-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Biochemistry
p-ISSN: 2163-3010 e-ISSN: 2163-3029
2019; 9(1): 3-6
doi:10.5923/j.ajb.20190901.02

Vitamin D and Parathyroid Hormone Levels in Egyptian Patients with Non-Alcoholic Fatty Liver Disease - A Case Control Study
Zakarya M. Zakarya1, Abdullah Gaafar2
1Department of Internal Medicine, Faculty of Medicine, Al-Azhar University, Cairo, Egypt
2Department of Clinical Pathology, Faculty of Medicine, Al-Azhar University, Cairo, Egypt
Correspondence to: Zakarya M. Zakarya, Department of Internal Medicine, Faculty of Medicine, Al-Azhar University, Cairo, Egypt.
| Email: |  |
Copyright © 2019 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: Vitamin D plays a critical role in the adjustment of different genes linked to various metabolic disorders. This study was outlined to evaluate the association between serum 25 hydroxy vitamin D (25 (OH) D) and parathyroid hormone (PTH) levels, insulin levels and insulin resistance (IR) in patients with non-alcoholic fatty liver disease (NAFLD). Methods: A case-control study was done on 100 patients with NAFLD and age-matched subjects without NAFLD (n= 50) as the control group. After 8-12 hours fasting, serum 25 (OH) D, PTH, insulin and fasting blood glucose (FPG) levels were assessed. The multivariate logistic regression analysis model was used to evaluate the association between vitamin D, PTH and IR in the occurrence of NAFLD by adjusting for the independent variables. Results: Patients with NAFLD had significantly decreased serum 25(OH) D levels and elevated levels of PTH, insulin and FPG compared to controls (p< 0.001). The relationship between NAFLD and the decreased 25(OH) vitamin D values was independent of confounders. Conclusion: Our results showed that decreased serum 25 hydroxy vitamin D concentration and elevated insulin and IR values were independently associated with increased likelihood of having NAFLD.
Keywords: 25 hydroxy vitamin D, Insulin resistance, Parathyroid hormone, NAFLD
Cite this paper: Zakarya M. Zakarya, Abdullah Gaafar, Vitamin D and Parathyroid Hormone Levels in Egyptian Patients with Non-Alcoholic Fatty Liver Disease - A Case Control Study, American Journal of Biochemistry, Vol. 9 No. 1, 2019, pp. 3-6. doi: 10.5923/j.ajb.20190901.02.
1. Introduction
- Non-alcoholic fatty liver disease (NAFLD) is one of the most common chronic liver disorders. Triglyceride fat droplets within liver cells constitute the pathological hallmark of the disease [1,2]. The pathophysiological mechanisms involved in NAFLD are not fully understood. However, the ‘multi-hit’ theory was proposed as an explanatory model. This theory suggests insulin resistance as the “first hit” which induces an aggregation of triglycerides in liver cells. Subsequently, oxidative stress “the second hit” results in inflammation and initiation of osteohepatitis; a process that recruits a wide range of inflammatory cytokines. The “third hit” consists of liver cell proliferation [3].In spite of the sunny weather, hypovitaminosis D is a common encounter in Middle Eastern countries with a prevalence ranging from 20% to 70.0 [3,4]. The condition is probably attributed to avoidance of sunlight or clothing which prevents adequate sunlight exposure [5,6].Deficiency of vitamin D is associated with glucose homeostatic abnormalities such as insulin resistance, impaired fasting glucose, metabolic syndrome and type 2 diabetes [7-9]. In addition, low levels of 25-Hydroxy vitamin-D together with elevated levels of parathyroid hormone (PTH) have been suggested as markers of metabolic syndrome and fatty liver disease [10]. The present study was carried out to study the association between the serum levels of 25 (OH) vitamin-D (25 (OH) D) and PTH and insulin resistance in Egyptian patients with NAFLD.
2. Subjects and Methods
- This case–control study included one hundred adult patients with NAFLD in the period between September 2018 to March 2019 and fifty age- and sex-matched healthy controls. Patients with NAFLD were collected from Outpatient Clinics of the Internal Medicine Department, El-Husein University Hospital and the controls were members of staff of the hospital. For diagnosis of NAFLD, abdominal ultrasonography was used; all studies were done by a single sonographist who was blind to the aims of the study, to prevent bias. The local Ethical Research and Review Committee approved the study protocol, and informed consent was obtained from the participants. Exclusion criteria included history of alcohol consumption, diabetes mellitus, glucocorticoid therapy, Cushing’s disease, overt hypothyroidism, chronic renal disease, chronic liver disease, hepatotoxic medications, obstruction of biliary tree, pregnancy, lactation, and postmenopause.All participants were subjected to careful history taking, thorough clinical examination and routine laboratory investigations. Serum 25(OH) D, the most stable circulating form of this molecule was assessed by Bioactiva 25 (OH) D ELISA kit, (bioactiva diagnostic, Germany); intraassay coefficient of variation (CV) for this kit was 4.6% and inter-assay CV was 5.4%. The sensitivity for the assessment was 0.57 ng/ml. Fasting PTH levels was determined via IBL kit (Parathyroid hormone intact ELISA kit, IBL International GMBH, Germany); intra-assay CV was 14.3% and inter-assay CV was 13.6%, for this kit. The sensitivity for PTH assessment was 1.57 pg/ml. Fasting insulin levels were determined by Monobind kit (Monobind ELISA kit, Accubind, USA); intra-assay CV for this kit was 5.6% while inter-assay CV was 7.3%. For fasting insulin level assessment, the sensitivity was 0.75 µIU/ml. Glucose-oxidase method was used to determine the fasting serum glucose levels. Homeostasis model assessment for insulin resistance (HOMA-IR), as an indicator of insulin resistance, was calculated using the following formula: fasting glucose (mg/dL) X fasting insulin (μU/mL)/405. The collected data were analysed using the statistical package for the social sciences (SPSS 23) (IBM, USA). Independent student’s t test was used to test the differences in the mean values for the continuous variables. Chi-square test was used to test the differences in the proportion of the categorical variables. Multivariate logistic regression was applied to evaluate the association between vitamin D, PTH and insulin resistance in the occurrence of NAFLD by adjusting for the independent predictors. The Pearson correlation coefficient (r) was used to assess the correlation between variables. Statistical significance was set at P < 0.05.
3. Results
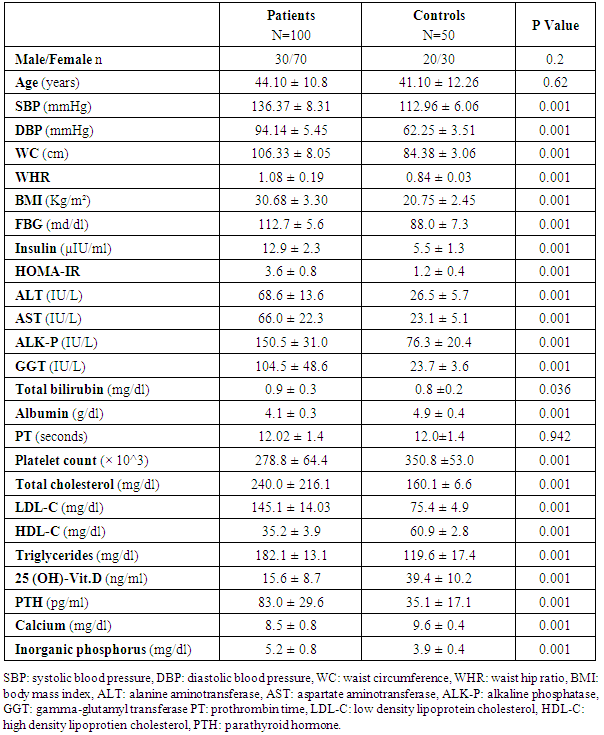
- Comparison between the studied groups regarding clinical and laboratory data is shown in table-1. Of note, HOMA-IR and PTH were significantly elevated and serum 25 (OH) D was significantly lowered in NAFLD patients when compared to healthy subjects.
|
|
|
|
4. Discussion
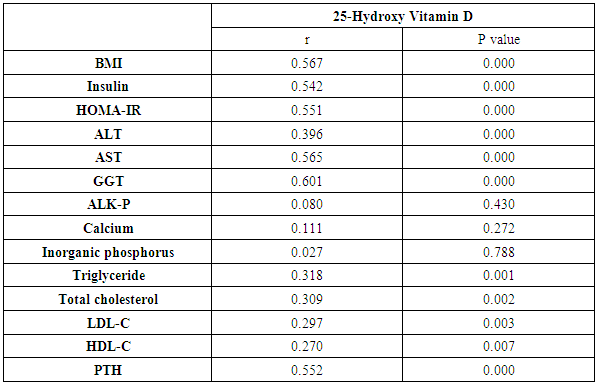
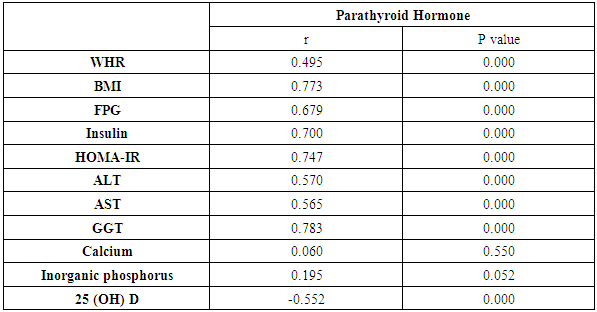
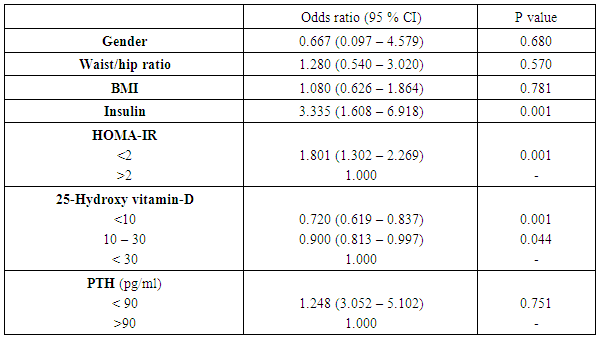
- This study proved that individuals affected by NAFLD have lowered serum 25 hydroxy vitamin D levels compared to control subjects without NAFLD. Decreased levels of serum 25-hydroxy vitamin D were independently related with an elevated oppurtunities of NAFLD. Prior studies have shown an almost identical result. Barchetta et al. [11] found that low 25(OH) vitamin D levels are independently associated with the existence of NAFLD regardless metabolic syndrome, diabetes and insulin-resistance. Also, Rhee et al. [12] studied the association of serum 25(OH) D levels with NAFLD and found that subjects with higher serum 25(OH)D showed a significantly lower risk for NAFLD compared to those with low serum 25(OH)D3 levels; a finding unrelated to obesity and metabolic syndrome. In another study, vitamin D concentration was significantly decreased in NAFLD patients compared to healthy controls. Moreover, low plasma vitamin D concentration could independently predict NAFLD advancement [13]. In this study, our results showed that HOMA-IR and serum insulin levels were significantly elevated in patients with NAFLD if compared to controls. Elevated HOMA scores were independently related to increased odds of NAFLD. Similar findings have previously been reported. Loomba et al. [2] demonstrated a significant relationship between NAFLD and liver biomarkers with the parameters of insulin resistance. Also, El-Karaksy et al. [14] found that IR was significantly associated with NAFLD. Insulin resistance, a key risk predictor in the pathogenesis of NAFLD, is related to the occurrence of oxidative stress and lipotoxicity. It is now accepted that proinflammatory cytokines such as TNF-α and IL-6, and adipokines such as adiponectin and leptin play a critical role in the advancement of steatosis to steatohepatitis [15]. In the present study, the relationship between 25-Hydroxy vitamin D, parathyroid hormone and insulin resistance was evaluated. It was demonstrated that serum 25 (OH) D level was negatively correlated with HOMA-IR scores in NAFLD patients (r=-0.551, P=0.000) and positively correlated with HOMA-IR in control group (r=0.363, P=0.009). The association between serum PTH and HOMA-IR was significantly positive in NAFLD patients (r: 0.747 p=0.000) while this correlation was non-significant among the healthy subjects. Kayaniyil et al. [16] study on obese individuals with NAFLD revealed a relationship between insufficient vitamin D status and low insulin sensitivity. In contrast, Bril et al. [17] failed to prove a relation between plasma vitamin D levels and insulin resistance, amount of liver fat accumulation, or the severity of NASH. In this study, there were statistically significant higher levels of parathyroid hormone in patients with NAFLD than in the control group. However, using multivariate analysis, PTH was not decided as a predictor of NAFLD development inspite of the positive correlation of it with 25-hydroxy vitamin D, serum insulin, and HOMA-IR. In contradiction with these results, the study by Bhatt et al. [10] who analysed the associations of 25 hydroxy vitamin D [25(OH) D] and parathyroid hormone (PTH) levels with clinical, anthropometric, biochemical and body composition parameters in Asian Indians with NAFLD, they concluded that low serum 25(OH) D and high PTH levels were independently associated with the presence of NAFLD. However, our findings are consistently correlated with [18], who revealed that decreased levels of 25-hydroxy vitamin D, elevated HOMA indices, but not parathyroid hormone (PTH) were independently associated with increased risk of NAFLD.Our study had some limitations. First, exposure of our patients to sunlight, as a source of vitamin D, was not assessed to evaluate the impact of sufficiency of vitamin D on improvement of NAFLD. Second, dietary supplementations were not examined in our study to evaluate their impact on vitamin D status in NAFLD patients. Third, although abdominal ultrasonography is a practical approach commonly used to detect liver steatosis, it is not the gold standard method for quantitative hepatic fat assessment. Fourth, factors known to influence the serum levels of vitamin D, PTH and, insulin, were considered as confounders in this study; however, it is possible that some unknown predictors also influence these results. Our study had some strengths. This case-control study was one of few studies to investigate the association between both vitamin D and PTH, with NAFLD. Similar previous studies had not examined these two factors simultaneously in relation to fatty liver.
5. Conclusions
- Serum concentrations of vitamin D, insulin and HOMA-IR index are independent predictors of NAFLD risk. Serum PTH concentration inspite of its elevation had nonsignificant relationship with NAFLD. Further studies may be required in the future to investigate the role of vitamin D supplementation for treatment or even prevention of NAFLD.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML