN was noted after N no of taps. The behavior of powder, granule and direct compression formulations in the tapping procedure were compared using numerical constants obtained from the Kawakita plots. The Kawakita equation, which is used for assessing the flow properties of powders, is given by:
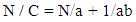 | (2) |
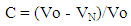 | (3) |
2.5.4. Compaction Studies
- 2.5.4.1. Preparation of Compacts Containing 500 mg of Symphonia globuliferaCompacts containing 500 mg of Symphonia globulifera (SG)were made for stem bark powder, granules and direct compression formulations using a hydraulic single press (Cadmach Machinery Co, Serial no: 193/2/07-08, Type: SSF: 3, Ahmedabad, 45, India). Compression loads were used in the range of 10 Kg/cm2 to 95 Kg/cm2. Three compacts each were made from stem bark powder, granules, and direct compression. Before compression, the die (12.5 mm diameter) and the flat- faced punches were lubricated with a 2% w/v dispersion of magnesium stearate in ethanol ether (1:1). The compacts were stored over silica gel for 24 hours (to allow for elastic recovery, hardening and prevent falsely low yield values) before evaluations. The dimensions (thickness and diameter) and weight uniformity of three compacts were determined. The relative density ρr was calculated as the ratio of apparent density ρA of the compact to the true density ρT, of the powder. The data obtained using this ‘ejected tablet method’ were used to obtain the Heckel plots. Linear regression analysis was carried out over a compression range of 10 Kg/cm2 to 95 Kg/cm2 and parameters from Heckel plots [22, 23] were calculated.2.5.4.2. Heckel EquationThe compaction characteristics of the powder were studied with the Heckel equation:
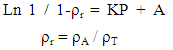 | (4) |
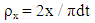 where x is hardness (in Kg/cm2) and d and t are the diameter and thickness of the compacts (in mm), respectively.Leuenberger analysis was performed by fitting the data in the following equation [25]. A nonlinear plot of tensile strength with respect to product of compaction pressure. P and relative density ρr was obtained using statistical software (Graph Pad Prism 4):
where x is hardness (in Kg/cm2) and d and t are the diameter and thickness of the compacts (in mm), respectively.Leuenberger analysis was performed by fitting the data in the following equation [25]. A nonlinear plot of tensile strength with respect to product of compaction pressure. P and relative density ρr was obtained using statistical software (Graph Pad Prism 4): 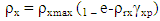 Where, ρ x max is maximum tensile strength (kg/cm2) when p will be infinite and ρr will be equal to 1, and γ is compression susceptibility.
Where, ρ x max is maximum tensile strength (kg/cm2) when p will be infinite and ρr will be equal to 1, and γ is compression susceptibility.2.5.5. Preparation of Tablet
- Tablets containing 500 mg of Symphonia globulifera (SG) were produced by compressing the granules and direct compression formulations using a single station tablet punching machine (Cadmach Machinery Co Pvt. Ltd., Type: SSF:3, Serial no: 193/2/107-08, Mumbai, India) equipped with 12.5 mm circular, flat and plain punches. 2.5.5.1. Evaluation Tests for Tablets The prepared tablets of SG stem bark powder by both wet granulation and direct compression were subjected to standard quality control tests for tablets. Weight variation was determined by weighing 20 tablets individually, the average weight was calculated and the percent variation of each tablet was determined. Crushing strength was determined by taking 6 tablets form each formulation using a digital tablet hardness tester (Monsanto, Serial no: 09091082, Model: MHT-20, India) and the average of applied pressure (Kg/cm2) for crushing the tablet was determined. Friability was determined by first weighing 10 tablets and placing them in a friability tester (Tablet Friability test Apparatus, Type: DT/2D, UK), which was rotated for 4 min at 25 rpm. After dusting, the total remaining weight of the tablets was recorded and the percent friability was calculated. Disintegration time for the tablets was determined in 900 ml of distilled water using a disintegration test apparatus (Model: Copley ZT2, Erweka Apparatebau GMBH, Heusenstamm, Germany). Statistical AnalysisAll parameters, values and determinations were calculated using Graph Pad Prism 5 (GraphPad Software Inc. San Diego, USA).
3. Results and Discussion
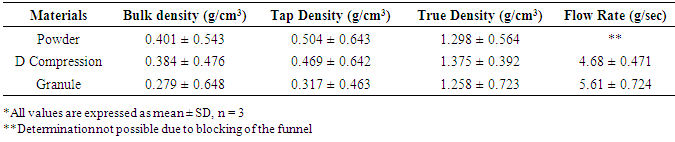
- The fundamental flow properties of the SG stem bark powder exhibit no flow through funnel, which revealed that it was not up to the theoretical level for processing into tablet dosage form. Flow rate of direct compression formulation and granule revealed a significant improvement in the flowability (Table 1). One of the most important factors affecting bulk density of a powder and its flow properties, is the interparticulate interaction [26]. Desirable micromeritic properties and the optimal presence of water diminish the cohesiveness of the powder, resulting in an increased bulk density for granule and direct compression formulation revealed enhanced flowability [27]. Similarly, increased tapped density for granule and direct compression formulation indicated better degree of compactibility as a function of applied pressure [28] (Table 1). True density value of powder and direct compression formulation was quite close to each other whereas it was less in case of granules.
|
3.1. Flow Properties
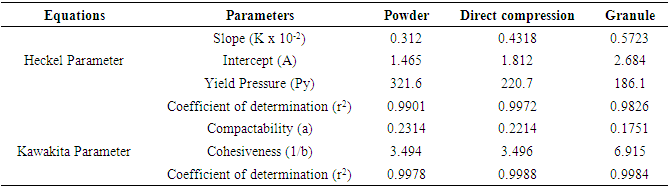
- Plots of N/C versus N (Kawakita plots) for SG stem bark powder and formulations, gave the linear relationship. Kawakita constants indicate the behavior of the powder from the bulk density state to the tap density state. The constants of the Kawakita equation were resolved from the slope and intercept of the line from graphs N/C versus N (Table 2). Granule densified the least (small compressible value) but attained the final packing state most slowly. On the other hand, direct compression formulation densified considerably but achieved the final packing state rather quickly than powder and granule. Lower value of ‘a’ for granule revealed better flowability than direct compression formulation. Whereas, lower value of 1/b for direct compression formulation showed that it is less cohesive than granule [29].
|
3.2. Compaction Properties
3.2.1. Heckel Analysis
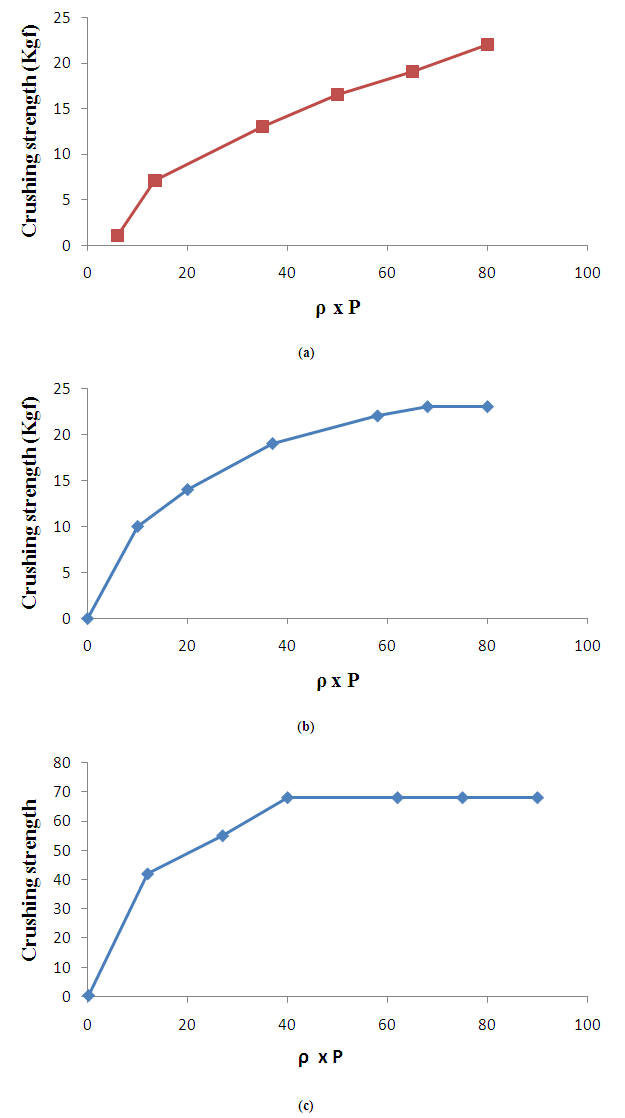
- The Heckel plots (Fig. 1) for direct compression formulation and granule showed no linearity at early stages of compression, because of particle rearrangement and the initial fragmentation. Granule showed highest value for die filling in initial stages of rearrangement as indicated by their intercept A values. These features of the later could result to formation of bridges and arches, which could in turn prevent close packing of the particles in the bulk state. Higher value of A for granule implies higher degree of fragmentation. At low pressure, the large granules were fractured into small ones, which facilitated the further rearrangement. When the compression pressure was increased, the granule showed plastic deformation [30]. Greater slopes indicate a greater degree of plasticity of material.
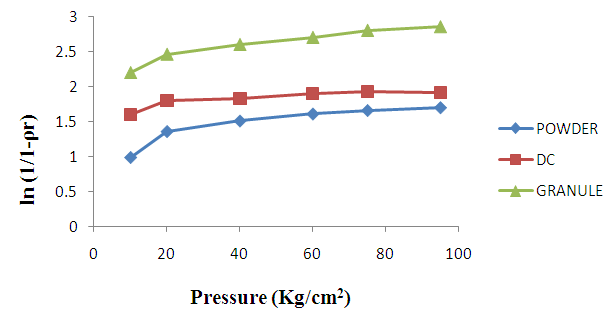 | Figure 1. Heckel plot for Symphonia globulifera (stem bark powder), direct compression formulation (D.C) and wet granulation formulation (granule) at various compression pressures (mean ± SD, n = 3) |
3.2.2. Leuenberger Equation
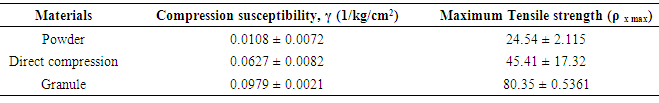
- The compression susceptibility parameter (Fig. 2) for compact formed by direct compression and wet granulation technique indicated that the maximum crushing strength is reached faster at lower pressures of compression as opposed to SG stem bark powder. Higher value for σx max was observed in case of granule than direct compression formulation. It showed that granule can build a compact with a higher strength than direct compression formulation. Lower value of compression susceptibility for SG stem powder demonstrated that maximum tensile strength could be obtained slowly at higher pressure.
|
3.2.3. Evaluation Tests for Tablets
- All the batches of tablets were produced under similar conditions to avoid processing variables. Weight variation for the SG tablets prepared by wet granulation and direct compression method were in the range of 625 ± 16 mg and 624 ± 21 mg respectively. Crushing strength of tablets was higher for tablets prepared by wet granulation method (5.2 ± 1.23 Kg/cm2) than the direct compression method (4.8 ± 2.31 Kg/cm2). The percentage friability for tablets prepared by direct compression method (0.75 ± 0.28%) was more than for tablets prepared by wet granulation method (0.46 ± 0.16%). The values of crushing strength and percent friability indicated good handling property of the prepared SG stem bark tablets. Disintegration time was 10 ± 2.5 and 12 ± 1.5 min for tablets prepared by direct compression and wet granulation methods respectively.
4. Conclusions
- Both wet granulation and direct compression method could be used successfully for developing tablet formulation of Symphonia globulifera stem bark powder. Granules prepared using wet granulation showed better flowability, compressibility and compactibility compared to direct compression formulation.
ACKNOWLEDGEMENTS
- Authors wish to thank the technical staff of the Department of Pharmaceutics and Pharmaceutical Technology, Faculty of Pharmacy, University of Uyo, Uyo, Akwa-Ibom State for their cooperation during the course of this study.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML